Abstract
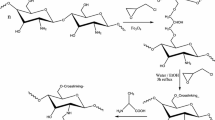
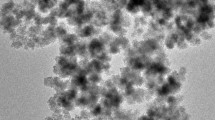
The recovery of three rare earth (RE) metals ions [Yb(III), Dy(III) and Nd(III), belonging to heavy, mild and light REs, respectively] was investigated using hybrid chitosan-magnetic nano-based particles functionalized by diethylenetriamine (DETA). The effect of pH on sorption performance was analyzed: the optimum initial pH value was found close to 5 (equilibrium pH value close to 6.5). The nanometric size of sorbent particles (30–50 nm) minimized the contribution of resistance to intraparticle diffusion on the control of uptake kinetics, which is efficiently modeled using the pseudo-second order rate equation: under selected experimental conditions the contact time required for reaching equilibrium was less than 1 h. Sorption isotherms were efficiently modeled using the Langmuir equation: maximum sorption capacities reached about 50 mg metal g−1, regardless of the RE. The temperature had a very limited effect on sorption capacity (in the range 300–320 K). The thermodynamic parameters were determined: the sorption was endothermic (positive values of ΔH°), spontaneous (negative values of ΔG°) and contributed to increasing the disorder of the system (positive values of ΔS°). The three REs have similar sorption properties on DETA-functionalized chitosan magnetic nano-based particles: the selective separation of these elements seems to be difficult. The sorbed metal ions can be removed from loaded sorbents using thiourea, and the sorbent can be recycled for at least five sorption/desorption cycles with a limited loss in sorption performance (by less than 6 %). The saturation magnetization was close to 20 emu g−1; this means that nano-based superparamagnetic particles can be readily recovered by an external magnetic field, making the processing of these materials easy.




Similar content being viewed by others
References
Abdel-Rahman AAH, El Aassy IE, Ahmed FY, Hamza MF (2010) Studies on the uptake of rare earth elements on polyacrylamidoxime resins from natural concentrate leachate solutions. J Dispersion Sci Technol 31:1128–1135. doi:10.1080/01932690903224821
Abreu RD, Morais CA (2014) Study on separation of heavy rare earth elements by solvent extraction with organophosphorus acids and amine reagents. Miner Eng 61:82–87. doi:10.1016/j.mineng.2014.03.015
Aghayan H, Mahjoub AR, Khanchi AR (2013) Samarium and dysprosium removal using 11-molybdo-vanadophosphoric acid supported on Zr modified mesoporous silica SBA-15. Chem Eng J 225:509–519. doi:10.1016/j.cej.2013.03.092
Allouche F-N, Guibal E, Mameri N (2014) Preparation of a new chitosan-based material and its application for mercury sorption. Colloids Surf A 446:224–232. doi:10.1016/j.colsurfa.2014.01.025
Barron L, O’Toole M, Diamond D, Nesterenko PN, Paull B (2008) Separation of transition metals on a poly-iminodiacetic acid grafted polymeric resin column with post-column reaction detection utilising a paired emitter-detector diode system. J Chromatogr A 1213:31–36. doi:10.1016/j.chroma.2008.08.049
Cadogan EI, Lee C-H, Popuri SR, Lin H-Y (2014) Efficiencies of chitosan nanoparticles and crab shell particles in europium uptake from aqueous solutions through biosorption: synthesis and characterization. Int Biodeterior Biodegrad Part A 95:232–240. doi:10.1016/j.ibiod.2014.06.003
Cotton S (2006) Lanthanide and actinide chemistry. Wiley, Chichester
Das N, Das D (2013) Recovery of rare earth metals through biosorption: an overview. J Rare Earths 31:933–943. doi:10.1016/s1002-0721(12)60382-2
Diniz V, Volesky B (2005a) Biosorption of La, Eu and Yb using Sargassum biomass. Water Res 39:239–247. doi:10.1016/j.waters.2004.09.009
Diniz V, Volesky B (2005b) Effect of counterions on lanthanum biosorption by Sargassum polycystum. Water Res 39:2229–2236. doi:10.1016/j.watres.2005.04.004
Dubinin MM, Zaverina ED, Radushkevich LV (1947) Sorption and structure of active carbons. I. Adsorption of organic vapors. Zh Fiz Khim 21:1351–1362
El-Didamony H, Ali MM, Awwad NS, Fawzy MM, Attallah MF (2012) Treatment of phosphogypsum waste using suitable organic extractants. J Radioanal Nucl Chem 291:907–914. doi:10.1007/s10967-011-1547-3
Elwakeel KZ, Atia AA (2014) Uptake of U(VI) from aqueous media by magnetic Schiff’s base chitosan composite. J Cleaner Prod 70:292–302. doi:10.1016/j.jclepro.2014.02.017
Elwakeel KZ, Atia AA, Donia AM (2009) Removal of Mo(VI) as oxoanions from aqueous solutions using chemically modified magnetic chitosan resins. Hydrometallurgy 97:21–28. doi:10.1016/j.hydromet.2008.12.009
Elwakeel KZ, Atia AA, Guibal E (2014) Fast removal of uranium from aqueous solutions using tetraethylenepentamine modified magnetic chitosan resin. Bioresour Technol 160:107–114. doi:10.1016/j.biortech.2014.01.037
Elyamani IS, Shabana EI (1985) Solvent-extraction of Lanthanum(III) from sulfuric-acid solutions by Primene JMT. J Less Common Met 105:255–261. doi:10.1016/0022-5088(85)90412-6
Foo KY, Hameed BH (2010) Insights into the modeling of adsorption isotherm systems. Chem Eng J 156:2–10. doi:10.1016/j.cej.2009.09.013
Freundlich HMF (1906) Uber die adsorption in lasungen. Z Phys Chem 57:385–470
Gao YH, Oshita K, Lee KH, Oshima M, Motomizu S (2002) Development of column-pretreatment chelating resins for matrix elimination/multi-element determination by inductively coupled plasma-mass spectrometry. Analyst 127:1713–1719. doi:10.1039/b208341h
Greenwood NN, Earnshaw A (1997) Chemistry of the elements, 2nd edn. Butterworth-Heinemann, Oxford
Guibal E (2004) Interactions of metal ions with chitosan-based sorbents: a review. Sep Purif Technol 38:43–74. doi:10.1016/j.seppur.2003.10.004
Hakim L, Sabarudin A, Oshima M, Motomizu S (2007) Synthesis of novel chitosan resin derivatized with serine diacetic acid moiety and its application to on-line collection/concentration of trace elements and their determination using inductively coupled plasma-atomic emission spectrometry. Anal Chim Acta 588:73–81. doi:10.1016/j.aca.2007.01.066
Hosomomi Y, Baba Y, Kubota F, Kamiya N, Goto M (2013) Biosorption of rare earth elements by Escherichia coli. J Chem Eng Jpn 46:450–454. doi:10.1252/jcej.13we031
Hu X-J et al (2011) Adsorption of chromium (VI) by ethylenediamine-modified cross-linked magnetic chitosan resin: isotherms, kinetics and thermodynamics. J Hazard Mater 185:306–314. doi:10.1016/j.jhazmat.2010.09.034
Innocenzi V, De Michelis I, Kopacek B, Veglio F (2014) Yttrium recovery from primary and secondary sources: a review of main hydrometallurgical processes. Waste Manage (Oxford) 34:1237–1250. doi:10.1016/j.wasman.2014.02.010
Jayakumar R, Prabaharan M, Reis RL, Mano JF (2005) Graft copolymerized chitosan—present status and applications. Carbohydr Polym 62:142–158. doi:10.1016/j.carbpol.2005.07.017
Koochaki-Mohammadpour SMA, Torab-Mostaedi M, Talebizadeh-Rafsanjani A, Naderi-Behdani F (2014) Adsorption isotherm, kinetic, thermodynamic, and desorption studies of lanthanum and dysprosium on oxidized multiwalled carbon nanotubes. J Dispers Sci Technol 35:244–254. doi:10.1080/01932691.2013.785361
Kozhevnikova NM, Tsybikova NL (2008) Sorption of neodymium(III) ions by natural mordenite-containing tuff. Russ J Appl Chem 81:42–45. doi:10.1134/s1070427208010102
Krishna PG, Gladis JM, Rao TP, Naidu GR (2005) Selective recognition of neodymium(III) using ion imprinted polymer particles. J Mol Recognit 18:109–116. doi:10.1002/jmr.720
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Amer Chem Soc 40:1361–1402
Lee GS, Uchikoshi M, Mimura K, Isshiki M (2010) Separation of major impurities Ce, Pr, Nd, Sm, Al, Ca, Fe, and Zn from La using bis(2-ethylhexyl)phosphoric acid (D2EHPA)-impregnated resin in a hydrochloric acid medium. Sep Purif Technol 71:186–191. doi:10.1016/j.seppur.2009.11.020
Lokshin EP, Ivanenko VI, Tareeva OA, Korneikov RI (2013) Sorption of rare earth elements of waste solution of leaching uranium. Russ J Appl Chem 86:450–452. doi:10.1134/s1070427213030269
Mahfouz MG, Galhoum AA, Gomaa NA, Abdel-Rehem SS, Atia AA, Vincent T, Guibal E (2015) Uranium extraction using magnetic nano-based particles of diethylenetriamine-functionalized chitosan: equilibrium and kinetic studies. Chem Eng J 262:198–209. doi:10.1016/j.cej.2014.09.061
Marcus Y (1997) Ion properties. Marcel Dekker Inc., New York
Martins TS, Isolani PC (2005) Rare earths: industrial and biological applications. Quim Nova 28:111–117. doi:10.1590/s0100-40422005000100020
Melnyk IV, Goncharyk VP, Kozhara LI, Yurchenko GR, Matkovsky AK, Zub YL, Alonso B (2012a) Sorption properties of porous spray-dried microspheres functionalized by phosphonic acid groups. Microporous Mesoporous Mater 153:171–177. doi:10.1016/j.micromeso.2011.12.027
Melnyk IV, Goncharyk VP, Stolyarchuk NV, Kozhara LI, Lunochkina AS, Alonso B, Zub YL (2012b) Dy(III) sorption from water solutions by mesoporous silicas functionalized with phosphonic acid groups. J Porous Mater 19:579–585. doi:10.1007/s10934-011-9508-3
Morf LS, Gloor R, Haag O, Haupt M, Skutan S, Di Lorenzo F, Boeni D (2013) Precious metals and rare earth elements in municipal solid waste—Sources and fate in a Swiss incineration plant. Waste Manage (Oxford) 33:634–644. doi:10.1016/j.wasman.2012.09.010
Namdeo M, Bajpai SK (2008) Chitosan-magnetite nanocomposites (CMNs) as magnetic carrier particles for removal of Fe(III) from aqueous solutions. Colloids Surf A 320:161–168. doi:10.1016/j.colsurfa.2008.01.053
Oliveira RC, Jouannin C, Guibal E, Garcia O Jr (2011) Samarium(III) and praseodymium(III) biosorption on Sargassum sp.: batch study. Proc Biochem 46:736–744. doi:10.1016/j.procbio.2010.11.021
Oliveira RC, Guibal E, Garcia O Jr (2012) Biosorption and desorption of lanthanum(III) and neodymium(III) in fixed-bed columns with Sargassum sp.: perspectives for separation of rare earth metals. Biotechnol Progr 28:715–722. doi:10.1002/btpr.1525
Oshita K, Takayanagi T, Oshima M, Motomizu S (2007) Adsorption behavior of cationic and anionic species on chitosan resins possessing amino acid moieties. Anal Sci 23:1431–1434. doi:10.2116/analsci.23.1431
Oshita K, Sabarudin A, Takayanagi T, Oshima M, Motomizu S (2009) Adsorption behavior of uranium(VI) and other ionic species on cross-linked chitosan resins modified with chelating moieties. Talanta 79:1031–1035. doi:10.1016/j.talanta.2009.03.035
Park H-J, Tavlarides LL (2010) Adsorption of neodymium(III) from aqueous solutions using a phosphorus functionalized adsorbent. Ind Eng Chem Res 49:12567–12575. doi:10.1021/ie100403b
Pearson RG (1966) Acids and bases. Science 151:172–177. doi:10.1126/science.151.3707.172
Qadeer R, Hanif J (1995) Adsorption of dysprosium ions on activated-charcoal from aqueous solutions. Carbon 33:215–220. doi:10.1016/0008-6223(94)00135-m
Qiu H, Lv L, Pan B, Zhang Q, Zhang W, Zhang Q (2009) Review: critical review in adsorption kinetic models. J Zhejiang Univ Sci A 10:716–724
Qu Y, Lian B (2013) Bioleaching of rare earth and radioactive elements from red mud using Penicillium tricolor RM-10. Bioresour Technol 136:16–23. doi:10.1016/j.biortech.2013.03.070
Rabatho JP, Tongamp W, Takasaki Y, Haga K, Shibayama A (2013) Recovery of Nd and Dy from rare earth magnetic waste sludge by hydrometallurgical process. J Mater Cycles Waste Manage 15:171–178. doi:10.1007/s10163-012-0105-6
Rahmati A, Ghaemi A, Samadfam M (2012) Kinetic and thermodynamic studies of uranium(VI) adsorption using Amberlite IRA-910 resin. Ann Nucl Energy 39:42–48. doi:10.1016/j.anucene.2011.09.006
Ren Y, Abbood HA, He F, Peng H, Huang K (2013) Magnetic EDTA-modified chitosan/SiO2/Fe3O4 adsorbent: preparation, characterization, and application in heavy metal adsorption. Chem Eng J 226:300–311. doi:10.1016/j.cej.2013.04.059
Repo E, Warchol JK, Bhatnagar A, Mudhoo A, Sillanpaa M (2013) Aminopolycarboxylic acid functionalized adsorbents for heavy metals removal from water. Water Res 47:4812–4832. doi:10.1016/j.watres.2013.06.020
Roosen J, Binnemans K (2014) Adsorption and chromatographic separation of rare earths with EDTA- and DTPA-functionalized chitosan biopolymers. J Mater Chem A 2:1530–1540. doi:10.1039/c3ta14622g
Roosen J, Spooren J, Binnemans K (2014) Adsorption performance of functionalized chitosan-silica hybrid materials toward rare earths. J Mater Chem A 2:19415–19426. doi:10.1039/c4ta04518a
Ruiz M, Sastre AM, Guibal E (2002a) Pd and Pt recovery using chitosan gel beads: II. Influence of chemical and physical modification on sorption properties. Sep Sci Technol 37:2385–2403
Ruiz MA, Sastre AM, Guibal E (2002b) Pd and Pt recovery using chitosan gel beads: I. Influence of drying process on diffusion properties. Sep Sci Technol 37:2143–2166
Russo P, Acierno D, Palomba M, Carotenuto G, Rosa R, Rizzuti A, Leonelli C (2012) Ultrafine magnetite nanopowder: synthesis, characterization, and preliminary use as filler of polymethylmethacrylate nanocomposites. J Nanotechnol 2012:8. doi:10.1155/2012/728326
Sapsford DJ, Bowell RJ, Geroni JN, Penman KM, Dey M (2012) Factors influencing the release rate of uranium, thorium, yttrium and rare earth elements from a low grade ore. Miner Eng 39:165–172. doi:10.1016/j.mineng.2012.08.002
Sui N, Huang K, Zhang C, Wang N, Wang F, Liu H (2013) Light, middle, and heavy rare-earth group separation: a new approach via a liquid-liquid-liquid three-phase system. Ind Eng Chem Res 52:5997–6008. doi:10.1021/ie4002553
Temkin VP (1940) Kinetics of ammonia synthesis on promoted iron catalysts. Acta Physiochim 12:217–222
Texier AC, Andres Y, Le Cloirec P (1999) Selective biosorption of lanthanide (La, Eu, Yb) ions by Pseudomonas aeruginosa. Environ Sci Technol 33:489–495. doi:10.1021/es9807744
Tunsu C, Ekberg C, Foreman M, Retegan T (2014) Studies on the solvent extraction of rare earth metals from fluorescent lamp waste using Cyanex 923. Solvent Extr Ion Exch 32:650–668. doi:10.1080/07366299.2014.925297
Vander Hoogerstraete T, Binnemans K (2014) Highly efficient separation of rare earths from nickel and cobalt by solvent extraction with the ionic liquid trihexyl (tetradecyl) phosphonium nitrate: a process relevant to the recycling of rare earths from permanent magnets and nickel metal hydride batteries. Green Chem 16:1594–1606. doi:10.1039/c3gc41577e
Vijayaraghavan K, Sathishkumar M, Balasubramanian R (2011) Interaction of rare earth elements with a brown marine alga in multi-component solutions. Desalination 265:54–59. doi:10.1016/j.desal.2010.07.030
Vlachou A, Symeopoulos BD, Koutinas AA (2009) A comparative study of neodymium sorption by yeast cells. Radiochim Acta 97:437–441. doi:10.1524/ract.2009.1632
Wan Ngah WS, Endud CS, Mayanar R (2002) Removal of copper(II) ions from aqueous solution onto chitosan and cross-linked chitosan beads. React Funct Polym 50:181–190
Wang J-S, Peng R-T, Yang J-H, Liu Y-C, Hu X-J (2011) Preparation of ethylenediamine-modified magnetic chitosan complex for adsorption of uranyl ions. Carbohydr Polym 84:1169–1175. doi:10.1016/j.carbpol.2011.01.007
Wu D, Zhang L, Wang L, Zhu B, Fan L (2011) Adsorption of lanthanum by magnetic alginate-chitosan gel beads. J Chem Technol Biotechnol 86:345–352. doi:10.1002/jctb.2522
Xie F, Zhang TA, Dreisinger D, Doyle F (2014) A critical review on solvent extraction of rare earths from aqueous solutions. Miner Eng 56:10–28. doi:10.1016/j.mineng.2013.10.021
Xiong C, Zheng Z (2010) Evaluation of D113 cation exchange resin for the removal of Eu(III) from aqueous solution. J Rare Earths 28:862–867. doi:10.1016/s1002-0721(09)60231-3
Xiong C, Yao C, Wang Y (2006) Sorption behaviour and mechanism of ytterbium(III) on imino-diacetic acid resin. Hydrometallurgy 82:190–194. doi:10.1016/j.hydromet.2006.03.012
Xu T, Peng H (2009) Formation cause, composition analysis and comprehensive utilization of rare earth solid wastes. J Rare Earths 27:1096–1102. doi:10.1016/s1002-0721(08)60394-4
Xu J, Chen M, Zhang C, Yi Z (2013) Adsorption of uranium(VI) from aqueous solution by diethylenetriamine-functionalized magnetic chitosan. J Radioanal Nucl Chem 298:1375–1383. doi:10.1007/s10967-013-2571-2
Xue X, Wang J, Mei L, Wang Z, Qi K, Yang B (2013) Recognition and enrichment specificity of Fe3O4 magnetic nanoparticles surface modified by chitosan and Staphylococcus aureus enterotoxins A antiserum. Colloids Surf B 103:107–113. doi:10.1016/j.colsurfb.2012.10.013
Yoon H-S et al (2014) Leaching kinetics of neodymium in sulfuric acid of rare earth elements (REE) slag concentrated by pyrometallurgy from magnetite ore. Korean J Chem Eng 31:1766–1772. doi:10.1007/s11814-014-0078-3
Zhang S-G, Yang M, Liu H, Pan D-A, Tian J-J (2013) Recovery of waste rare earth fluorescent powders by two steps acid leaching. Rare Met 32:609–615. doi:10.1007/s12598-013-0170-6
Zhao F, Repo E, Yin D, Sillanpaa MET (2013) Adsorption of Cd(II) and Pb(II) by a novel EGTA-modified chitosan material: kinetics and isotherms. J Colloid Interface Sci 409:174–182. doi:10.1016/j.jcis.2013.07.062
Zhao FP, Repo E, Sillanpaa M, Meng Y, Yin DL, Tang WZ (2015) Green synthesis of magnetic EDTA- and/or DTPA-cross-linked chitosan adsorbents for highly efficient removal of metals. Ind Eng Chem Res 54:1271–1281. doi:10.1021/ie503874x
Zhou J, Duan W, Zhou X, Zhang C (2007) Application of annular centrifugal contactors in the extraction flowsheet for producing high purity yttrium. Hydrometallurgy 85:154–162. doi:10.1016/j.hydromet.2006.08.010
Zhou L, Xu J, Liang X, Liu Z (2010) Adsorption of platinum(IV) and palladium(II) from aqueous solution by magnetic cross-linking chitosan nanoparticles modified with ethylenediamine. J Hazard Mater 182:518–524. doi:10.1016/j.jhazmat.2010.06.062
Acknowledgments
This work was supported by the French Government through a fellowship granted to A.G. by the French Embassy in Egypt (Institut Français d’Egypte). Special dedication to the memory of Prof. Dr. Ahmed Donia.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Galhoum, A.A., Mahfouz, M.G., Abdel-Rehem, S.T. et al. Diethylenetriamine-functionalized chitosan magnetic nano-based particles for the sorption of rare earth metal ions [Nd(III), Dy(III) and Yb(III)]. Cellulose 22, 2589–2605 (2015). https://doi.org/10.1007/s10570-015-0677-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-015-0677-0