Abstract
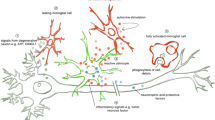
Amyotrophic lateral sclerosis is a devastating motor neuron disease and to this day not curable. While 5–10% of patients inherit the disease (familiar ALS), up to 95% of patients are diagnosed with the sporadic form (sALS). ALS is characterized by the degeneration of upper motor neurons in the cerebral cortex and of lower motor neurons in the brainstem and spinal cord. The wobbler mouse resembles almost all phenotypical hallmarks of human sALS patients and is therefore an excellent motor neuron disease model. The motor neuron disease of the wobbler mouse develops over a time course of around 40 days and can be divided into three phases: p0, presymptomatic; p20, early clinical; and p40, stable clinical phase. Recent findings suggest an essential implication of the NAD+-producing enzyme Nmnat2 in neurodegeneration as well as maintenance of healthy axons. Here, we were able to show a significant downregulation of both gene and protein expression of Nmnat2 in the spinal cord of the wobbler mice at the stable clinical phase. The product of the enzyme NAD+ is also significantly reduced, and the values of the reactive oxygen species are significantly increased in the spinal cord of the wobbler mouse at p40. Thus, the deregulated expression of Nmnat2 appears to have a great influence on the cellular stress in the spinal cord of wobbler mice.





Similar content being viewed by others
References
Bruijn LI, Miller TM, Cleveland DW (2004) Unraveling the mechanisms involved in motor neuron degeneration in ALS. Annu Rev Neurosci 27:723–749. https://doi.org/10.1146/annurev.neuro.27.070203.144244
Renton AE, Chio A, Traynor BJ (2014) State of play in amyotrophic lateral sclerosis genetics. Nat Neurosci 17(1):17–23. https://doi.org/10.1038/nn.3584
Cozzolino M, Carrì MT (2012) Mitochondrial dysfunction in ALS. The neurotoxicity of mutant proteins: 20 years after the discovery of the first mutant gene involved in neurodegeneration 97(2): 54–66. doi: https://doi.org/10.1016/j.pneurobio.2011.06.003
Kiernan MC, Vucic S, Cheah BC, Turner MR, Eisen A, Hardiman O, Burrell JR, Zoing MC (2011) Amyotroph Lateral Scler. Lancet 377(9769):942–955. https://doi.org/10.1016/S0140-6736(10)61156-7
Chen S, Sayana P, Zhang X, le W (2013) Genetics of amyotrophic lateral sclerosis: An update. Mol Neurodegener 8:28. https://doi.org/10.1186/1750-1326-8-28
Alami NH, Smith RB, Carrasco MA, Williams LA, Winborn CS, Han SSW, Kiskinis E, Winborn B et al (2014) Axonal transport of TDP-43 mRNA granules is impaired by ALS-causing mutations. Neuron 81(3):536–543. https://doi.org/10.1016/j.neuron.2013.12.018
Evans MC, Couch Y, Sibson N, Turner MR (2013) Inflammation and neurovascular changes in amyotrophic lateral sclerosis. Neuroinflammation Neurodegener Neurodysfunct 53:34–41. https://doi.org/10.1016/j.mcn.2012.10.008
Tadic V, Prell T, Lautenschlaeger J, Grosskreutz J (2014) The ER mitochondria calcium cycle and ER stress response as therapeutic targets in amyotrophic lateral sclerosis. Front Cell Neurosci 8:147. https://doi.org/10.3389/fncel.2014.00147
Ott B, Dahlke C, Meller K, Napirei M, Schmitt-John T, Brand-Saberi B, Theiss C, Saberi D (2015) Implementation of a manual for working with wobbler mice and criteria for discontinuation of the experiment. Ann Anat 200:118–124. https://doi.org/10.1016/j.aanat.2015.03.007
Dahlke C, Saberi D, Ott B, Brand-Saberi B, Schmitt-John T, Theiss C (2015) Inflammation and neuronal death in the motor cortex of the wobbler mouse, an ALS animal model. J Neuroinflammation 12:215. https://doi.org/10.1186/s12974-015-0435-0
Saberi D, Ott B, Dahlke C, Matschke V, Schmitt-John T, Theiss C (2016) The spatiotemporal pattern of degeneration in the cerebellum of the wobbler mouse. J Neuropathol Exp Neurol 75(4):347–357. https://doi.org/10.1093/jnen/nlw005
Duchen LW, Strich SJ (1968) An hereditary motor neurone disease with progressive denervation of muscle in the mouse: the mutant ‘wobbler’. J Neurol Neurosurg Psychiatry 31(6):535–542
Schmitt-John T, Drepper C, Mussmann A et al (2005) Mutation of Vps54 causes motor neuron disease and defective spermiogenesis in the wobbler mouse. Nat Genet 37(11):1213–1215. https://doi.org/10.1038/ng1661
Ferraiuolo L, Kirby J, Grierson AJ, Sendtner M, Shaw PJ (2011) Molecular pathways of motor neuron injury in amyotrophic lateral sclerosis. Nat Rev Neurol 7(11):616–630. https://doi.org/10.1038/nrneurol.2011.152
Moser JM, Bigini P, Schmitt-John T (2013) The wobbler mouse, an ALS animal model. Mol Gen Genomics 288(5–6):207–229. https://doi.org/10.1007/s00438-013-0741-0
Achi EY, Rudnicki SA (2012) ALS and frontotemporal dysfunction: a review. Neurol Res Int 2012: 806306:1–9. https://doi.org/10.1155/2012/806306
Heimann P, Laage S, Jockusch H (1991) Defect of sperm assembly in a neurological mutant of the mouse, wobbler (WR). Differentiation 47(2):77–83
Verdin E (2015) NAD(+) in aging, metabolism, and neurodegeneration. Science 350(6265):1208–1213. https://doi.org/10.1126/science.aac4854
Coleman MP, Freeman MR (2010) Wallerian degeneration, Wld S, and Nmnat. Annu Rev Neurosci 33(1):245–267. https://doi.org/10.1146/annurev-neuro-060909-153248
Gilley J, Coleman MP, Barres BA (2010) Endogenous Nmnat2 is an essential survival factor for maintenance of healthy axons. PLoS Biol 8(1):e1000300. https://doi.org/10.1371/journal.pbio.1000300
Rueda CB, Llorente-Folch I, Traba J, Amigo I, Gonzalez-Sanchez P, Contreras L, Juaristi I, Martinez-Valero P et al (2016) Glutamate excitotoxicity and Ca2+-regulation of respiration: Role of the Ca2+ activated mitochondrial transporters (CaMCs). Biochim Biophys Acta 1857(8):1158–1166. https://doi.org/10.1016/j.bbabio.2016.04.003
Berger F, Lau C, Dahlmann M, Ziegler M (2005) Subcellular compartmentation and differential catalytic properties of the three human nicotinamide mononucleotide adenylyltransferase isoforms. J Biol Chem 280(43):36334–36341. https://doi.org/10.1074/jbc.M508660200
Houtkooper RH, Cantó C, Wanders RJ, Auwerx J (2010) The secret life of NAD +: an old metabolite controlling new metabolic signaling pathways. Endocr Rev 31(2):194–223. https://doi.org/10.1210/er.2009-0026
Yan T, Feng Y, Zheng J, Ge X, Zhang Y, Wu D, Zhao J, Zhai Q (2010) Nmnat2 delays axon degeneration in superior cervical ganglia dependent on its NAD synthesis activity. Neurochem Int 56(1):101–106. https://doi.org/10.1016/j.neuint.2009.09.007
Mayer PR, Huang N, Dewey CM, Dries DR, Zhang H, Yu G (2010) Expression, localization, and biochemical characterization of nicotinamide mononucleotide adenylyltransferase 2. J Biol Chem 285(51):40387–40396. https://doi.org/10.1074/jbc.M110.178913
Milde S, Fox AN, Freeman MR, Coleman MP (2013) Deletions within its subcellular targeting domain enhance the axon protective capacity of Nmnat2 in vivo. Sci Rep 3:2567. https://doi.org/10.1038/srep02567
Sasaki Y, Vohra BP, Baloh RH et al (2009) Transgenic mice expressing the Nmnat1 protein manifest robust delay in axonal degeneration in vivo. J Neurosci 29(20):6526–6534. https://doi.org/10.1523/JNEUROSCI.1429-09.2009
Yahata N, Yuasa S, Araki T (2009) Nicotinamide mononucleotide adenylyltransferase expression in mitochondrial matrix delays Wallerian degeneration. J Neurosci 29(19):6276–6284. https://doi.org/10.1523/JNEUROSCI.4304-08.2009
Gilley J, Orsomando G, Nascimento-Ferreira I, Coleman MP (2015) Absence of SARM1 rescues development and survival of NMNAT2-deficient axons. Cell Rep 10(12):1974–1981. https://doi.org/10.1016/j.celrep.2015.02.060
Coleman M (2005) Axon degeneration mechanisms: commonality amid diversity. Nat Rev Neurosci 6(11):889–898. https://doi.org/10.1038/nrn1788
Mack TG, Reiner M, Beirowski B et al (2001) Wallerian degeneration of injured axons and synapses is delayed by a Ube4b/Nmnat chimeric gene. Nat Neurosci 4(12):1199–1206. https://doi.org/10.1038/nn770
Conforti L, Gilley J, Coleman MP (2014) Wallerian degeneration: an emerging axon death pathway linking injury and disease. Nat Rev Neurosci 15(6):394–409. https://doi.org/10.1038/nrn3680
Harlan BA, Pehar M, Sharma DR, Beeson G, Beeson CC, Vargas MR (2016) Enhancing NAD+ salvage pathway reverts the toxicity of primary astrocytes expressing amyotrophic lateral sclerosis-linked mutant superoxide dismutase 1 (SOD1). J Biol Chem 291(20):10836–10846. https://doi.org/10.1074/jbc.M115.698779
Ferrante RJ, Browne SE, Shinobu LA, Bowling AC, Baik MJ, MacGarvey U, Kowall NW, Brown RH Jr et al (1997) Evidence of increased oxidative damage in both sporadic and familial amyotrophic lateral sclerosis. J Neurochem 69(5):2064–2074
Shaw PJ, Ince PG, Falkous G, Mantle D (1995) Oxidative damage to protein in sporadic motor neuron disease spinal cord. Ann Neurol 38(4):691–695. https://doi.org/10.1002/ana.410380424
Pollari E, Goldsteins G, Bart G et al (2014) The role of oxidative stress in degeneration of the neuromuscular junction in amyotrophic lateral sclerosis. Front Cell Neurosci 8:131. https://doi.org/10.3389/fncel.2014.00131
Joyce PI, Fratta P, Fisher EMC, Acevedo-Arozena A (2011) SOD1 and TDP-43 animal models of amyotrophic lateral sclerosis: recent advances in understanding disease toward the development of clinical treatments. Mamm Genome 22(7–8):420–448. https://doi.org/10.1007/s00335-011-9339-1
McGoldrick P, Joyce PI, Fisher EMC, Greensmith L (2013) Rodent models of amyotrophic lateral sclerosis. Biochim Biophys Acta 1832(9):1421–1436. https://doi.org/10.1016/j.bbadis.2013.03.012
Smith RG, Henry YK, Mattson MP, Appel SH (1998) Presence of 4-hydroxynonenal in cerebrospinal fluid of patients with sporadic amyotrophic lateral sclerosis. Ann Neurol 44(4):696–699. https://doi.org/10.1002/ana.410440419
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Imai S, Guarente L (2014) NAD+ and sirtuins in aging and disease. Trends Cell Biol 24(8):464–471. https://doi.org/10.1016/j.tcb.2014.04.002
Jasper H (2013) Sirtuins: longevity focuses on NAD+. Nat Chem Biol 9(11):666–667. https://doi.org/10.1038/nchembio.1369
Trammell SAJ, Schmidt MS, Weidemann BJ, Redpath P, Jaksch F, Dellinger RW, Li Z, Abel ED et al (2016) Nicotinamide riboside is uniquely and orally bioavailable in mice and humans. Nat Commun 7:12948. https://doi.org/10.1038/ncomms12948
Trammell SA, Yu L, Redpath P, Migaud ME, Brenner C (2016) Nicotinamide riboside is a major NAD+ precursor vitamin in cow milk. J Nutr 146(5):957–963. https://doi.org/10.3945/jn.116.230078
Freitas AA, de Magalhaes JP (2011) A review and appraisal of the DNA damage theory of ageing. Mutat Res 728(1–2):12–22. https://doi.org/10.1016/j.mrrev.2011.05.001
Gorbunova V, Seluanov A, Mao Z, Hine C (2007) Changes in DNA repair during aging. Nucleic Acids Res 35(22):7466–7474. https://doi.org/10.1093/nar/gkm756
Lavado-Roldan A, Fernandez-Chacon R (2016) Two for the price of one: a neuroprotective chaperone kit within NAD synthase protein NMNAT2. PLoS Biol 14(7):e1002522. https://doi.org/10.1371/journal.pbio.1002522
Ali YO, Allen HM, Yu L, Li-Kroeger D, Bakhshizadehmahmoudi D, Hatcher A, McCabe C, Xu J et al (2016) NMNAT2:HSP90 complex mediates proteostasis in proteinopathies. PLoS Biol 14(6):e1002472. https://doi.org/10.1371/journal.pbio.1002472
Gerdts J, Summers DW, Milbrandt J, DiAntonio A (2016) Axon self-destruction: new links among SARM1, MAPKs, and NAD+ metabolism. Neuron 89(3):449–460. https://doi.org/10.1016/j.neuron.2015.12.023
Jayaram HN, Kusumanchi P, Yalowitz JA (2011) NMNAT expression and its relation to NAD metabolism. CMC 18(13):1962–1972. https://doi.org/10.2174/092986711795590138
Di Stefano M, Nascimento-Ferreira I, Orsomando G et al (2015) A rise in NAD precursor nicotinamide mononucleotide (NMN) after injury promotes axon degeneration. Cell Death Differ 22(5):731–742. https://doi.org/10.1038/cdd.2014.164
Di Stefano M, Loreto A, Orsomando G et al (2017) NMN Deamidase delays Wallerian degeneration and rescues axonal defects caused by NMNAT2 deficiency in vivo. Curr Biol 27(6):784–794. https://doi.org/10.1016/j.cub.2017.01.070
Blokhuis AM, Groen EJN, Koppers M, van den Berg LH, Pasterkamp RJ (2013) Protein aggregation in amyotrophic lateral sclerosis. Acta Neuropathol 125(6):777–794. https://doi.org/10.1007/s00401-013-1125-6
Eisele YS, Monteiro C, Fearns C, Encalada SE, Wiseman RL, Powers ET, Kelly JW (2015) Targeting protein aggregation for the treatment of degenerative diseases. Nat Rev Drug Discov 14(11):759–780. https://doi.org/10.1038/nrd4593
Cushman M, Johnson BS, King OD, Gitler AD, Shorter J (2010) Prion-like disorders: blurring the divide between transmissibility and infectivity. J Cell Sci 123(8):1191–1201. https://doi.org/10.1242/jcs.051672
Stefani M, Dobson CM (2003) Protein aggregation and aggregate toxicity: new insights into protein folding, misfolding diseases and biological evolution. J Mol Med (Berl) 81(11):678–699. https://doi.org/10.1007/s00109-003-0464-5
Salminen A, Kaarniranta K, Kauppinen A (2013) Crosstalk between oxidative stress and SIRT1: impact on the aging process. Int J Mol Sci 14(2):3834–3859. https://doi.org/10.3390/ijms14023834
Bonora M, Bononi A, de Marchi E et al (2014) Role of the c subunit of the F O ATP synthase in mitochondrial permeability transition. Cell Cycle 12(4):674–683. https://doi.org/10.4161/cc.23599
Ying W (2007) NAD+ and NADH in neuronal death. Jrnl Neuroimmune Pharm 2(3):270–275. https://doi.org/10.1007/s11481-007-9063-5
Rowe LA, Degtyareva N, Doetsch PW (2008) DNA damage-induced reactive oxygen species (ROS) stress response in Saccharomyces cerevisiae. Free Radic Biol Med 45(8):1167–1177. https://doi.org/10.1016/j.freeradbiomed.2008.07.018
Acknowledgements
The authors gratefully acknowledge A. Lodwig, C. Grzelak, and S. Wenderdel for technical assistance, as well as A. Lenz for secretarial work. This research was supported by FoRUM - F874N-2016 (Ruhr-University Bochum).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Röderer, P., Klatt, L., John, F. et al. Increased ROS Level in Spinal Cord of Wobbler Mice due to Nmnat2 Downregulation. Mol Neurobiol 55, 8414–8424 (2018). https://doi.org/10.1007/s12035-018-0999-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-0999-7