Abstract
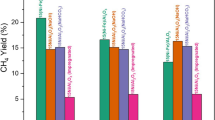
In the present study, a set of methanol synthesis catalysts with different Cu/Zn/Al or Cu/Zn/Zr molar ratios were synthesized by a co-precipitation method. Silicotungstic acid and tungstophosphoric acid were impregnated into these materials to synthesize new bifunctional catalytic materials to be used in direct synthesis of dimethyl ether (DME) from syngas. In the case of methanol production, synthesized Cu/Zn/Al catalysts exhibited quite high methanol selectivity, reaching to approximately 87%. These results indicated that the dispersion of copper particles profoundly affected the selectivity of methanol. Direct synthesis of DME was investigated in the presence of heteropoly acid impregnated copper-based novel hybrid type bifunctional catalysts. Results revealed that product distributions were strongly influenced by the reaction temperature, pressure, heteropoly acid content, and type. Results proved that silicotungstic acid (STA) impregnated bifunctional catalysts showed much better catalytic performance than the tungstophosphoric acid (TPA) impregnated ones in DME synthesis from syngas. Very high dimethyl ether selectivity values were achieved in direct synthesis of dimethyl ether over the 25%, and 30% STA impregnated methanol synthesis catalysts, at 275 °C and 50 bar. DME selectivity presented an increasing trend due to the increase in heteropoly acid content over methanol synthesis catalysts. The silicotungstic acid incorporated (30%) Cu/Zn/Al catalyst, having a composition of 6/3/1 (30STA@CZA:631), showed the highest CO conversion and DME selectivity. However, coke formation over this catalyst was much more than the catalyst containing 25% STA. This is mainly due to the higher Bronsted acidity of 30STA@CZA:631 than 25STA@CZA:631. Low coke formation, together with quite high DME selectivity values achieved with 25STA@CZA:631, is a worthy distinction of this catalyst for the direct synthesis of DME from synthesis gas.
Graphic Abstract
Novel hybrid type catalysts are successfully synthesized for direct synthesis of dimethyl ether process. Heteropoly acid incorporated methanol synthesis catalysts are very active and stable in the direct synthesis of dimethyl ether process. Silicotungstic acid (STA) impregnated copper-based catalyst exhibits a superior performance than the tungstophosphoric acid impregnated catalyst.


















Similar content being viewed by others
References
Tasdemir HM (2019) Catal Lett 149:473–485. https://doi.org/10.1007/s10562-018-2634-7
Gündüz S, Dogu T (2015) Appl Catal B 168–169:497–508. https://doi.org/10.1016/j.apcatb.2015.01.006
Semelsberger TA, Borup RL, Greene HL (2006) J Power Sources 156:497–511. https://doi.org/10.1016/j.jpowsour.2005.05.082
Ereña J, Garoña R, Arandes JM, Aguayo AT, Bilbao J (2005) Catal Today 107–108:467–473. https://doi.org/10.1016/j.cattod.2005.07.116
Woo J, Jun Y, Ho S, Yoo PJ, Lee DH, Jun KW, Bae WB (2012) Appl Catal B 126:1–8. https://doi.org/10.1016/j.apcatb.2012.06.026
Song F, Tan Y, Xie H, Zhang Q, Han Y (2014) Fuel Process Technol 126:88–94. https://doi.org/10.1016/j.fuproc.2014.04.021
Mao D, Yang W, Xia J, Zhang B, Song Q, Chen Q (2005) J Catal 230:140–149. https://doi.org/10.1016/j.jcat.2004.12.007
Erena J, Sierra I, Aguayo AT, Ateka A, Olaazar M, Bilbao J (2011) Chem Eng J 174:660–667. https://doi.org/10.1016/j.cej.2011.09.067
Erena J, Garona R, Arandes JM, Aguayo AT, Bilbao J (2005) Int J Chem React Eng. https://doi.org/10.2202/1542-6580.1295
Wang L, Fang D, Huang X, Zhang S, Qi Y, Liul Z (2006) J Nat Gas Chem 15:38–44. https://doi.org/10.1016/S1003-9953(06)60005-4
Sang K, Kim J, Park M, Kim S, Joo O, Jung K (2007) Appl Catal A 330:57–62. https://doi.org/10.1016/j.apcata.2007.07.007
Bayat A, Dogu T (2016) Ind Eng Chem Res 55:11431–11439. https://doi.org/10.1021/acs.iecr.6b03001
Arbag H (2018) Int J Hydrogen Energy 43:6561–6574. https://doi.org/10.1016/j.ijhydene.2018.02.063
Ciftci A, Varisli D, Dogu T (2010) Int J Chem React Eng. https://doi.org/10.2202/1542-6580.2151
Varisli D, Dogu T, Dogu G (2010) Chem Eng Sci 65:153–159. https://doi.org/10.1016/j.ces.2009.01.066
Varisli D, Dogu T, Dogu G (2008) Ind Eng Chem Res 47:4071–4076. https://doi.org/10.1021/ie800192t
Lei H, Hou Z, Xie J (2016) Fuel 164:191–198. https://doi.org/10.1016/j.fuel.2015.09.082
Dibenedetto A, Abgelini A, Stufano P (2013) J Chem Technol Biotechnol 89:334–353. https://doi.org/10.1002/jctb.4229
Witoon T, Permsirivanich T, Donphai W (2013) Fuel Process Technol 116:72–78. https://doi.org/10.1016/j.fuproc.2013.04.024
Baltes C, Vukojevis S, Schüth F (2008) J Catal 258:334–344. https://doi.org/10.1016/j.jcat.2008.07.004
Grabowski R, Olszewski P, Kozłowska A, Stoch J, Lachowska M, Skrzypek J (2006) Appl Catal A 310:127–137. https://doi.org/10.1016/j.apcata.2006.05.035
Gao P, Li F, Zhao N, Xiao F, Wei W, Zhong L (2013) Appl Catal A 468:442–452. https://doi.org/10.1016/j.apcata.2013.09.026
Angelo L, Kobl K, Marcela L, Tejada M, Zimmermann Y, Parkhomenko K (2015) C R Chim 18:250–260. https://doi.org/10.1016/j.crci.2015.01.001
Zhang L, Zhang Y, Chen S (2012) Appl Catal A 415–416:118–123. https://doi.org/10.1016/j.apcata.2011.12.013
Fujitani T, Nakamura J (1998) Catal Lett 56:119–124. https://doi.org/10.1023/A:1019000927366
Moradi G, Ahmadpour J, Nazari M, Yaripour F (2008) Ind Eng Chem Res 47:7672–7679. https://doi.org/10.1021/ie800888z
Celik G, Arinan A, Bayat A, Ozbelge HO, Dogu T, Varisli D (2013) Top Catal 56:1764–1774. https://doi.org/10.1007/s11244-013-0112-4
Phienluphon R, Pinkaew K, Yang G, Li J, Wei Q (2015) Chem Eng J 270:605–611. https://doi.org/10.1016/j.cej.2015.02.071
Contador MS, Ateka A, Ibane M, Bilbao J, Aguayo AT (2019) Renew Energy 138:585–597. https://doi.org/10.1016/j.renene.2019.01.093
Pinkaew K, Yang G, Vitidsant T, Jin Y, Zeng C (2013) Fuel 111:727–732. https://doi.org/10.1016/j.fuel.2013.03.027
Nie R, Lei H, Pan S, Wang L, Fei J, Hou Z (2012) Fuel 96:419–425. https://doi.org/10.1016/j.fuel.2011.12.048
Saito M, Fujitani T, Takeuchi M, Watanabe T (1996) Appl Catal A 138:311–318. https://doi.org/10.1016/0926-860X(95)00305-3
Pekmezci B, Cakiryilmaz N, Arbag H, Oktar N, Dogu G, Dogu T (2017) Int J Hydrogen Energy 42:26257–26269. https://doi.org/10.1016/j.ijhydene.2017.08.155
Tasdemir HM, Yagızatlı Y, Yasyerli S, Yasyerli N (2019) Int J Chem React Eng. https://doi.org/10.1515/ijcre-2018-0157
Li D, Xu S, Cai Y, Chen C, Zhan Y, Jiang L (2017) Ind Eng Chem Res 56:3175–3183. https://doi.org/10.1021/acs.iecr.6b04337
Kurr P, Kasatkin I, Girgsdies F, Trunschke A, Ressler T, Schlo R (2008) Appl Catal A 348:153–164. https://doi.org/10.1016/j.apcata.2008.06.020
Ciftci A, Varisli D, Tokay KC, Sezgi NA, Dogu T (2012) Chem Eng J 207–208:85–93. https://doi.org/10.1016/j.cej.2012.04.016
Basahel SN, Mokhtar M, Alsharaeh EH, Ali TT, Mahmoud HA, Narasimharao K (2016) Catalysts 6:57. https://doi.org/10.3390/catal6040057
Guo W, Duan Z, Mabayoje O, Chemelewski W, Xiao P, Henkelman G, Zhang Y, Mullins CB (2016) J Electrochem Soc 163(10):H970–H975. https://doi.org/10.1149/2.0701610jes
Ikizer B, Oktar N, Dogu T (2010) Fuel Process Technol 138:570–577. https://doi.org/10.1016/j.fuproc.2015.06.039
Iyer SS, Renganathan T, Pushpavanam S, Vasudeva M (2015) Biochem Pharmacol 10:95–104. https://doi.org/10.1016/j.jcou.2015.01.006
Liu X, Lu GQ, Yan Z (2003) Beltramini. J Ind Eng Chem Res 42:6518–6530. https://doi.org/10.1021/ie020979s
Carter JH, Liu X, He Q, Althahban S, Nowicka E, Freakley SJ, Niu L, Morgan DJ, Li Y, Niemantsverdriet JWH, Golunski S, Kiely CJ, Hutchings GJ (2017) Angew Chem Int Ed 56:16037–16041. https://doi.org/10.1002/anie.201709708
Obalı Z, Sezgi NA, Dogu T (2009) Chem Eng Commun 196:116–130. https://doi.org/10.1080/00986440802301537
Erdogan B, Arbag H, Yasyerli N (2018) Int J Hydrog Energy 43:1396–1405. https://doi.org/10.1016/j.ijhydene.2017.11.127
Arbag H, Yasyerli S, Yasyerli N, Dogu G, Dogu T, Crnivec IGO (2015) Ind Eng Chem Res 54:2290–2301
Acknowledgements
TUBITAK (115M377) and Gazi University Research Fund (06/2017-09 and 06/2018-23) were gratefully acknowledged for financial support. The authors also thank the Central Laboratory of METU, Prof. Dr. Emrah OZENSOY Research Group, Messaoud Harfouche, and SESAME Synchrotron Laboratory for some of the characterization results.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest for each contributing author.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pekmezci Karaman, B., Oktar, N., Doğu, G. et al. Bifunctional Silicotungstic Acid and Tungstophosphoric Acid Impregnated Cu–Zn–Al & Cu–Zn–Zr Catalysts for Dimethyl Ether Synthesis from Syngas. Catal Lett 150, 2744–2761 (2020). https://doi.org/10.1007/s10562-020-03171-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-020-03171-6