Abstract
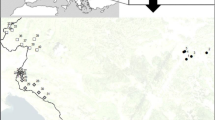
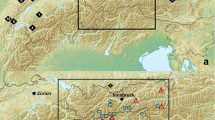
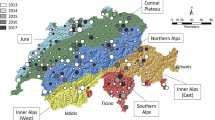
Niphanda fusca is an endangered myrmecophilous butterfly inhabiting environments at early stages of succession. Most of its habitats are places where succession is prevented by human activity. In some places, however, N. fusca lives in natural semi-bare areas, such as cliffs in mountains or grasslands around volcanos. We investigate the genetic structure of N. fusca in Japan and South Korea to address two questions. (1) Are populations in natural environments genetically different from those in secondary environments? and (2) Do populations in natural environments possess greater genetic diversity than those in secondary environments? The AMOVA results indicated that the populations in natural environments and those in secondary environments were differentiated to some extent; however, more than 80 % of genetic variation was found to occur within the same habitat type and within each population. We found no differences in genetic diversity between populations in the two environments. At present, we have not found a strong reason to consider populations in the two environments as different evolutionarily significant units. We think it is practical to conserve populations in natural environments at first, because in this case we need not manage habitats to protect N. fusca. We have only to inhibit habitat destruction. In contrast, in order to conserve populations in secondary environments, we would have to continue managing the habitats. This is far more difficult than inhibiting habitat destruction.



Similar content being viewed by others
References
Als TD, Vila R, Kandul NP, Nash DR, Yen S-H, Hsu Y-F, Mignault AA, Boomsma JJ, Pierce NE (2004) The evolution of alternative parasitic life histories in large blue butterflies. Nature 432:386–390. doi:10.1038/nature03020
Bossart JL, Antwi JB (2013) Species-specific traits predict genetic structure but not genetic diversity of three fragmented Afrotropical forest butterfly species. Conserv Genet 14:511–528. doi:10.1007/s10592-012-0436-9
Clement M, Posada D, Crandall K (2000) TCS: a computer program to estimate gene genealogies. Mol Ecol 9:1657–1660. doi:10.1046/j.1365-294x.2000.01020.x
Crandall KA, Bininda-Emonds ORP, Mace GM, Wayne RK (2000) Considering evolutionary processes in conservation biology. Trends Ecol Evol 15:290–295. doi:10.1016/S0169-5347(00)01876-0
de Jong MA, Wahlberg N, van Eijk M, Brakefield PM, Zwaan BJ (2011) Mitochondrial DNA signature for range-wide populations of Bicyclus anynana suggests a rapid expansion from recent refugia. PLoS One 6:6. doi:10.1371/journal.pone.0021385
DeChaine EG, Martin AP (2004) Historic cycles of fragmentation and expansion in Parnassius smintheus (Papilionidae) inferred using mitochondrial DNA. Evolution 58:113–127. doi:10.1111/j.0014-3820.2004.tb01578.x
Downey MH, Nice CC (2013) A role for both ecology and geography as mechanisms of genetic differentiation in specialized butterflies. Evol Ecol 27:565–578. doi:10.1007/s10682-012-9626-7
Dupanloup I, Schneider S, Excoffier L (2002) A simulated annealing approach to define the genetic structure of populations. Mol Ecol 11:2571–2581
Eastwood R, Pierce NE, Kitching RL, Hughes JM (2006) Do ants enhance diversification in lycaenid butterflies? Phylogeographic evidence from a model myrmecophile, Jalmenus evagoras. Evolution 60:315–327
Excoffier L, Lischer HEL (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analysis under Linux and Windows. Mol Ecol Res 10:564–567. doi:10.1111/j.1755-0998.2010.02847.x
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131:479–491
Frankham R, Ballou JD, Briscoe DA (2002) Introduction to conservation genetics. Cambridge University Press, Cambridge
Fukuda H, Hama E, Kuzuya K, Takahashi A, Takahashi M, Tanaka B, Tanaka H, Wakabayashi M, Watanabe Y (1984) The pictorial guide to ecology of butterflies in Japan 4. Hoikusha, Osaka (in Japanese with English abstract)
Graham RI, Wilson K (2012) Male-killing Wolbachia and mitochondrial selective sweep in a migratory African insect. BMC Evol Biol 12:204. doi:10.1186/1471-2148-12-204
Hojo MK, Wada-Katsumata A, Akino T, Yamaguchi S, Ozaki M, Yamaoka R (2009) Chemical disguise as particular caste of host ants in the ant inquilines parasite Niphanda fusca (Lepidoptera: Lycaenidae). Proc R Soc Lond B 276:551–558. doi:10.1098/rspb.2008.1064
Imai H, Kihara A, Kondoh M et al (2004) Ants of Japan. Gakken, Tokyo (in Japanese)
Kira T (1971) Nature seen from ecological viewpoint. Kawadeshoboushinsha, Tokyo (in Japanese)
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948. doi:10.1093/bioinformatics/btm404
Laurance WF, Delamônica P, Laurance SG, Vasconcelos HL, Lovejoy TE (2000) Rainforest fragmentation kills big trees. Nature 404:836. doi:10.1038/35009032
Mano T, Fujii H (2009) Decline and conservation of butterflies and moths in Japan VI. The Lepidopterological Society of Japan, Tokyo (in Japanese)
Ministry of Environment of Japan (2012) Red list, 4th ed. http://www.env.go.jp/press/file_view.php?serial=20563&hou_id=15619 (in Japanese) Accessed 28 Jul 2013
Monteiro A, Pierce NE (2000) Phylogeny of Bicyclus (Lepidoptera: Nymphalidae) inferred from COI, COII, and EF-1α gene sequences. Mol Phyl Evol 18:264–281. doi:10.1006/mpev.2000.0872
Nakamura Y (2011) Conservation of butterflies in Japan: status, action and strategy. J Insect Conserv 15:5–22. doi:10.1007/s10841-010-9299-x
Narita S, Nomura M, Kato Y, Fukatsu T (2006) Genetic structure of sibling butterfly species affected by Wolbachia infection sweep: evolutionary and biogeographical implications. Mol Ecol 15:1095–1108. doi:10.1111/j.1365-294X.2006.02857.x
Nash DR, Als TD, Maile R, Jones GR, Boomsma JJ (2008) A mosaic of chemical coevolution in a large blue butterfly. Science 319:88–90. doi:10.1126/science.1149180
Nei M (1987) Molecular evolutionary genetics. Colombia University Press, New York
Neve G, Pavlicko A, Konvicka M (2009) Loss of genetic diversity through spontaneous colonization in the bog fritillary butterfly, Proclossiana eunomia (Lepidoptera: Nymphalidae) in the Czech Republic. Eur J Entomol 106:11–19
Pellissier L, Litsios G, Guisan A, Alvarez N (2012) Molecular substitution rate increases in myrmecophilous lycaenid butterflies (Lepidoptera). Zool Scr 41:651–658. doi:10.1111/j.1463-6409.2012.00556.x
Pierce NE, Braby MF, Heath A, Lohman DJ, Mathew J, Rand DB, Travassos MA (2002) The ecology and evolution of ant association in the Lycaenidae (Lepidoptera). Annu Rev Entomol 47:733–771. doi:10.1146/annurev.ento.47.091201.145257
Pimm SL, Jenkins CN, Abell R, Brooks TM, Gittleman JL, Joppa LN, Raven PH, Roberts CM, Sexton JO (2014) The biodiversity of species and their rates of extinction, distribution, and protection. Science 344:987. doi:10.1126/science.1246752
Sakamoto Y, Hirai N, Tanikawa T, Yago M, Ishii M (2015) Population genetic structure and Wolbachia infection in an endangered butterfly, Zizina emelina (Lepidoptera, Lycaenidae), in Japan. Bull Entomol Res. doi:10.1017/S0007485314000819
Sielezniew M, Ponikwicka-Tyszko D, Ratkiewicz M, Dziekańska I, Kostro-Ambroziak A, Rutkowski R (2011) Divergent patterns in the mitochondrial and nuclear diversity of the specialized butterfly Plebejus argus (Lepidoptera: Lycaenidae). Eur J Entomol 108:537–545
Sielezniew M, Rutkowski R, Ponikwicka-Tyszko D, Ratkiewicz M, Dziekańska I, Švitra G (2012) Differences in genetic variability between two ecotypes of the endangered myrmecophilous butterfly Phengaris (=Maculinea) alcon—the setting of conservation priorities. Insect Conserv Diver 5:223–236. doi:10.1111/j.1752-4598.2011.00163.x
Simon C, Frati F, Beckenbach AT, Crespi BJ, Liu H, Flook P (1994) Evolution, weighting, and phylogenetic utility of mitochondrial gene sequences and a compilation of conserved polymerase chain reaction primers. Ann Entomol Soc Am 87:651–701
Thomas JA, Simcox DJ, Clarke RT (2009) Successful conservation of a threatened Maculinea butterfly. Science 325:80–83. doi:10.1126/science.1175726
Ugelvig LV, Nielsen PS, Boomsma JJ, Nash DR (2011) Reconstructing eight decades of genetic variation in an isolated Danish population of the large blue butterfly Maculinea arion. BMC Evol Biol 11:201. doi:10.1186/1471-2148-11-201
Washitani I (2001) Traditional sustainable ecosystem ‘SATOYAMA’ and biodiversity crisis in Japan: conservation ecological perspective. Global Environ Res 5:119–133
Wu L-W, Yen S-H, Lees DC, Hsu Y-F (2010) Elucidating genetic signatures of native and introduced populations of the Cycad Blue, Chilades pandava to Taiwan: a threat both to Sago Palm and to native Cycas populations worldwide. Biol Invasions 12:2649–2669. doi:10.1007/s10530-009-9672-4
Yagi T, Sakai G, Takebe H (1999) Phylogeny of Japanese Papilionid butterflies inferred from nucleotide sequences of the mitochondrial ND5 gene. J Mol Evol 48:42–48. doi:10.1007/PL00006443
Acknowledgments
We are grateful to S. Asano, H. Fukuda, T. Ichikawa, H. Inoue, K. Kamata, T. Kobayashi, C. Kudo, T. Kumon, Y. Matsuoka, H. Nagayama, T. Nishiguchi, H. Oh, T. Tani, and O. Yamamoto for collecting samples. We are also grateful to T. Okamoto for valuable comments on the manuscript. This work was supported by a grant from the Fujiwara Natural History Foundation, a grant for start up for young scientists of Kyoto University, Global COE program A06 of Kyoto University, and MEXT Grants for Excellent Graduate Schools.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takeuchi, T., Takahashi, J., Kiyoshi, T. et al. Genetic differentiation in the endangered myrmecophilous butterfly Niphanda fusca: a comparison of natural and secondary habitats. Conserv Genet 16, 979–986 (2015). https://doi.org/10.1007/s10592-015-0717-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-015-0717-1