Abstract
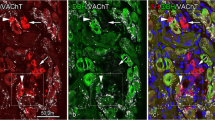
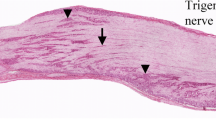
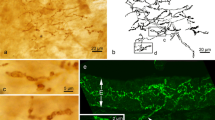
The beta-2 subunit of the mammalian brain voltage-gated sodium channel (SCN2B) was examined in the rat trigeminal ganglion (TG) and trigeminal sensory nuclei. In the TG, 42.6 % of sensory neurons were immunoreactive (IR) for SCN2B. These neurons had various cell body sizes. In facial skins and oral mucosae, corpuscular nerve endings contained SCN2B-immunoreactivity. SCN2B-IR nerve fibers formed nerve plexuses beneath taste buds in the tongue and incisive papilla. However, SCN2B-IR free nerve endings were rare in cutaneous and mucosal epithelia. Tooth pulps, muscle spindles and major salivary glands were also innervated by SCN2B-IR nerve fibers. A double immunofluorescence method revealed that about 40 % of SCN2B-IR neurons exhibited calcitonin gene-related peptide (CGRP)-immunoreactivity. However, distributions of SCN2B- and CGRP-IR nerve fibers were mostly different in facial, oral and cranial structures. By retrograde tracing method, 60.4 and 85.3 % of TG neurons innervating the facial skin and tooth pulp, respectively, showed SCN2B-immunoreactivity. CGRP-immunoreactivity was co-localized by about 40 % of SCN2B-IR cutaneous and tooth pulp TG neurons. In trigeminal sensory nuclei of the brainstem, SCN2B-IR neuronal cell bodies were common in deep laminae of the subnucleus caudalis, and the subnuclei interpolaris and oralis. In the mesencephalic trigeminal tract nucleus, primary sensory neurons also exhibited SCN2B-immunoreactivity. In other regions of trigeminal sensory nuclei, SCN2B-IR cells were very infrequent. SCN2B-IR neuropil was detected in deep laminae of the subnucleus caudalis as well as in the subnuclei interpolaris, oralis and principalis. These findings suggest that SCN2B is expressed by various types of sensory neurons in the TG. There appears to be SCN2B-containing pathway in the TG and trigeminal sensory nuclei.





Similar content being viewed by others
References
Chahine M, O’Leary ME (2011) Regulatory role of voltage-gated Na channel β subunits in sensory neurons. Front Pharmacol 2:70
Ho C, Zhao J, Malinowski S, Chahine M, O’Leary ME (2012) Differential expression of sodium channel β subunits in dorsal root ganglion sensory neurons. J Biol Chem 287:15044–15053
Ichikawa H, Sugimoto T (1997) Parvalbumin- and calbindin D-28 k-immunoreactive innervation of orofacial tissues in the rat. Exp Neurol 146:414–418
Ichikawa H, Sugimoto T (2000) VRL-1-immunoreactive primary sensory neurons in the rat trigeminal nervous system. Neuroscience 101:719–725
Ichikawa H, Sugimoto T (2001) VR1-immunoreactive primary sensory neurons in the rat trigeminal ganglion. Brain Res 890:184–188
Ichikawa H, Sugimoto T (2003) Calcium-binding protein-immunoreactive innervation of the rat vibrissa. Brain Res 970:226–231
Ichikawa H, Deguchi T, Nakago T, Jacobowitz DM, Sugimoto T (1994) Parvalbumin, calretinin and carbonic anhydrase in the trigeminal and spinal primary neurons of the rat. Brain Res 655:241–245
Ichikawa H, Deguchi T, Nakago T, Jacobowitz DM, Sugimoto T (1995) Parvalbumin- and calretinin-immunoreactive trigeminal neurons innervating the rat molar tooth pulp. Brain Res 679:205–211
Ichikawa H, Jacobowitz DM, Sugimoto T (1997) Coexpression of calretinin and parvalbumin in Ruffini-like endings in the rat incisor periodontal ligament. Brain Res 770:294–297
Ichikawa H, Hidaka H, Sugimoto T (2000) Neurocalcin-immunoreactive primary sensory neurons in the trigeminal ganglion provide myelinated innervation to the tooth pulp and periodontal ligament. Brain Res 864:152–156
Ichikawa H, Yamashita K, Takano-Yamamoto T, Sugimoto T (2001) Osteopontin-immunoreactivity in the rat trigeminal ganglion and trigeminal sensory nuclei. Brain Res 919:147–154
Ichikawa H, Schulz S, Höllt V, Sugimoto T (2005) Delta-opioid receptor-immunoreactive neurons in the rat cranial sensory ganglia. Brain Res 1043:225–230
Iino S, Kato M, Hidaka H, Kobayashi S (1998) Neurocalcin-immunopositive neurons in the rat sensory ganglia. Brain Res 781:236–243
Isom LL, Ragsdale DS, De Jongh KS, Westenbroek RE, Reber BF, Scheuer T, Catterall WA (1995) Structure and function of the beta 2 subunit of brain sodium channels, a transmembrane glycoprotein with a CAM motif. Cell 83:433–442
Kim DY, Ingano LA, Carey BW, Pettingell WH, Kovacs DM (2005) Presenilin/gamma-secretase-mediated cleavage of the voltage-gated sodium channel beta2-subunit regulates cell adhesion and migration. J Biol Chem 17:23251–23261
Kruger L, Sternini C, Brecha NC, Mantyh PW (1988) Distribution of calcitonin gene-related peptide immunoreactivity in relation to the rat central somatosensory projection. J Comp Neurol 273:149–162
Lopez-Santiago LF, Pertin M, Morisod X, Chen C, Hong S, Wiley J, Decosterd I, Isom LL (2006) Sodium channel beta2 subunits regulate tetrodotoxin-sensitive sodium channels in small dorsal root ganglion neurons and modulate the response to pain. J Neurosci 26:7984–7994
O’Malley HA, Shreiner AB, Chen GH, Huffnagle GB, Isom LL (2009) Loss of Na+ channel beta2 subunits is neuroprotective in a mouse model of multiple sclerosis. Mol Cell Neurosci 40:143–155
Pertin M, Ji RR, Berta T, Powell AJ, Karchewski L, Tate SN, Isom LL, Woolf CJ, Gilliard N, Spahn DR, Decosterd I (2005) Upregulation of the voltage-gated sodium channel beta2 subunit in neuropathic pain models: characterization of expression in injured and non-injured primary sensory neurons. J Neurosci 25:10970–10980
Renehan WE, Munger BL (1986) Degeneration and regeneration of peripheral nerve in the rat trigeminal system. I. Identification and characterization of the multiple afferent innervation of the mystacial vibrissae. J Comp Neurol 246:129–145
Rice FL, Mance A, Munger BL (1986) A comparative light microscopic analysis of the sensory innervation of the mystacial pad. I. Innervation of vibrissal follicle-sinus complexes. J Comp Neurol 252:154–174
Sato T, Fujita M, Shimizu Y, Kanetaka H, Chu LW, Côté PD, Ichikawa H (2015) Glial reaction in the spinal cord of the degenerating muscle mouse (Scn8a (dmu)). Neurochem Res 40:124–129
Silverman JD, Kruger L (1989) Calcitonin gene-related peptide-immunoreactive innervation of the rat head with emphasis on specialized sensory structures. J Comp Neurol 280:303–330
Stefanini M, De Martino C, Zamboni L (1967) Fixation of ejaculated spermatozoa for electron microscopy. Nature 216:173–174
Sugimoto T, Fujiyoshi Y, Xiao C, He YF, Ichikawa H (1997) Central projection of calcitonin gene-related peptide (CGRP)- and substance P (SP)-immunoreactive trigeminal primary neurons in the rat. J Comp Neurol 378:425–442
Takahashi N, Kikuchi S, Dai Y, Kobayashi K, Fukuoka T, Noguchi K (2003) Expression of auxiliary beta subunits of sodium channels in primary afferent neurons and the effect of nerve injury. Neuroscience 121:441–450
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors do not have any conflict of interest.
Rights and permissions
About this article
Cite this article
Shimada, Y., Sato, T., Yajima, T. et al. SCN2B in the Rat Trigeminal Ganglion and Trigeminal Sensory Nuclei. Cell Mol Neurobiol 36, 1399–1408 (2016). https://doi.org/10.1007/s10571-016-0340-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-016-0340-9