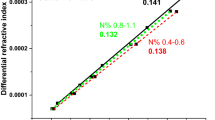
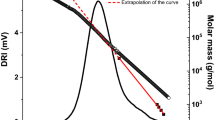
Abstract
Dialdehyde cellulose (DAC) has gained increasing interest because its (masked) aldehyde functionalities promise a rich follow-up chemistry and further conversion into different derivatives with varying properties. So far, the question of degradation upon DAC production and the resulting molar mass changes have remained unanswered since the problem of molar mass determination of DACs had not been solved. In this study, we present a comparison of two methods based on aqueous eluants, size exclusion chromatography and asymmetric flow field-flow fractionation (AsFlFFF), to determine the molar mass distribution of highly oxidized and thus water-soluble DACs, and to elucidate the impact of oxidation on the chain length of different cellulose substrates. Molar masses were determined by multi-angle laser light scattering in the case of both separation systems. The measurements were complemented by dynamic light scattering. AsFlFFF demonstrated an overall better separation efficiency, but at the expense of increased time consumption for method development. The results of molar mass analysis strongly depended on the eluant. Hemiacetal crosslinks are stable in near-neutral, low ionic strength eluants, while they have a tendency to be cleaved in acidic eluants or in solvents of higher ionic strength. By comparison of different dissolution strategies the optimal conditions for DAC analysis are proposed for both separation techniques. It is concluded that the extent of degradation upon dissolution can be significantly minimized by controlling the dissolution parameters, but degradation cannot be avoided completely.









Similar content being viewed by others
References
Berggren R, Berthold F, Sjöholm E, Lindström M (2003) Improved methods for evaluating the molar mass distributions of cellulose in kraft pulp. J Appl Polym Sci 88:1170–1179. doi:10.1002/app.11767
Calvini P, Gorassini A, Luciano G, Franceschi E (2006) FTRI and WAXS analysis of periodate oxycellulose: evidence for a cluster mechanism of oxidation. Vib Spectrosc 40:177–183. doi:10.1016/j.vibspec.2005.08.004
Carmali S, Brocchini S (2014) Chapter 13: polyacetals. In: Deng SGKTL (ed) Natural and synthetic biomedical polymers. Elsevier, Oxford, pp 219–233
Cheong K-L, Wu D-T, Zhao J, Li S-P (2015) A rapid and accurate method for the quantitative estimation of natural polysaccharides and their fractions using high performance size exclusion chromatography coupled with multi-angle laser light scattering and refractive index detector. J Chromatogr A 1400:98–106. doi:10.1016/j.chroma.2015.04.054
Fiedorowicz M, Para A (2006) Structural and molecular properties of dialdehyde starch. Carbohydr Polym 63:360–366. doi:10.1016/j.carbpol.2005.08.054
Habibi Y, Chanzy H, Vignon M (2006) Tempo-mediated surface oxidation of cellulose whiskers. Cellulose 13:679–687. doi:10.1007/s10570-006-9075-y
Hasani M, Henniges U, Idstroem A, Nordstierna L, Westman G, Rosenau T, Potthast A (2013) Nano-cellulosic materials: the impact of water on their dissolution in DMAc/LiCl. Carbohydr Polym 98:1565–1572. doi:10.1016/j.carbpol.2013.07.001
Kim U-J, Kuga S, Wada M, Okano T, Kondo T (2000) Periodate oxidation of crystalline cellulose. Biomacromolecules 1:488–492. doi:10.1021/bm0000337
Kim U-J, Wada M, Kuga S (2004) Solubilization of dialdehyde cellulose by hot water. Carbohydr Polym 56:7–10. doi:10.1016/j.carbpol.2003.10.013
Li J, Wan Y, Li L, Liang H, Wang J (2009) Preparation and characterization of 2,3-dialdehyde bacterial cellulose for potential biodegradable tissue engineering scaffolds. Mater Sci Eng C 29:1635–1642. doi:10.1016/j.msec.2009.01.006
Li H, Wu B, Mu C, Lin W (2011) Concomitant degradation in periodate oxidation of carboxymethyl cellulose. Carbohydr Polym 84:881–886. doi:10.1016/j.carbpol.2010.12.026
Maekawa E, Kosaki T, Koshijima T (1986) Periodate oxidation of mercerized cellulose and regenerated cellulose. Wood Res Bull Wood Res Inst Kyoto Univ 73:44–49
Marianne M-F (1998) Molecular weights and molecular-weight distributions of cellulose and cellulose nitrates during ultrasonic and mechanical degradation. Cellul Deriv Am Chem Soc 688:184–193
Perlin AS (2006) Glycol-cleavage oxidation. In: Derek H (ed) Advances in carbohydrate chemistry and biochemistry. Academic Press, New York, pp 183–250
Potthast A, Rosenau T, Kosma P (2006) Analysis of oxidized functionalities in cellulose. In: Klemm D (ed) Polysaccharides II. Springer, Berlin, pp 1–48
Potthast A, Kostic M, Schiehser S, Kosma P, Rosenau T (2007) Studies on oxidative modifications of cellulose in the periodate system: molecular weight distribution and carbonyl group profiles. Holzforschung 61:662–667. doi:10.1515/HF.2007.099
Potthast A, Schiehser S, Rosenau T, Kostic M (2009) Oxidative modifications of cellulose in the periodate system: reduction and beta-elimination reactions. Holzforschung 63:12–17. doi:10.1515/HF.2009.108
Potthast A, Radosta S, Saake B, Lebioda S, Heinze T, Henniges U, Isogai A, Koschella A, Kosma P, Rosenau T, Schiehser S, Sixta H, Strlič M, Strobin G, Vorwerg W, Wetzel H (2015) Comparison testing of methods for gel permeation chromatography of cellulose: coming closer to a standard protocol. Cellulose 22:1591–1613. doi:10.1007/s10570-015-0586-2
Rosenau T, Potthast A, Krainz K, Yoneda Y, Dietz T, Shields Z-I, French A (2011) Chromophores in cellulosics, VI. First isolation and identification of residual chromophores from aged cotton linters. Cellulose 18:1623–1633. doi:10.1007/s10570-011-9585-0
Rosenau T, Potthast A, Krainz K, Hettegger H, Henniges U, Yoneda Y, Rohrer C, French A (2014) Chromophores in cellulosics, XI: isolation and identification of residual chromophores from bacterial cellulose. Cellulose 21:2271–2283. doi:10.1007/s10570-014-0289-0
Sihtola H (1960) Chemical properties of modified celluloses. Makromol Chem 35:250–265. doi:10.1002/macp.1960.020350113
Siller M, Amer H, Bacher M, Roggenstein W, Rosenau T, Potthast A (2015) Effects of periodate oxidation on cellulose polymorphs. Cellulose. doi:10.1007/s10570-015-0648-5
Stefanovic B, Rosenau T, Potthast A (2013) Effect of sonochemical treatments on the integrity and oxidation state of cellulose. Carbohydr Polym 92:921–927. doi:10.1016/j.carbpol.2012.09.039
Varma AJ, Chavan VB (1995) A study of crystallinity changes in oxidised celluloses. Polym Degrad Stab 49:245–250. doi:10.1016/0141-3910(95)87006-7
Vold IMN, Christensen BE (2005) Periodate oxidation of chitosans with different chemical compositions. Carbohydr Res 340:679–684. doi:10.1016/j.carres.2005.01.002
Vold IMN, Kristiansen KA, Christensen BE (2006) A study of the chain stiffness and extension of alginates, in vitro epimerized alginates, and periodate-oxidized alginates using size-exclusion chromatography combined with light scattering and viscosity detectors. Biomacromolecules 7:2136–2146. doi:10.1021/bm060099n
Xiang Q, Lee YY, Pettersson PO, Torget RW (2003) Heterogeneous aspects of acid hydrolysis of [alpha]-cellulose. Appl Biochem Biotechnol 107:505–514. doi:10.1385/ABAB:107:1-3:505
Acknowledgments
The authors acknowledge the financial support of Christian Doppler Laboratory “Advanced Cellulose Chemistry and Analytics”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sulaeva, I., Klinger, K.M., Amer, H. et al. Determination of molar mass distributions of highly oxidized dialdehyde cellulose by size exclusion chromatography and asymmetric flow field-flow fractionation. Cellulose 22, 3569–3581 (2015). https://doi.org/10.1007/s10570-015-0769-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-015-0769-x