Abstract
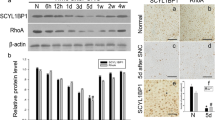
SYF2 is a putative homolog of human p29 in Saccharomyces cerevisiae. It seems to be involved in pre-mRNA splicing and cell cycle progression. Disruption of SYF2 leads to reduced α-tubulin expression and delayed nerve system development in zebrafish. Due to the potential of SYF2 in modulating microtubule dynamics in nervous system, we investigated the spatiotemporal expression of SYF2 in a rat sciatic nerve crush (SNC) model. We found that SNC resulted in a significant upregulation of SYF2 from 3 days to 1 week and subsequently returned to the normal level at 4 weeks. At its peak expression, SYF2 distributed predominantly in Schwann cells. In addition, upregulation of SYF2 was approximately in parallel with Oct-6, and numerous Schwann cells expressing SYF2 were Oct-6 positive. In vitro, we observed enhanced expression of SYF2 during the process of cyclic adenosine monophosphate (cAMP)-induced Schwann cell differentiation. SYF2-specific siRNA-transfected Schwann cells did not show significant morphological change in the process of Schwann cell differentiation. Also, we found shorter and disorganized microtubule structure and a decreased migration in SYF2-specific siRNA-transfected Schwann cells. Together, these findings indicated that the upregulation of SYF2 was associated with Schwann cell differentiation and migration following sciatic nerve crush.








Similar content being viewed by others
Abbreviations
- SNC:
-
Sciatic nerve crush
- PNI:
-
Peripheral nerve injury
- cAMP:
-
Cyclic adenosine monophosphate
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- P0:
-
Myelin protein zero
References
Aguayo AJ, Epps J, Charron L, Bray GM (1976) Multipotentiality of Schwann cells in cross-anastomosed and grafted myelinated and unmyelinated nerves: quantitative microscopy and radioautography. Brain Res 104(1):1–20
Arroyo EJ, Bermingham JR Jr, Rosenfeld MG, Scherer SS (1998) Promyelinating Schwann cells express Tst-1/SCIP/Oct-6. J Neurosci 18(19):7891–7902
Ben-Yehuda S, Dix I, Russell CS, McGarvey M, Beggs JD, Kupiec M (2000) Genetic and physical interactions between factors involved in both cell cycle progression and pre-mRNA splicing in Saccharomyces cerevisiae. Genetics 156(4):1503–1517
Beuche W, Friede R (1984) The role of non-resident cells in Wallerian degeneration. J Neurocytol 13(5):767–796
Brück W (1997) The role of macrophages in Wallerian degeneration. Brain Pathol 7(2):741–752
Brück W, Huitinga I, Dijkstra CD (1996) Liposome-mediated monocyte depletion during Wallerian degeneration defines the role of hematogenous phagocytes in myelin removal. J Neurosci Res 46(4):477–484
Chang MS, Chang CL, Huang CJ, Yang YC (2000) p29, a novel GCIP-interacting protein, localizes in the nucleus. Biochem Biophys Res Commun 279(2):732–737. doi:10.1006/bbrc.2000.3992
Chen Z-L, Yu W-M, Strickland S (2007) Peripheral regeneration. Annu Rev Neurosci 30:209–233
Chen C-H, Chu P-C, Lee L, Lien H-W, Lin T-L, Fan C–C, Chi P, Huang C-J, Chang M-S (2012a) Disruption of murine mp29/Syf2/Ntc31 gene results in embryonic lethality with aberrant checkpoint response. PLoS One 7(3):e33538
Chen CH, Chu PC, Lee L, Lien HW, Lin TL, Fan CC, Chi P, Huang CJ, Chang MS (2012b) Disruption of murine mp29/Syf2/Ntc31 gene results in embryonic lethality with aberrant checkpoint response. PLoS One 7(3):e33538. doi:10.1371/journal.pone.0033538
Cheng C, Qin Y, Shao X, Wang H, Gao Y, Cheng M, Shen A (2007) Induction of TNF-alpha by LPS in Schwann cell is regulated by MAPK activation signals. Cell Mol Neurobiol 27(7):909–921. doi:10.1007/s10571-007-9215-4
Chu PC, Yang YC, Lu YT, Chen HT, Yu LC, Chang MS (2006) Silencing of p29 affects DNA damage responses with UV irradiation. Cancer Res 66(17):8484–8491. doi:10.1158/0008-5472.CAN-05-3229
Dahan O, Kupiec M (2002a) Mutations in genes of Saccharomyces cerevisiae encoding pre-mRNA splicing factors cause cell cycle arrest through activation of the spindle checkpoint. Nucleic Acids Res 30(20):4361–4370
Dahan O, Kupiec M (2002b) Mutations in genes of Saccharomyces cerevisiae encoding pre-mRNA splicing factors cause cell cycle arrest through activation of the spindle checkpoint. Nucleic Acids Res 30(20):4361–4370
Drewes G, Trinczek B, Illenberger S, Biernat J, Schmitt-Ulms G, Meyer HE, Mandelkow E-M, Mandelkow E (1995) Microtubule-associated protein/microtubule affinity-regulating kinase (p110mark). A novel protein kinase that regulates tau-microtubule interactions and dynamic instability by phosphorylation at the Alzheimer-specific site serine 262. J Biol Chem 270(13):7679–7688
Eser F, Bodur H, Atan Ç (2009) Etiological factors of traumatic peripheral nerve injuries. Neurol India 57(4):434–437
Etienne-Manneville S (2010) From signaling pathways to microtubule dynamics: the key players. Curr Opin Cell Biol 22(1):104–111
Fawcett JW, Keynes RJ (1990) Peripheral nerve regeneration. Annu Rev Neurosci 13:43–60. doi:10.1146/annurev.ne.13.030190.000355
Feneley MR, Fawcett JW, Keynes RJ (1991) The role of Schwann cells in the regeneration of peripheral nerve axons through muscle basal lamina grafts. Exp Neurol 114(3):275–285
Fricker FR, Bennett DL (2011) The role of neuregulin-1 in the response to nerve injury. Future neurol 6(6):809–822
Gaudet AD, Popovich PG, Ramer MS (2011) Wallerian degeneration: gaining perspective on inflammatory events after peripheral nerve injury. J Neuroinflammation 8(1):110
Hokfelt T, Zhang X, Wiesenfeld-Hallin Z (1994) Messenger plasticity in primary sensory neurons following axotomy and its functional implications. Trends Neurosci 17(1):22–30
Horie H, Kadoya T, Hikawa N, Sango K, Inoue H, Takeshita K, Asawa R, Hiroi T, Sato M, Yoshioka T (2004) Oxidized galectin-1 stimulates macrophages to promote axonal regeneration in peripheral nerves after axotomy. J Neurosci 24(8):1873–1880
Horner PJ, Gage FH (2000) Regenerating the damaged central nervous system. Nature 407(6807):963–970
Jessen KR, Mirsky R (2005) The origin and development of glial cells in peripheral nerves. Nat Rev Neurosci 6(9):671–682. doi:10.1038/nrn1746
Ji Y, Tao T, Cheng C, Yang H, Wang Y, Yang J, Liu H, He X, Wang H, Shen A (2010) SSeCKS is a suppressor in Schwann cell differentiation and myelination. Neurochem Res 35(2):219–226. doi:10.1007/s11064-009-0045-2
Karlsson JE, Rosengren L, Wang S, Danielsen N, Haglid K (1993) Glial and neuronal marker proteins in the silicone chamber model for nerve regeneration. J Neurochem 60(3):1098–1104
Kidd G, Andrews SB, Trapp BD (1996) Axons regulate the distribution of Schwann cell microtubules. J Neurosci 16(3):946–954
Kiefer R, Kieseier BC, Stoll G, Hartung H-P (2001) The role of macrophages in immune-mediated damage to the peripheral nervous system. Prog Neurobiol 64(2):109–127
Konigsmark BW, Murphy EA (1970) Neuronal populations in the human brain. Nature 228:1335–1336
La Fleur M, Underwood JL, Rappolee DA, Werb Z (1996) Basement membrane and repair of injury to peripheral nerve: defining a potential role for macrophages, matrix metalloproteinases, and tissue inhibitor of metalloproteinases-1. J Exp Med 184(6):2311–2326
Lee SK, Wolfe SW (2000) Peripheral nerve injury and repair. J Am Acad Orthop Surg 8(4):243–252
Li H, Yang H, Liu Y, Huan W, Zhang S, Wu G, Lu Q, Wang Q, Wang Y (2011) The cyclin-dependent kinase inhibitor p27(Kip1) is a positive regulator of Schwann cell differentiation in vitro. J Mol Neurosci 45(2):277–283. doi:10.1007/s12031-011-9518-2
Liu HM, Yang LH, Yang YJ (1995) Schwann cell properties: 3. C-fos expression, bFGF production, phagocytosis and proliferation during Wallerian degeneration. J Neuropathol Exp Neurol 54(4):487–496
Liu Y, Chen Y, Lu X, Wang Y, Duan Y, Cheng C, Shen A (2012) SCYL1BP1 modulates neurite outgrowth and regeneration by regulating the Mdm2/p53 pathway. Mol Biol Cell 23(23):4506–4514
Long L, Huang Y, Wu H, Luan W, Zhang Q, Wen H, Ding T, Wang Y (2013) Dynamic change of Prohibitin2 expression in rat sciatic nerve after crush. Cell Mol Neurobiol 33(5):689–698. doi:10.1007/s10571-013-9935-6
Lou D, Sun B, Wei H, Deng X, Chen H, Xu D, Li G, Xu H, Wang Y (2012) Spatiotemporal expression of testicular protein kinase 1 after rat sciatic nerve injury. J Mol Neurosci 47(1):180–191
Lundborg G (2003) Richard P. Bunge memorial lecture. Nerve injury and repair–a challenge to the plastic brain. J Peripher Nerv Syst 8(4):209–226
Martini R, Fischer S, López-Vales R, David S (2008) Interactions between Schwann cells and macrophages in injury and inherited demyelinating disease. Glia 56(14):1566–1577
Meunier S, Vernos I (2012) Microtubule assembly during mitosis–from distinct origins to distinct functions? J Cell Sci 125(12):2805–2814
Mirsky R, Jessen KR (1996) Schwann cell development, differentiation and myelination. Curr Opin Neurobiol 6(1):89–96
Morell P, Quarles R, Norton W (1999) Basic neurochemistry: molecular, cellular, and medical aspects. Myelin Formation, Structure, and Biochemistry Lippincott-Raven Publishers:79-93
Murinson BB, Archer DR, Li Y, Griffin JW (2005) Degeneration of myelinated efferent fibers prompts mitosis in Remak Schwann cells of uninjured C-fiber afferents. J Neurosci 25(5):1179–1187. doi:10.1523/JNEUROSCI.1372-04.2005
Pellegrino RG, Politis MJ, Ritchie JM, Spencer PS (1986) Events in degenerating cat peripheral nerve: induction of Schwann cell S phase and its relation to nerve fibre degeneration. J Neurocytol 15(1):17–28
Robinson LR (2000a) Traumatic injury to peripheral nerves. Muscle Nerve 23(6):863–873
Robinson LR (2000b) Traumatic injury to peripheral nerves. Muscle Nerve 23(6):863–873
Salzer JL, Bunge RP (1980) Studies of Schwann cell proliferation. I. An analysis in tissue culture of proliferation during development, Wallerian degeneration, and direct injury. J Cell Biol 84(3):739–752
Scherer SS, Wang DY, Kuhn R, Lemke G, Wrabetz L, Kamholz J (1994) Axons regulate Schwann cell expression of the POU transcription factor SCIP. J Neurosci 14(4):1930–1942
Spencer P, Politis M, Pellegrino R, Weinberg H (1981) Control of Schwann cell behavior during nerve degeneration and regeneration. Raven Press, New York
Su X, Ohi R, Pellman D (2012) Move in for the kill: motile microtubule regulators. Trends Cell Biol 22(11):567–575
Tang EI, Mruk DD, Cheng CY (2013) MAP/microtubule affinity-regulating kinases, microtubule dynamics, and spermatogenesis. J Endocrinol 217(2):R13–R23
Trapp BD, Hauer P, Lemke G (1988) Axonal regulation of myelin protein mRNA levels in actively myelinating Schwann cells. J Neurosci 8(9):3515–3521
Wade RH, Hyman AA (1997) Microtubule structure and dynamics. Curr Opin Cell Biol 9(1):12–17
Wang Y, Liu Y, Chen Y, Shi S, Qin J, Xiao F, Zhou D, Lu M, Lu Q, Shen A (2009) Peripheral nerve injury induces down-regulation of Foxo3a and p27kip1 in rat dorsal root ganglia. Neurochem Res 34(5):891–898
Weinberg HJ, Spencer PS (1976) Studies on the control of myelinogenesis. II. Evidence for neuronal regulation of myelin production. Brain Res 113(2):363–378
White FV, Toews AD, Goodrum JF, Novicki DL, Bouldin TW, Morell P (1989) Lipid metabolism during early stages of Wallerian degeneration in the rat sciatic nerve. J Neurochem 52(4):1085–1092
Xu W, Cao M, Zheng H, Tan X, Li L, Cui G, Xu J, Cao J, Ke K, Wu Q (2014a) Upregulation of SYF2 is associated with neuronal apoptosis caused by reactive astrogliosis to neuroinflammation. J Neurosci Res 92(3):318–328
Xu W, Cao M, Zheng H, Tan X, Li L, Cui G, Xu J, Cao J, Ke K, Wu Q (2014b) Upregulation of SYF2 is associated with neuronal apoptosis caused by reactive astrogliosis to neuroinflammation. J Neurosci Res 92(3):318–328. doi:10.1002/jnr.23312
Yang DP, Zhang DP, Mak KS, Bonder DE, Pomeroy SL, Kim HA (2008) Schwann cell proliferation during Wallerian degeneration is not necessary for regeneration and remyelination of the peripheral nerves: axon-dependent removal of newly generated Schwann cells by apoptosis. Mol Cell Neurosci 38(1):80–88. doi:10.1016/j.mcn.2008.01.017
Yao L, Cao J, Sun H, Guo A, Li A, Ben Z, Zhang H, Wang X, Ding Z, Yang X (2014) FBP1 and p27kip1 expression after sciatic nerve injury: implications for Schwann cells proliferation and differentiation. J Cell Biochem 115(1):130–140
Zhu X, Yao L, Yang X, Sun H, Guo A, Li A, Yang H (2014) Spatiotemporal expression of KHSRP modulates Schwann cells and neuronal differentiation after sciatic nerve injury. Int J biochem Cell Biol 48:1–10
Zorick TS, Lemke G (1996) Schwann cell differentiation. Curr Opin Cell Biol 8(6):870–876
Zorick TS, Syroid DE, Arroyo E, Scherer SS, Lemke G (1996) The transcription factors SCIP and Krox-20 mark distinct stages and cell fates in Schwann cell differentiation. Mol Cell Neurosci 8(2–3):129–145. doi:10.1006/mcne.1996.0052
Acknowledgments
This work was supported by the National Natural Scientific Foundation of China Grant (No. 31300902), the Colleges and Universities in Natural Science Research Project of Jiangsu Province (13KJB310009), and a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Conflict of interest
All the authors of this study declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Zhengming Zhou, Yang Liu, Yonghua Liu and Huiguang Yang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhou, Z., Liu, Y., Nie, X. et al. Involvement of Upregulated SYF2 in Schwann Cell Differentiation and Migration After Sciatic Nerve Crush. Cell Mol Neurobiol 34, 1023–1036 (2014). https://doi.org/10.1007/s10571-014-0078-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-014-0078-1