Abstract
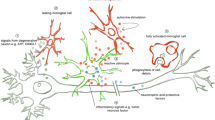
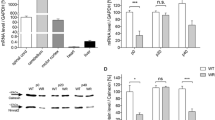
The Wobbler mouse is an accepted model of sporadic amyotrophic lateral sclerosis. The spinal cord of clinically symptomatic animals (3–5 months old) shows vacuolar motoneuron degeneration, inflammation, and gliosis accompanied by motor impairment. However, data are not conclusive concerning pathological changes appearing early after birth. To answer this question, we used postnatal day (PND) 6 genotyped Wobbler pups to determine abnormalities of glia and neurons at this early age period in the spinal cord. We found astrogliosis, microgliosis with morphophenotypic changes pointing to active ameboid microglia, enhanced expression of the proinflammatory markers TLR4, NFkB, TNF, and inducible nitric oxide synthase. The astrocytic enzyme glutamine synthase and the glutamate-aspartate transporter GLAST were also reduced in PND 6 Wobbler pups, suggesting excitotoxicity due to impaired glutamate homeostasis. At the neuronal level, PND 6 Wobblers showed swollen soma, increased choline acetyltransferase immunofluorescence staining, and low expression of the neuronal nuclear antigen NeuN. However, vacuolated motoneurons, a typical signature of older clinically symptomatic Wobbler mice, were absent in the spinal cord of PND 6 Wobblers. The results suggest predominance of neuroinflammation and abnormalities of microglia and astrocytes at this early period of Wobbler life, accompanied by some neuronal changes. Data support the non-cell autonomous hypothesis of the Wobbler disorder, and bring useful information with regard to intervening molecular inflammatory mechanisms at the beginning stage of human motoneuron degenerative diseases.
Graphical Abstract





Similar content being viewed by others
Data Availability
Enquiries about data availability should be directed to the authors.
References
Ait-Ikhlef A, Hantaz-Ambroise D, Jacque C, Belkadi L, Rieger F (1999) Astrocyte proliferation induced by wobbler astrocyte conditioned medium is blocked by tumor necrosis factor-alpha (TNF-alpha) and interleukin-1beta (IL-1beta) neutralizing antibodies in vitro. Cell Mol Biol (noisy-Le-Grand) 45(4):393–400
Augustin M, Heimann P, Rathke S, Jockusch H (1997) Spinal muscular atrophy gene wobbler of the mouse: evidence from chimeric spinal cord and testis for cell-autonomous function. Dev Dyn 209(3):286–295. https://doi.org/10.1002/(SICI)1097-0177(199707)209:3%3c286
Beers DR, Appel SH (2019) Immune dysregulation in amyotrophic lateral sclerosis: mechanisms and emerging therapies. Lancet Neurol 18(2):211–220. https://doi.org/10.1016/S1474-4422(18)30394-6
Bensimon G, Ludolph A, Agid Y, Vidailhet M, Payan C, Leigh PN, Group NS (2009) Riluzole treatment, survival and diagnostic criteria in Parkinson plus disorders: the NNIPPS study. Brain 132(Pt 1):156–171. https://doi.org/10.1093/brain/awn291
Blondet B, Hantaz-Ambroise D, Ait-Ikhlef A, Cambier D, Murawsky M, Rieger F (1995) Astrocytosis in wobbler mouse spinal cord involves a population of astrocytes which is glutamine synthetase-negative. Neurosci Lett 183(3):179–182
Blondet B, Carpentier G, Ait-Ikhlef A, Murawsky M, Rieger F (2002) Motoneuron morphological alterations before and after the onset of the disease in the wobbler mouse. Brain Res 930(1–2):53–57. https://doi.org/10.1016/s0006-8993(01)03405-9
Boillee S, Viala L, Peschanski M, Dreyfus PA (2001) Differential microglial response to progressive neurodegeneration in the murine mutant Wobbler. Glia 33(4):277–287
Boillee S, Peschanski M, Junier MP (2003) The wobbler mouse: a neurodegeneration jigsaw puzzle. Mol Neurobiol 28(1):65–106. https://doi.org/10.1385/MN:28:1:65
Brenner D, Freischmidt A (2022) Update on genetics of amyotrophic lateral sclerosis. Curr Opin Neurol 35(5):672–677. https://doi.org/10.1097/WCO.0000000000001093
Brettschneider J, Toledo JB, Van Deerlin VM, Elman L, McCluskey L, Lee VM, Trojanowski JQ (2012) Microglial activation correlates with disease progression and upper motor neuron clinical symptoms in amyotrophic lateral sclerosis. PLoS ONE. https://doi.org/10.1371/journal.pone.0039216
Cihankaya H, Theiss C, Matschke V (2021) Little helpers or mean rogue-role of microglia in animal models of amyotrophic lateral sclerosis. Int J Mol Sci. https://doi.org/10.3390/ijms22030993
Crain JM, Nikodemova M, Watters JJ (2013) Microglia express distinct M1 and M2 phenotypic markers in the postnatal and adult central nervous system in male and female mice. J Neurosci Res 91(9):1143–1151. https://doi.org/10.1002/jnr.23242
Dahlke C, Saberi D, Ott B, Brand-Saberi B, Schmitt-John T, Theiss C (2015) Inflammation and neuronal death in the motor cortex of the wobbler mouse, an ALS animal model. J Neuroinflammation 12:215. https://doi.org/10.1186/s12974-015-0435-0
Dave KR, Bradley WG, Perez-Pinzon MA (2003) Early mitochondrial dysfunction occurs in motor cortex and spinal cord at the onset of disease in the Wobbler mouse. Exp Neurol 182(2):412–420. https://doi.org/10.1016/s0014-4886(03)00091-8
De Nicola AF, Coronel F, Garay LI, Gargiulo-Monachelli G, Gonzalez Deniselle MC, Gonzalez SL, Labombarda F, Meyer M, Guennoun R, Schumacher M (2013) Therapeutic effects of progesterone in animal models of neurological disorders. CNS Neurol Disord: Drug Targets 12(8):1205–1218
De Nicola AF, Meyer M, Guennoun R, Schumacher M, Hunt H, Belanoff J, de Kloet ER, Gonzalez Deniselle MC (2020) Insights into the therapeutic potential of glucocorticoid receptor modulators for neurodegenerative diseases. Int J Mol Sci 21(6):10. https://doi.org/10.3390/ijms21062137
De Paola M, Mariani A, Bigini P, Peviani M, Ferrara G, Molteni M, Gemma S, Veglianese P, Castellaneta V, Boldrin V, Rossetti C, Chiabrando C, Forloni G, Mennini T, Fanelli R (2012) Neuroprotective effects of toll-like receptor 4 antagonism in spinal cord cultures and in a mouse model of motor neuron degeneration. Mol Med 18:971–981. https://doi.org/10.2119/molmed.2012.00020
Diana V, Ottolina A, Botti F, Fumagalli E, Calcagno E, De Paola M, Cagnotto A, Invernici G, Parati E, Curti D, Mennini T (2010) Neural precursor-derived astrocytes of wobbler mice induce apoptotic death of motor neurons through reduced glutamate uptake. Exp Neurol 225(1):163–172. https://doi.org/10.1016/j.expneurol.2010.06.008
Dockery P, Tang Y, Morais M, Vacca-Galloway LL (1997) Neuron volume in the ventral horn in Wobbler mouse motoneuron disease: a light microscope stereological study. J Anat 191(Pt 1):89–98. https://doi.org/10.1046/j.1469-7580.1997.19110089.x
Duchen LW, Strich SJ (1968) An hereditary motor neurone disease with progressive denervation of muscle in the mouse: the mutant “wobbler.” J Neurol Neurosurg Psychiatry 31(6):535–542. https://doi.org/10.1136/jnnp.31.6.535
Dukkipati SS, Garrett TL, Elbasiouny SM (2018) The vulnerability of spinal motoneurons and soma size plasticity in a mouse model of amyotrophic lateral sclerosis. J Physiol 596(9):1723–1745. https://doi.org/10.1113/JP275498
Filipi T, Hermanova Z, Tureckova J, Vanatko O, Anderova AM (2020) Glial cells-the strategic targets in amyotrophic lateral sclerosis treatment. J Clin Med. https://doi.org/10.3390/jcm9010261
Frank-Cannon TC, Alto LT, McAlpine FE, Tansey MG (2009) Does neuroinflammation fan the flame in neurodegenerative diseases? Mol Neurodegener 4:47. https://doi.org/10.1186/1750-1326-4-47
Fricker M, Tolkovsky AM, Borutaite V, Coleman M, Brown GC (2018) Neuronal cell death. Physiol Rev 98(2):813–880. https://doi.org/10.1152/physrev.00011.2017
Friese A, Kaltschmidt JA, Ladle DR, Sigrist M, Jessell TM, Arber S (2009) Gamma and alpha motor neurons distinguished by expression of transcription factor Err3. Proc Natl Acad Sci USA 106(32):13588–13593. https://doi.org/10.1073/pnas.0906809106
Garay LI, Gonzalez Deniselle MC, Brocca ME, Lima A, Roig P, De Nicola AF (2012) Progesterone down-regulates spinal cord inflammatory mediators and increases myelination in experimental autoimmune encephalomyelitis. Neuroscience 226:40–50. https://doi.org/10.1016/j.neuroscience.2012.09.032
Gargiulo-Monachelli G, Meyer M, Lara A, Garay L, Lima A, Roig P, De Nicola AF, Gonzalez Deniselle MC (2019) Comparative effects of progesterone and the synthetic progestin norethindrone on neuroprotection in a model of spontaneous motoneuron degeneration. J Steroid Biochem Mol Biol. https://doi.org/10.1016/j.jsbmb.2019.105385
Gibson SB, Figueroa KP, Bromberg MB, Pulst SM, Cannon-Albright L (2014) Familial clustering of ALS in a population-based resource. Neurology 82(1):17–22. https://doi.org/10.1212/01.wnl.0000438219.39061.da
Gonzalez Deniselle MC, Lopez-Costa JJ, Saavedra JP, Pietranera L, Gonzalez SL, Garay L, Guennoun R, Schumacher M, De Nicola AF (2002) Progesterone neuroprotection in the Wobbler mouse, a genetic model of spinal cord motor neuron disease. Neurobiol Dis 11(3):457–468
Gonzalez Deniselle MC, Garay L, Lopez-Costa JJ, Gonzalez S, Mougel A, Guennoun R, Schumacher M, De Nicola AF (2004) Progesterone treatment reduces NADPH-diaphorase/nitric oxide synthase in Wobbler mouse motoneuron disease. Brain Res 1014(1–2):71–79. https://doi.org/10.1016/j.brainres.2004.04.004
Gonzalez Deniselle MC, Garay L, Gonzalez S, Saravia F, Labombarda F, Guennoun R, Schumacher M, De Nicola AF (2007) Progesterone modulates brain-derived neurotrophic factor and choline acetyltransferase in degenerating Wobbler motoneurons. Exp Neurol 203(2):406–414. https://doi.org/10.1016/j.expneurol.2006.08.019
Gonzalez Deniselle MC, Carreras MC, Garay L, Gargiulo-Monachelli G, Meyer M, Poderoso JJ, De Nicola AF (2012) Progesterone prevents mitochondrial dysfunction in the spinal cord of wobbler mice. J Neurochem 122(1):185–195. https://doi.org/10.1111/j.1471-4159.2012.07753.x
Hall ED, Andrus PK, Oostveen JA, Fleck TJ, Gurney ME (1998) Relationship of oxygen radical-induced lipid peroxidative damage to disease onset and progression in a transgenic model of familial ALS. J Neurosci Res 53(1):66–77. https://doi.org/10.1002/(SICI)1097-4547(19980701)53:1%3c66::AID-JNR7%3e3.0.CO;2-H
Hantaz-Ambroise D, Jacque C, Ait Ikhlef A, Parmentier C, Leclerc P, Cambier D, Zadigue G, Rieger F (2001) Specific features of chronic astrocyte gliosis after experimental central nervous system (CNS) xenografting and in Wobbler neurological mutant CNS. Differentiation 69(2–3):100–107
Julien JP, Kriz J (2006) Transgenic mouse models of amyotrophic lateral sclerosis. Biochem Biophys Acta 1762(11–12):1013–1024. https://doi.org/10.1016/j.bbadis.2006.03.006
Kiernan MC, Vucic S, Cheah BC, Turner MR, Eisen A, Hardiman O, Burrell JR, Zoing MC (2011) Amyotrophic lateral sclerosis. Lancet 377(9769):942–955. https://doi.org/10.1016/S0140-6736(10)61156-7
Kishino A, Ishige Y, Tatsuno T, Nakayama C, Noguchi H (1997) BDNF prevents and reverses adult rat motor neuron degeneration and induces axonal outgrowth. Exp Neurol 144(2):273–286. https://doi.org/10.1006/exnr.1996.6367
Laage S, Zobel G, Jockusch H (1988) Astrocyte overgrowth in the brain stem and spinal cord of mice affected by spinal atrophy, wobbler. Dev Neurosci 10(3):190–198
Labombarda F, Gonzalez SL, Gonzalez DM, Guennoun R, Schumacher M, De Nicola AF (2002) Cellular basis for progesterone neuroprotection in the injured spinal cord. J Neurotrauma 19:343–355. https://doi.org/10.1089/089771502753594918
Lasiene J, Yamanaka K (2011) Glial cells in amyotrophic lateral sclerosis. Neurol Res Int 2011:718987. https://doi.org/10.1155/2011/718987
Levine JB, Kong J, Nadler M, Xu Z (1999) Astrocytes interact intimately with degenerating motor neurons in mouse amyotrophic lateral sclerosis (ALS). Glia 28(3):215–224
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
MacLean M, Juranek J, Cuddapah S, Lopez-Diez R, Ruiz HH, Hu J, Frye L, Li H, Gugger PF, Schmidt AM (2021) Microglia RAGE exacerbates the progression of neurodegeneration within the SOD1(G93A) murine model of amyotrophic lateral sclerosis in a sex-dependent manner. J Neuroinflammation 18(1):139. https://doi.org/10.1186/s12974-021-02191-2
Meyer M, Gonzalez Deniselle MC, Garay LI, Monachelli GG, Lima A, Roig P, Guennoun R, Schumacher M, De Nicola AF (2010) Stage dependent effects of progesterone on motoneurons and glial cells of wobbler mouse spinal cord degeneration. Cell Mol Neurobiol 30(1):123–135. https://doi.org/10.1007/s10571-009-9437-8
Meyer M, Gonzalez Deniselle MC, Gargiulo-Monachelli G, Garay LI, Schumacher M, Guennoun R, De Nicola AF (2012) Progesterone effects on neuronal brain-derived neurotrophic factor and glial cells during progression of Wobbler mouse neurodegeneration. Neuroscience 201:267–279. https://doi.org/10.1016/j.neuroscience.2011.11.034
Meyer M, Gonzalez Deniselle MC, Garay L, Sitruk-Ware R, Guennoun R, Schumacher M, De Nicola AF (2015) The progesterone receptor agonist Nestorone holds back proinflammatory mediators and neuropathology in the wobbler mouse model of motoneuron degeneration. Neuroscience 308:51–63. https://doi.org/10.1016/j.neuroscience.2015.09.007
Meyer M, Lara A, Hunt H, Belanoff J, de Kloet ER, Gonzalez Deniselle MC, De Nicola AF (2018) The selective glucocorticoid receptor modulator Cort 113176 reduces neurodegeneration and neuroinflammation in Wobbler mice spinal cord. Neuroscience 384:384–396. https://doi.org/10.1016/j.neuroscience.2018.05.042
Mora JS (2017) Edaravone for treatment of early-stage ALS. Lancet Neurol 16(10):772. https://doi.org/10.1016/S1474-4422(17)30289-2
Moser JM, Bigini P, Schmitt-John T (2013) The wobbler mouse, an ALS animal model. MGG 288(5–6):207–229. https://doi.org/10.1007/s00438-013-0741-0
Pirooznia SK, Dawson VL, Dawson TM (2014) Motor neuron death in ALS: programmed by astrocytes? Neuron 81(5):961–963. https://doi.org/10.1016/j.neuron.2014.02.024
Rathke-Hartlieb S, Schmidt VC, Jockusch H, Schmitt-John T, Bartsch JW (1999) Spatiotemporal progression of neurodegeneration and glia activation in the wobbler neuropathy of the mouse. NeuroReport 10(16):3411–3416
Rizzo F, Riboldi G, Salani S, Nizzardo M, Simone C, Corti S, Hedlund E (2014) Cellular therapy to target neuroinflammation in amyotrophic lateral sclerosis. CMLS 71(6):999–1015. https://doi.org/10.1007/s00018-013-1480-4
Santoro B, Bigini P, Levandis G, Nobile V, Biggiogera M, Botti F, Mennini T, Curti D (2004) Evidence for chronic mitochondrial impairment in the cervical spinal cord of a murine model of motor neuron disease. Neurobiol Dis 17(2):349–357. https://doi.org/10.1016/j.nbd.2004.07.003
Schmitt-John T (2015) VPS54 and the wobbler mouse. Front Neurosci 9:381. https://doi.org/10.3389/fnins.2015.00381
Trias E, Ibarburu S, Barreto-Nunez R, Barbeito L (2017) Significance of aberrant glial cell phenotypes in pathophysiology of amyotrophic lateral sclerosis. Neurosci Lett 636:27–31. https://doi.org/10.1016/j.neulet.2016.07.052
Tsuzaka K, Ishiyama T, Pioro EP, Mitsumoto H (2001) Role of brain-derived neurotrophic factor in wobbler mouse motor neuron disease. Muscle Nerve 24(4):474–480. https://doi.org/10.1002/mus.1029
Van Harten ACM, Phatnani H, Przedborski S (2021) Non-cell-autonomous pathogenic mechanisms in amyotrophic lateral sclerosis. Trends Neurosci 44(8):658–668. https://doi.org/10.1016/j.tins.2021.04.008
Vargas MR, Johnson JA (2010) Astrogliosis in amyotrophic lateral sclerosis: role and therapeutic potential of astrocytes. Neurotherapeutics 7(4):471–481. https://doi.org/10.1016/j.nurt.2010.05.012
Yang EJ, Kim SH, Yang SC, Lee SM, Choi SM (2011) Melittin restores proteasome function in an animal model of ALS. J Neuroinflammation 8:69. https://doi.org/10.1186/1742-2094-8-69
Acknowledgements
This work was supported by grants from the National Research Council of Argentina (PIP 2017-2019 #11220170100002CO and PIP 2017-2019 # 11220170100301CO), the Ministry of Science and Technology of Argentina (PICT 2017 Nº 1150 and PICT 2019 Nº 3292), the University of Buenos Aires (#20020170100224BA) and Rene Baron, Williams and Roemmers Foundations of Argentina. These funding sources did not have a role in the collection or analysis of data, writing of this manuscript or submission for publication.
Author information
Authors and Affiliations
Contributions
MM and MCGD performed the experiments, carried out statistical analysis, and made substantial contributions to interpretation of the data. AL provided technical support. MCGD and AFDeN designed the experiments and performed writing and editing of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Consent to Participate
All authors have given their consent to participate.
Ethical Approval
Animal procedures were approved by the Institutional Animal Care and Use Committee (IACUC) following the Guide for the Care and Use of Laboratory Animals (Animal Welfare Assurance, NIH certificate # F16-00065 A5072-01). Every effort was made to avoid animal suffering and treatments were supervised by IACUC personnel.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Meyer, M., Lima, A., Deniselle, M.C.G. et al. Early Signs of Neuroinflammation in the Postnatal Wobbler Mouse Model of Amyotrophic Lateral Sclerosis. Cell Mol Neurobiol 43, 2149–2163 (2023). https://doi.org/10.1007/s10571-022-01294-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-022-01294-5