Abstract
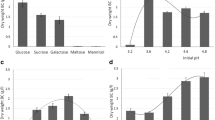
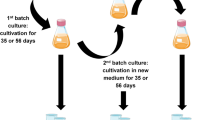
The morphology, crystallinity, crystallite size, and production yield of bacterial cellulose (BC) produced with six different carbon sources (glucose, fructose, lactose, maltitol, sucralose, and xylitol) in static and agitated fermentation conditions by five strains of Komagataeibacter xylinus (KX, TISTR 086, 428, 975, and 1011) which are locally available, were studied. In static condition, the BC pellicle was formed as a membrane sheet at the medium surface exposed to air, while in agitated condition, the spherical or asterisk-like shape BC particles were obtained in the culture media. The XRD and FT-IR analyses found no significant differences in the cellulose crystallinity, crystallite size or polymorphic distribution within the carbon sources. However, changes in crystallinity and mass fraction of the Iα allomorph were observed in BC produced from the different bacterial strains and incubation conditions. The BC samples produced by the same bacterial strain with the varying culture conditions showed the alteration of physical properties more clearly than the BC samples prepared by the opposite situation. These findings suggested that the strains of bacteria and fermentation conditions strongly affected on the physical structures of BC.





Similar content being viewed by others
References
Atalla RH, Vanderhart DL (1984) Native cellulose: a composite of two cistinct crystalline forms. Science 223:283–285. https://doi.org/10.1126/science.223.4633.283
Benziman M, Haigler CH, Brown RM, White AR, Cooper KM (1980) Cellulose biogenesis: polymerization and crystallization are coupled processes in Acetobacter xylinum. Proc Natl Acad Sci U S A 77:6678–6682
Bi JC, Liu SX, Li CF, Li J, Liu LX, Deng J, Yang YC (2014) Morphology and structure characterization of bacterial celluloses produced by different strains in agitated culture. J Appl Microbiol 117:1305–1311. https://doi.org/10.1111/jam.12619
Brown RM (2004) Cellulose structure and biosynthesis: what is in store for the 21st century? J Polym Sci Part A Polym Chem 42:487–495. https://doi.org/10.1002/pola.10877
Budhiono A, Rosidi B, Taher H, Iguchi M (1999) Kinetic aspects of bacterial cellulose formation in nata-de-coco culture system. Carbohydr Polym 40:137–143. https://doi.org/10.1016/S0144-8617(99)00050-8
Castro C, Zuluaga R, Putaux J-L, Caro G, Mondragon I, Gañán P (2011) Structural characterization of bacterial cellulose produced by Gluconacetobacter swingsii sp. from Colombian agroindustrial wastes. Carbohydr Polym 84:96–102. https://doi.org/10.1016/j.carbpol.2010.10.072
Chang C, Zhang L (2011) Cellulose-based hydrogels: present status and application prospects. Carbohydr Polym 84:40–53. https://doi.org/10.1016/j.carbpol.2010.12.023
Cheng K-C, Catchmark JM, Demirci A (2009) Enhanced production of bacterial cellulose by using a biofilm reactor and its material property analysis. J Biol Eng 3:12. https://doi.org/10.1186/1754-1611-3-12
Colvin JR, Leppard GG (1977) The biosynthesis of cellulose by Acetobacter xylinum and Acetobacter acetigenus. Can J Microbiol 23:701–709. https://doi.org/10.1139/m77-105
Czaja W, Romanovicz D, Rm Brown (2004) Structural investigations of microbial cellulose produced in stationary and agitated culture. Cellulose 11:403–411. https://doi.org/10.1023/b:cell.0000046412.11983.61
Delmer DP, Amor Y (1995) Cellulose biosynthesis. Plant Cell 7:987–1000
Dudman WF (1960) Cellulose production by Acetobacter strains in submerged culture. Microbiology 22:25–39. https://doi.org/10.1099/00221287-22-1-25
French AD (2014) Idealized powder diffraction patterns for cellulose polymorphs. Cellulose 21:885–896. https://doi.org/10.1007/s10570-013-0030-4
Gindl W, Keckes J (2004) Tensile properties of cellulose acetate butyrate composites reinforced with bacterial cellulose. Compos Sci Technol 64:2407–2413. https://doi.org/10.1016/j.compscitech.2004.05.001
Hestrin S, Schramm M (1954) Synthesis of cellulose by acetobacter xylinum. 2. Preparation of freeze-dried cells capable of polymerizing glucose to cellulose. Biochem J 58:345–352
Hirai A, Tsuji M, Horii F (2002) TEM study of band-like cellulose assemblies produced by acetobacter xylinum at 4 C. Cellulose 9:105–113. https://doi.org/10.1023/a:1020195205030
Hu Y, Catchmark JM (2011) Integration of cellulases into bacterial cellulose: toward bioabsorbable cellulose composites. J Biomed Mater Res Part B 97B:114–123. https://doi.org/10.1002/jbm.b.31792
Iguchi M, Yamanaka S, Budhiono A (2000) Bacterial cellulose—a masterpiece of nature’s arts. J Mater Sci 35:261–270. https://doi.org/10.1023/a:1004775229149
Jahn CE, Selimi DA, Barak JD, Charkowski AO (2011) The Dickeya dadantii biofilm matrix consists of cellulose nanofibres, and is an emergent property dependent upon the type III secretion system and the cellulose synthesis operon. Microbiology 157:2733–2744. https://doi.org/10.1099/mic.0.051003-0
Keshk S, Razek T, Sameshima K (2006) Bacterial cellulose production from beet molasses. Afr J Biotechnol 5:1519–1523
Klemm D, Heublein B, Fink H-P, Bohn A (2005) Cellulose: fascinating biopolymer and sustainableraw material. Angew Chem Int Ed 44:3358–3393. https://doi.org/10.1002/anie.200460587
Krystynowicz A, Czaja W, Wiktorowska-Jezierska A, Gonçalves-Miśkiewicz M, Turkiewicz M, Bielecki S (2002) Factors affecting the yield and properties of bacterial cellulose. J Ind Microbiol Biotech 29:189–195. https://doi.org/10.1038/sj.jim.7000303
Lee H, Zhao X (1999) Effects of mixing conditions on the production of microbial cellulose by Acetobacter xylinum. Biotechnol Bioprocess Eng 4:41–45. https://doi.org/10.1007/bf02931912
Mihranyan A, Llagostera AP, Karmhag R, Strømme M, Ek R (2004) Moisture sorption by cellulose powders of varying crystallinity. Int J Pharm 269:433–442. https://doi.org/10.1016/j.ijpharm.2003.09.030
Mohammadkazemi F, Azin M, Ashori A (2015) Production of bacterial cellulose using different carbon sources and culture media. Carbohydr Polym 117:518–523. https://doi.org/10.1016/j.carbpol.2014.10.008
Mohite B, Salunke B, Patil S (2013) Enhanced production ofbacterial cellulose by using Gluconacetobacter hansenii NCIM 2529 strain under shaking conditions. Appl Biochem Biotechnol 169:1497–1511. https://doi.org/10.1007/s12010-013-0092-7
Morgan JLW, Strumillo J, Zimmer J (2013) Crystallographic snapshot of cellulose synthesis and membrane translocation. Nature 493:181–186. https://doi.org/10.1038/nature11744
Nakagaito AN, Iwamoto S, Yano H (2005) Bacterial cellulose: the ultimate nano-scalar cellulose morphology for the production of high-strength composites. Appl Phys A 80:93–97. https://doi.org/10.1007/s00339-004-2932-3
Nguyen VT, Gidley MJ, Dykes GA (2008) Potential of a nisin-containing bacterial cellulose film to inhibit Listeria monocytogenes on processed meats. Food Microbiol 25:471–478. https://doi.org/10.1016/j.fm.2008.01.004
Omran A, Ahearn G, Bowers D, Swenson J, Coughlin C (2013) Metabolic effects of sucralose on environmental bacteria. J Toxicol 2013:6. https://doi.org/10.1155/2013/372986
Ross P, Mayer R, Benziman M (1991) Cellulose biosynthesis and function in bacteria. Microbiol Rev 55:35–58
Ruka DR, Simon GP, Dean KM (2012) Altering the growth conditions of Gluconacetobacter xylinus to maximize the yield of bacterial cellulose. Carbohydr Polym 89:613–622. https://doi.org/10.1016/j.carbpol.2012.03.059
Schramm M, Gromet Z, Hestrin S (1957) Synthesis of cellulose by Acetobacter xylinum. 3. Substrates and inhibitors. Biochem J 67:669–679
Tokoh C, Takabe K, Fujita M, Saiki H (1998) Cellulose synthesized by Acetobacter xylinum in the presence of acetyl glucomannan. Cellulose 5:249–261. https://doi.org/10.1023/a:1009211927183
Tsekos I (1999) The sites of cellulose synthesis in algae: diversity and evolution of cellulose-synthesizing enzyme complexes. J Phycol 35:635–655. https://doi.org/10.1046/j.1529-8817.1999.3540635.x
Watanabe K, Tabuchi M, Morinaga Y, Yoshinaga F (1998) Structural features andproperties of bacterial cellulose produced in agitated culture. Cellulose 5:187–200. https://doi.org/10.1023/a:1009272904582
Yamada Y, Yukphan P, Lan Vu HT, Muramatsu Y, Ochaikul D, Tanasupawat S, Nakagawa Y (2012) Description of Komagataeibacter gen. nov., with proposals of new combinations (Acetobacteraceae). J Gen Appl Microbiol 58:397–404. https://doi.org/10.2323/jgam.58.397
Yamamoto H, Horii F, Hirai A (1996) In situ crystallization of bacterial cellulose II. Influences of different polymeric additives on the formation of celluloses Iα and Iβ at the early stage of incubation. Cellulose 3:229–242. https://doi.org/10.1007/bf02228804
Yamanaka S, Watanabe K, Kitamura N, Iguchi M, Mitsuhashi S, Nishi Y, Uryu M (1989) The structure and mechanical properties of sheets prepared from bacterial cellulose. J Mater Sci 24:3141–3145. https://doi.org/10.1007/bf01139032
Yoshinaga F, Tonouchi N, Watanabe K (1997) Research progress in production of bacterial cellulose by aeration and agitation culture and its application as a new industrial material. Biosci Biotechnol Biochem 61:219–224. https://doi.org/10.1271/bbb.61.219
Zhu H, Jia S, Wan T, Jia Y, Yang H, Li J, Yan L, Zhong C (2011) Biosynthesis of spherical Fe3O4/bacterial cellulose nanocomposites as adsorbents for heavy metal ions. Carbohydr Polym 86:1558–1564. https://doi.org/10.1016/j.carbpol.2011.06.061
Acknowledgments
The Petroleum and Petrochemical College, Chulalongkorn University was thanked for supporting. This research was funded by the Doctoral Degree Chulalongkorn University 100th Year Birthday Anniversary Scholarship, and the Ratchadapisek Sompoch Endowment Fund (2016), Chulalongkorn University (CU-59-026-AM).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singhsa, P., Narain, R. & Manuspiya, H. Physical structure variations of bacterial cellulose produced by different Komagataeibacter xylinus strains and carbon sources in static and agitated conditions. Cellulose 25, 1571–1581 (2018). https://doi.org/10.1007/s10570-018-1699-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-018-1699-1