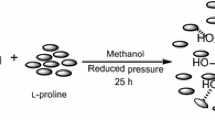
Abstract
A novel chiral BINOL–silica hybrid has been successfully prepared by the reaction of (S)-BINOL and SiCl4 following by gel polymerization under atmosphere condition. The synthesized catalyst was characterized by elemental analysis, Fourier-transform infrared spectroscopy, X-ray diffraction, scanning electron microscopy and energy-dispersive X-ray spectroscopy. Catalytic activity of the chiral BINOL–silica hybrid for diastereo- and enantioselective Diels–Alder and aza Michael reactions has been investigated. Mild reaction conditions, high yields, excellent diastereo- and enantiomeric excess make this powerful and effective catalyst as an attractive option for the synthesis of chiral organic compounds.
Graphical Abstract









Similar content being viewed by others
References
Wan KT, Davis ME (1994) Nature 370:449–450
Ohmatsu K, Ooi T (2015) Tetrahedron Lett 56:2043–2048
Ding K, Uozumi Y (2008) Handbook of asymmetric heterogeneous catalysis. Wiley, Weinheim
Ojima I (2000) Catalytic asymmetric synthesis, 2nd edn. Wiley, New York
Jacobsen EN, Pfaltz A, Yamamoto Y (1999) Comprehensive asymmetric catalysis. Springer, Berlin
Blaser HU (1991) Tetrahedron 2:843–866
Jannes G, Vincent D (1995) Chiral reactions in heterogeneous catalysis. Springer, Boston
Bhaduri S, Mukesh D (2014) Homogeneous catalysis: mechanisms and industrial applications, 2nd edn. Wiley, Weinheim
Blaser HU (1991) Tetrahedron 9:843–866
Gladysz JA (2002) Chem Rev 102:3215 – 3892
Baleizao C, Garcia H (2006) Chem Rev 106:3987–4043
Hutchings G (2004) Chem Soc Rev 33:108–122
Hahn R, Raabe G, Enders D (2006) Angew Chem Int Ed 45:4732–4762
Corma A, Garcia H (2003) Chem Rev 103:4307–4365
Ramazani A, Asiabi PA, Aghahosseini H, Gouranlou F (2017) Curr Org Chem 21:908–922
Li C, Liu Y (2014) Bridging heterogeneous and homogeneous catalysis: concepts, strategies, and applications, 1st edn. Wiley, Weinheim
Shylesh S, Thiel WR (2011) ChemCatChem 3:278–287
Margelefsky EL, Zeidan RK, Davis MK (2008) Chem Soc Rev 37:1118–1126
Haas KH (2000) Adv Eng Mater 2:571–582
Sanchez C, Ribot F (1994) New J Chem 18:1007–1047
Cauqui MA, Rodriguez-Izquierdo JM (1992) J Non-Cryst Solids 147–148:724–738
Brinker CH, Scherer GW (1990) Sol-gel science. Academic Press, London
Adima A, Moreau JJE, Wong C, Man M (2000) Chirality 12:411–420
Loy DA, Shea KJ (1995) Chem Rev 95:1431–1442
Fardood ST, Ramezani A, Moradi S (2017) J Sol-Gel Sci Technol 82:432–439
Schmidt HK (1997) J Sol-Gel Sci Technol 8:557–565
Shea KJ, Loy DA, Webster OW (1992) J Am Chem Soc114:6700–6710
Shea KJ, Loy DA (2001) Chem Mater 13:3306–3319
Chen Y, Yekta S, Yudin AK (2003) Chem Rev 103:3155–3211
Noyori R (1996) Acta Chem Scand 50:380–390
Mikami K, Itoh Y, Yamanaka M (2004) Chem Rev 104:1–16
Walsh P (2003) Chem Rev 103:3297–3344
Rouhani M, Ramazani A, Joo SW (2014) Ultrason Sonochem 21:262–267
Aghahosseini H, Ramazani A, Ślepokura K, Lis T (2018) J Colloid Interface Sci 511:222–232
Moyano A, Rios R (2011) Chem Rev 111:4703–4832
Nigam M, Rush B, Patel J, Castillo R, Dhar P (2016) J Chem Educ 93:753–756
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Electronic Supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Saeidian, H., Paghandeh, H., Parvin, Z. et al. Facile Synthesis of a New Chiral BINOL–Silica Hybrid Catalyst for Asymmetric Diels–Alder and Aza Michael Reactions. Catal Lett 148, 1366–1374 (2018). https://doi.org/10.1007/s10562-018-2346-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-018-2346-z