Abstract
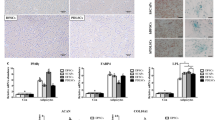
Stem-cell-based therapies were introduced aiming to overcome the limitations of the existing procedures for regeneration of mineralized tissues. Stem cells isolated from the endometrial tissue and dental pulp have the capacity to differentiate into various functional cells including osteoblasts. However, studies comparing their ability to regenerate mineralized tissue are lacking. The purpose of this study was to compare the proliferation and osteogenic differentiation potential of endometrial stem cells (EnSCs) and dental pulp stem cells (DPSCs) using in vitro cell culture technique. The DPSCs and EnSCs were isolated from human dental pulp and endometrium, respectively. Their proliferation and osteogenic potential were compared in the same osteogenic medium (OM) after 3, 5, 7 and 10 days using the methyl thiazol tetrazolium assay, alizarin red staining, and real-time quantitative reverse transcription polymerase chain reaction (Real-Time qRT-PCR). The EnSCs showed higher proliferation rate compared to DPSCs. Regarding osteogenesis, alizarin red-positive colonies appeared earlier and in greater amounts in DPSCs group. The real-time qRT-PCR demonstrated significantly greater osteogenic potential of DPSCs compared to EnSCs. Our findings revealed significant differences in stem cell properties based on the tissue source. The EnSCs had greater proliferation rate than DPSCs, while DPSCs showed greater osteogenic potential compared to EnSCs in the same OM.



Similar content being viewed by others
References
Alfotawei R, Naudi KB, Lappin D, Barbenel J, Di Silvio L, Hunter K, McMahon J, Ayoub A (2014) The use of TriCalcium Phosphate (TCP) and stem cells for the regeneration of osteoperiosteal critical-size mandibular bony defects, an in vitro and preclinical study. J Craniomaxillofac Surg 42:863–869. doi:10.1016/j.jcms.2013.12.006
Alge DL, Zhou D, Adams LL, Wyss BK, Shadday MD, Woods EJ, Gabriel Chu TM, Goebel WS (2010) Donor-matched comparison of dental pulp stem cells and bone marrow-derived mesenchymal stem cells in a rat model. J Tissue Eng Regen Med 4:73–81. doi:10.1002/term.220
Bilezikian JP, Raisz LG, Martin TJ (2008) Principles of bone biology: two-volume set. Academic Press, Cambridge
Caplan AI (2007) Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. J Cell Physiol 213:341–347. doi:10.1002/jcp.21200
Davies OG, Cooper PR, Shelton RM, Smith AJ, Scheven BA (2015) A comparison of the in vitro mineralisation and dentinogenic potential of mesenchymal stem cells derived from adipose tissue, bone marrow and dental pulp. J Bone Miner Metab 33(4):371–382. doi:10.1007/s00774-014-0601-y
De Ugarte DA, Morizono K, Elbarbary A, Alfonso Z, Zuk PA, Zhu M, Dragoo JL, Ashjian P, Thomas B, Benhaim P, Chen I, Fraser J, Hedrick MH (2003) Comparison of multi-lineage cells from human adipose tissue and bone marrow. Cells Tissues Organs 174(3):101–109. doi:10.1159/000071150
Elsalanty ME, Genecov DG (2009) Bone grafts in craniofacial surgery. Craniomaxillofac Trauma Reconstr 2(3):125–134. doi:10.1055/s-0029-1215875
Gargett CE, Schwab KE, Zillwood RM, Nguyen HPT, Wu D (2009) Isolation and culture of epithelial progenitors and mesenchymal stem cells from human endometrium. Biol Reprod 80(6):1136–1145. doi:10.1095/biolreprod.108.075226
Hakki SS, Kayis SA, Hakki EE, Bozkurt SB, Duruksu G, Unal ZS, Turaç G, Karaoz E (2015) Comparison of mesenchymal stem cells isolated from pulp and periodontal ligament. J Periodontol 86(2):283–291. doi:10.1902/jop.2014.140257
Kanafi M, Ramesh A, Gupta P, Bhonde R (2014) Dental pulp stem cells immobilized in alginate microspheres for applications in bone tissue engineering. Int Endod J 47(7):687–697. doi:10.1111/iej.12205
Li J-H, Liu D-Y, Zhang F-M, Wang F, Zhang W-K, Zhang Z-T (2011) Human dental pulp stem cell is a promising autologous seed cell for bone tissue engineering. Chin Medical J (Engl) 124(23):4022–4028
Lin Y, Luo E, Chen X, Liu L, Qiao J, Yan Z, Li Z, Tang W, Zheng X, Tian W (2005) Molecular and cellular characterization during chondrogenic differentiation of adipose tissue-derived stromal cells in vitro and cartilage formation in vivo. J Cell Mol Med 9(4):929–939. doi:10.1111/j.1582-4934.2005.tb00389.x
Liu TM, Martina M, Hutmacher DW, Hui JH, Lee EH, Lim B (2007) Identification of common pathways mediating differentiation of bone marrow- and adipose tissue-derived human mesenchymal stem cells into three mesenchymal lineages. Stem Cells 25(3):750–760. doi:10.1634/stemcells.2006-0394
Mutlu L, Hufnagel D, Taylor HS (2015) The endometrium as a source of mesenchymal stem cells for regenerative medicine. Biol Reprod 92(6):138. doi:10.1095/biolreprod.114.126771
Nandi S, Roy S, Mukherjee P, Kundu B, De D, Basu D (2010) Orthopaedic applications of bone graft & graft substitutes: a review. Indian J Med Res 132:15–30
Ponnaiyan D, Jegadeesan V (2014) Comparison of phenotype and differentiation marker gene expression profiles in human dental pulp and bone marrow mesenchymal stem cells. Eur J Dent 8(3):307–313. doi:10.4103/1305-7456.137631
Quarto R, Mastrogiacomo M, Cancedda R, Kutepov SM, Mukhachev V, Lavroukov A, Kon E, Marcacci M (2001) Repair of large bone defects with the use of autologous bone marrow stromal cells. N Engl J Med 344(5):385–386. doi:10.1056/NEJM200102013440516
Sakaguchi Y, Sekiya I, Yagishita K, Muneta T (2005) Comparison of human stem cells derived from various mesenchymal tissues: superiority of synovium as a cell source. Arthritis Rheum 52(8):2521–2529. doi:10.1002/art.21212
Schwab K, Gargett C (2007) Co-expression of two perivascular cell markers isolates mesenchymal stem-like cells from human endometrium. Human Reprod 22(11):2903–2911. doi:10.1093/humrep/dem265
Schwab KE, Chan RWS, Gargett CE (2005) Putative stem cell activity of human endometrial epithelial and stromal cells during the menstrual cycle. Fertil Steril 84:1124–1130. doi:10.1016/j.fertnstert.2005.02.056
Shi S, Gronthos S (2003) Perivascular niche of postnatal mesenchymal stem cells in human bone marrow and dental pulp. J Bone Miner Res 18(4):696–704. doi:10.1359/jbmr.2003.18.4.696
Stenderup K, Justesen J, Clausen C, Kassem M (2003) Aging is associated with decreased maximal life span and accelerated senescence of bone marrow stromal cells. Bone 33(6):919–926. doi:10.1016/j.bone.2003.07.005
Tabatabaei FS, Ai J, Kashi TSJ, Khazaei M, Kajbafzadeh A-M, Ghanbari Z (2013a) Effect of dentine matrix proteins on human endometrial adult stem-like cells: in vitro regeneration of odontoblasts cells. Arch Oral Biol 58(7):871–879. doi:10.1016/j.archoralbio.2013.01.013
Tabatabaei FS, Dastjerdi MV, Jazayeri M, Haghighipour N, Dastjerdie EV, Bordbar M (2013b) Comparison of osteogenic medium and uniaxial strain on differentiation of endometrial stem cells. Dent Res J (Isfahan) 10(2):190–196
Tabatabaei F, Jazayeri M, Ghahari P, Haghighipour N (2014) Effects of equiaxial strain on the differentiation of dental pulp stem cells without using biochemical reagents. Mol Cell Biomech 11(3):209–220
Takahashi M, Yamada Y, Ozawa R, Ohya M, Ito K, Ueda M (2004) Expression of odontoblastic-related genes in human dental follicle cells, dental pulp stem cells, and oral mucosal cells. Int J Oral-Med Sci 3(1):41–48. doi:10.5466/ijoms.3.41
Wexler SA, Donaldson C, Denning-Kendall P, Rice C, Bradley B, Hows JM (2003) Adult bone marrow is a rich source of human mesenchymal ‘stem’cells but umbilical cord and mobilized adult blood are not. Br J Haematol 121(2):368–374
Yoshimura H, Muneta T, Nimura A, Yokoyama A, Koga H, Sekiya I (2007) Comparison of rat mesenchymal stem cells derived from bone marrow, synovium, periosteum, adipose tissue, and muscle. Cell Tissue Res 327(3):449–462. doi:10.1007/s00441-006-0308-z
Acknowledgements
This study was financially supported by the School of Dentistry, Shahid Beheshti University of Medical Sciences, Tehran, Iran (Grant No. 9009).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Tabatabaei, F.S., Torshabi, M. In vitro proliferation and osteogenic differentiation of endometrial stem cells and dental pulp stem cells. Cell Tissue Bank 18, 239–247 (2017). https://doi.org/10.1007/s10561-017-9620-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-017-9620-y