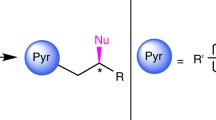
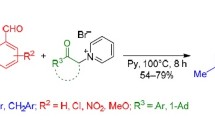
Several synthetic approaches are known for the preparation of chiral 4-aryl-3,4-dihydrocoumarins (DHCs). This article considers recently discovered methods for the synthesis of chiral DHCs: enantio-selective Rh(I)-catalyzed 1,4-addition of arylboronic acids to α,β-unsaturated carbonyl compounds, asymmetric reduction of coumarins, oxidation of chiral indanones, hydroesterification of alkenylphenols, and the addition of 1,3-dicarbonyl compounds to o-quinone methides.

Similar content being viewed by others
References
Wungsintaweekul, B.; Umehara, K.; Miyase, T.; Noguchi, H. Phytochemistry 2011, 72, 495.
Zhang, X.-F.; Wang, H.-M.; Song, Y.-L.; Nie, L.-H.; Wang, L.-F.; Lui, B.; Shen, P.-P.; Lui, Y. Bioorg. Med. Chem. Lett. 2006, 16, 949.
Wu, P.-L.; Hsu, Y.-L.; Zao, C.-W.; Damu, A. G.; Wu, T.-S. J. Nat. Prod. 2005, 68, 1180.
Seo, E.-K.; Wani, M. C.; Wall, M. E.; Navarro, H.; Mukherjee, R.; Farnsworth, N. R.; Kinghorn, A. D. Phytochemistry 2000, 55, 35.
Murata, T.; Miyase, T.; Yoshizaki, F. Chem. Pharm. Bull. 2011, 59, 88.
Khan, P. M.; El-Gendy, B. E.-D. M.; Kumar, N.; Garcia-Ordonez, R.; Lin, L.; Ruiz, C. H.; Cameron, M. D.; Griffin, P. R.; Kamenecka, T. M. Bioorg. Med. Chem. Lett. 2013, 23, 532.
(a) Gillberg, P.-G.; Sundquist, S.; Nilvebrant, L. Eur. J. Pharmacol. 1998, 349, 285. (b) Breitenbach, A.; Meese, C.; Wolff, H.-M.; Drews, R. WO Patent 2004089872.
Li, J.; Chang, W.; Ren, W.; Liu, W.; Wanga, H.; Shi, Y. Org. Biomol. Chem. 2015, 13, 10341.
Wang, H.; Dong, B.; Wang, Y.; Li, J.; Shi, Y. Org. Lett. 2014, 16, 186.
Gallagher, B. D.; Taft, B. R.; Lipshutz, B. H. Org. Lett. 2009, 11, 5374.
Barancelli, D. A.; Salles, A. G.; Taylor, J. G.; Correia, C. R. D. Org. Lett. 2012, 14, 6036.
Defieber, C.; Paquin, J.-F.; Serna, S.; Carreira, E. M. Org. Lett. 2004, 6, 3873.
Luo Y.; Carnell, A. J. Angew. Chem., Int. Ed. 2010, 49, 2750.
Colombo, L.; Rossi, R.; Allegrini, P.; Castaldi, G. EP Patent 1584621.
Chen, G.; Tokunaga, N.; Hayashi, T. Org. Lett. 2005, 7, 2285.
Korenaga, T.; Maenishi, R.; Osaki, K.; Sakai, T. Heterocycles 2010, 80, 157.
Mino, T.; Miura, K.; Taguchi, H.; Watanabe, K.; Sakamoto, M. Tetrahedron: Asymmetry 2015, 26, 1065.
Park, J. O.; Youn, S. W. Org. Lett. 2010, 12, 2258.
Brönnimann, R.; Chun, S.; Marti, R. Helv. Chim. Acta 2012, 95, 1809.
Wei, W.-T.; Yeh, J.-Y.; Kuo, T.-S.; Wu, H.-L. Chem.–Eur. J. 2011, 17, 11405.
Allen, J. C.; Kociok-Köhn, G.; Frost, C. G. Org. Biomol. Chem. 2012, 10, 32.
Yue, G.; Lei, K.; Hirao, H.; Zhou, J. Angew. Chem., Int. Ed. 2015, 54, 6531.
Caruana, L.; Mondatori, M.; Corti, V.; Morales, S.; Mazzanti, A.; Fochi, M.; Bernardi, L. Chem.–Eur. J. 2015, 21, 6037.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2016, 52(8), 527–529
Rights and permissions
About this article
Cite this article
Leitis, Z. Synthesis of enantiomerically enriched 4-aryl-3,4-dihydrocoumarins (microreview). Chem Heterocycl Comp 52, 527–529 (2016). https://doi.org/10.1007/s10593-016-1922-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-016-1922-7