Abstract
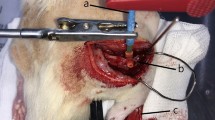
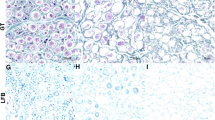
The use of processed nerve allografts as an alternative to autologous nerve grafts, the gold standard treatment for peripheral nerve defects, is increasing. However, it is not widely used in Korea due to cost and insurance issues. Moreover, the main detergent used in the conventional Hudson method is unavailable. Therefore, a new nerve allograft decellularization process is needed. We aimed to compare the traditional Hudson method with a novel decellularization process that may remove cellular content more efficiently while preserving the extracellular matrix (ECM) structure using low concentration sodium dodecyl sulfate (SDS) and nuclease. After each decellularization process, DNA content was measured in nerve tissue. Masson's trichrome staining and scanning electron microscopy were performed to determine the state of preservation of the ECM. A significantly greater amount of DNA content was removed in the novel method, and the ECM structure was preserved in both methods. For the in vivo study, a 15-mm long sciatic nerve defect was created in two groups of Sprague–Dawley rats, and processed nerve allografts decellularized using the Hudson or novel method were transplanted. Functional and histological recovery results were measured 12 weeks post-transplantation. Ankle contracture angle, maximal isometric tetanic force of the tibialis anterior (TA), and the TA mass were compared between the groups, as well as the percent neural tissue (100 × neural area/intrafascicular area). There was no significant difference in functional and histological nerve recovery between the methods. The novel method is appropriate for developing a processed nerve allograft.








Similar content being viewed by others
Availability of data and material
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Brooks DN, Weber RV, Chao JD, Rinker BD, Zoldos J, Robichaux MR, Ruggeri SB, Anderson KA, Bonatz EE, Wisotsky SM, Cho MS, Wilson C, Cooper EO, Ingari JV, Safa B, Parrett BM, Buncke GM (2012) Processed nerve allografts for peripheral nerve reconstruction: a multicenter study of utilization and outcomes in sensory, mixed, and motor nerve reconstructions. Microsurgery 32:1–14. https://doi.org/10.1002/micr.20975
Cebotari S, Tudorache I, Jaekel T, Hilfiker A, Dorfman S, Ternes W, Haverich A, Lichtenberg A (2010) Detergent decellularization of heart valves for tissue engineering: toxicological effects of residual detergents on human endothelial cells. Artif Organs 34:206–210. https://doi.org/10.1111/j.1525-1594.2009.00796.x
Crapo PM, Gilbert TW, Badylak SF (2011) An overview of tissue and whole organ decellularization processes. Biomaterials 32:3233–3243. https://doi.org/10.1016/j.biomaterials.2011.01.057
Du L, Wu X, Pang K, Yang Y (2011) Histological evaluation and biomechanical characterisation of an acellular porcine cornea scaffold. Br J Ophthalmol 95:410–414. https://doi.org/10.1136/bjo.2008.142539
Freed AD, Doehring TC (2005) Elastic model for crimped collagen fibrils. J Biomech Eng 127:587–593. https://doi.org/10.1115/1.1934145
Han LW, Xu G, Guo MY, Chang YA, Zhang Y, Zhao YT, Li ZH (2020) Comparison of SB-SDS and other decellularization methods for the acellular nerve graft: biological evaluation and nerve repair in vitro and in vivo. Synapse 74:e22143. https://doi.org/10.1002/syn.22143
Hudson TW, Liu SY, Schmidt CE (2004a) Engineering an improved acellular nerve graft via optimized chemical processing. Tissue Eng 10:1346–1358. https://doi.org/10.1089/ten.2004.10.1641
Hudson TW, Zawko S, Deister C, Lundy S, Hu CY, Lee K, Schmidt CE (2004b) Optimized acellular nerve graft is immunologically tolerated and supports regeneration. Tissue Eng 10:1641–1651. https://doi.org/10.1089/ten.2004.10.1641
Karlsson G, Hinz AC, Henriksson E, Winge S (2002) Determination of triton X-100 in plasma-derived coagulation factor VIII and factor IX products by reversed-phase high-performance liquid chromatography. J Chromatogr A 946:163–168. https://doi.org/10.1016/s0021-9673(01)01565-5
Keane TJ, Swinehart IT, Badylak SF (2015) Methods of tissue decellularization used for preparation of biologic scaffolds and in vivo relevance. Methods 84:25–34. https://doi.org/10.1016/j.ymeth.2015.03.005
Korossis SA, Booth C, Wilcox HE, Watterson KG, Kearney JN, Fisher J, Ingham E (2002) Tissue engineering of cardiac valve prostheses II: biomechanical characterization of decellularized porcine aortic heart valves. J Heart Valve Dis 11:463–471
Lee JY, Giusti G, Wang H, Friedrich PF, Bishop AT, Shin AY (2013) Functional evaluation in the rat sciatic nerve defect model: a comparison of the sciatic functional index, ankle angles, and isometric tetanic force. Plast Reconstr Surg 132:1173–1180. https://doi.org/10.1097/PRS.0b013e3182a3bfeb
Lin FM, Pan YC, Hom C, Sabbahi M, Shenaq S (1996) Ankle stance angle: a functional index for the evaluation of sciatic nerve recovery after complete transection. J Reconstr Microsurg 12:173–177. https://doi.org/10.1055/s-2007-1006472
Lumpkins SB, Pierre N, McFetridge PS (2008) A mechanical evaluation of three decellularization methods in the design of a xenogeneic scaffold for tissue engineering the temporomandibular joint disc. Acta Biomater 4:808–816. https://doi.org/10.1016/j.actbio.2008.01.016
Means KR Jr, Rinker BD, Higgins JP, Payne SH Jr, Merrell GA, Wilgis EFS (2016) A multicenter, prospective, randomized, pilot study of outcomes for digital nerve repair in the hand using hollow conduit compared with processed allograft nerve. Hand (N Y) 11:144–151. https://doi.org/10.1177/1558944715627233
Moore AM, MacEwan M, Santosa KB, Chenard KE, Ray WZ, Hunter DA, Mackinnon SE, Johnson PJ (2011) Acellular nerve allografts in peripheral nerve regeneration: a comparative study. Muscle Nerve 44:221–234. https://doi.org/10.1002/mus.22033
Nakayama KH, Batchelder CA, Lee CI, Tarantal AF (2010) Decellularized rhesus monkey kidney as a three-dimensional scaffold for renal tissue engineering. Tissue Eng Part A 16:2207–2216. https://doi.org/10.1089/ten.tea.2009.0602
Neubauer D, Graham JB, Muir D (2007) Chondroitinase treatment increases the effective length of acellular nerve grafts. Exp Neurol 207:163–170. https://doi.org/10.1016/j.expneurol.2007.06.006
Rbia N, Bulstra LF, Saffari TM, Hovius SER, Shin AY (2019) Collagen nerve conduits and processed nerve allografts for the reconstruction of digital nerve gaps: a single-institution case series and review of the literature. World Neurosurg 127:e1176–e1184. https://doi.org/10.1016/j.wneu.2019.04.087
Rinker B, Zoldos J, Weber RV, Ko J, Thayer W, Greenberg J, Leversedge FJ, Safa B, Buncke G (2017) Use of processed nerve allografts to repair nerve injuries greater than 25 mm in the hand. Ann Plast Surg 78:S292–S295. https://doi.org/10.1097/SAP.0000000000001037
Schaner PJ, Martin ND, Tulenko TN, Shapiro IM, Tarola NA, Leichter RF, Carabasi RA, Dimuzio PJ (2004) Decellularized vein as a potential scaffold for vascular tissue engineering. J Vasc Surg 40:146–153. https://doi.org/10.1016/j.jvs.2004.03.033
Shin RH, Vathana T, Giessler GA, Friedrich PF, Bishop AT, Shin AY (2008) Isometric tetanic force measurement method of the tibialis anterior in the rat. Microsurgery 28:452–457. https://doi.org/10.1002/micr.20520
Shin YH, Park SY, Kim JK (2019) Comparison of systematically combined detergent and nuclease-based decellularization methods for acellular nerve graft: an ex vivo characterization and in vivo evaluation. J Tissue Eng Regen Med 13:1241–1252. https://doi.org/10.1002/term.2874
Sondell M, Lundborg G, Kanje M (1998) Regeneration of the rat sciatic nerve into allografts made acellular through chemical extraction. Brain Res 795:44–54. https://doi.org/10.1016/s0006-8993(98)00251-0
Whitlock EL, Tuffaha SH, Luciano JP, Yan Y, Hunter DA, Magill CK, Moore AM, Tong AY, Mackinnon SE, Borschel GH (2009) Processed allografts and type I collagen conduits for repair of peripheral nerve gaps. Muscle Nerve 39:787–799. https://doi.org/10.1002/mus.21220
Williams C, Liao J, Joyce EM, Wang B, Leach JB, Sacks MS, Wong JY (2009) Altered structural and mechanical properties in decellularized rabbit carotid arteries. Acta Biomater 5:993–1005. https://doi.org/10.1016/j.actbio.2008.11.028
Wilshaw SP, Rooney P, Berry H, Kearney JN, Homer-Vanniasinkam S, Fisher J, Ingham E (2012) Development and characterization of acellular allogeneic arterial matrices. Tissue Eng Part A 18:471–483. https://doi.org/10.1089/ten.tea.2011.0287
Zilic L, Wilshaw SP, Haycock JW (2016) Decellularisation and histological characterisation of porcine peripheral nerves. Biotechnol Bioeng 113:2041–2053. https://doi.org/10.1002/bit.25964
Acknowledgements
We would like to thank Editage (www.editage.co.kr) for English language editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that there is no conflict of interest.
Ethical approval
All of animal care and experimental procedures were approved by the Catholic University of Korea College of Medicine, St. Vincent’s Hospital Institutional Animal Care and Use Committee (IRB17-7).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kang, HV., Im, JH., Chung, YG. et al. Comparison of two different decellularization methods for processed nerve allograft. Cell Tissue Bank 22, 575–585 (2021). https://doi.org/10.1007/s10561-021-09965-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-021-09965-1