Abstract
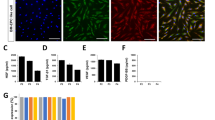
Endothelial progenitor cells (EPC) derived from the circulation may be used to enhance neovascularization. Since the combination of granulocyte colony-stimulating factor (GCSF) and CXCR4 antagonist AMD3100 efficiently mobilizes hematopoietic stem cells into peripheral circulation, it may increase the pool of endogenously circulating EPC. We tested this hypothesis by administering GCSF and AMD3100 to adult rabbits and rats, isolating mononuclear cells from peripheral blood by Ficoll density gradient centrifugation, and characterizing the blood-derived EPC based on morphology, immunophenotyping, gene expression and other functional analyses. These EPC showed clonal growth similar to that of human umbilical vein endothelial cells when cultured in complete EGM-2 medium on collagen I-precoated culture plates. The EPC exhibited a typical cobblestone-like morphology and were relatively homogeneous by the third passage. The cells expressed the typical endothelial marker CD31 based on flow cytometry and fluorescence microscopy, formed capillary-like structures when cultured in Matrigel, internalized DiI-acetylated low-density lipoprotein, bound Ulex europaeus agglutinin-1, and expressed CD31 and several other endothelial markers (VEGFR2, VE-cadherin, Tie-2, eNOS, vWF) at significantly higher levels than bone marrow-derived mesenchymal stem cells. These results suggest that the combination of GCSF and AMD3100 can efficiently release stem cells into peripheral circulation and generate EPC that show the desired morphological, immunophenotypic and functional characteristics. This minimally invasive approach may be useful for autologous cell transplantation for postnatal neovasculogenesis and tissue repair.





Similar content being viewed by others
References
Amini AR, Laurencin CT, Nukavarapu SP (2012) Differential analysis of peripheral blood- and bone marrow-derived endothelial progenitor cells for enhanced vascularization in bone tissue engineering. J Orthop Res 30:1507–1515. doi:10.1002/jor.22097
Baudin B, Bruneel A, Bosselut N, Vaubourdolle M (2007) A protocol for isolation and culture of human umbilical vein endothelial cells. Nat Protoc 2:481–485. doi:10.1038/nprot.2007.54
Boilson BA et al (2008) Endothelial progenitor cells exist in abundance in adipose tissue but not in peripheral blood. J Am Coll Cardiol 51:A295–A295
Campanelli R et al (2010) G-Csf alone or in combination with chemotherapy does not mobilize endothelial progenitor cells into peripheral blood. Haematol-Hematol J 95:289
Chen JK, Deng YP, Jiang GJ, Liu YZ, Zhao T, Shen FM (2013) Establishment of tube formation assay of bone marrow-derived endothelial progenitor cells. CNS Neurosci Ther 19:533–535. doi:10.1111/cns.12102
Damon LE, Damon LE (2009) Mobilization of hematopoietic stem cells into the peripheral blood. Expert Rev Hematol 2:717–733. doi:10.1586/ehm.09.54
Das R, Jahr H, van Osch GJ, Farrell E (2010) The role of hypoxia in bone marrow-derived mesenchymal stem cells: considerations for regenerative medicine approaches. Tissue Eng B Rev 16:159–168. doi:10.1089/ten.TEB.2009.0296
Dome B et al (2009) Circulating endothelial cells, bone marrow-derived endothelial progenitor cells and proangiogenic hematopoietic cells in cancer: from biology to therapy. Crit Rev Oncol Hemat 69:108–124. doi:10.1016/j.critrevonc.2008.06.009
Duong HT, Erzurum SC, Asosingh K (2011) Pro-angiogenic hematopoietic progenitor cells and endothelial colony-forming cells in pathological angiogenesis of bronchial and pulmonary circulation. Angiogenesis 14:411–422. doi:10.1007/s10456-011-9228-y
Grisar JC, Haddad F, Gomari FA, Wu JC (2011) Endothelial progenitor cells in cardiovascular disease and chronic inflammation: from biomarker to therapeutic agent. Biomarkers Med 5:731–744. doi:10.2217/bmm.11.92
Gulati R, Jevremovic D, Peterson TE, Chatterjee S, Shah V, Vile RG, Simari RD (2003) Diverse origin and function of cells with endothelial phenotype obtained from adult human blood. Circ Res 93:1023–1025. doi:10.1161/01.Res.0000105569.77539.21
Hirschi KK, Ingram DA, Yoder MC (2008) Assessing identity, phenotype, and fate of endothelial progenitor cells. Arterioscler Thromb Vasc Biol 28:1584–1595. doi:10.1161/ATVBAHA.107.155960
Honold J et al (2006a) Effects of granulocyte colony simulating factor on functional activities of endothelial progenitor cells in patients with chronic ischemic heart disease. Arterioscler Thromb Vasc Biol 26:2238–2243. doi:10.1161/01.ATV.0000240248.55172.dd
Honold J et al (2006b) Effects of granulocyte colony stimulating factor on functional activities of endothelial progenitor cells in patients with chronic ischemic heart disease. Arterioscler Throm Vasc Biol 26:2238–2243. doi:10.1161/01.Atv.0000240248.55172.Dd
Hur J et al (2004) Characterization of two types of endothelial progenitor cells and their different contributions to neovasculogenesis. Arterioscler Throm Vasc Biol 24:288–293. doi:10.1161/01.Atv.0000114236.77009.06
Ingram DA et al (2004) Identification of a novel hierarchy of endothelial progenitor cells using human peripheral and umbilical cord blood. Blood 104:2752–2760. doi:10.1182/blood-2004-04-1396
Kolonin MG, Simmons PJ (2009) Combinatorial stem cell mobilization. Nat Biotechnol 27:252–253. doi:10.1038/nbt0309-252
Lin RZ, Dreyzin A, Aamodt K, Dudley AC, Melero-Martin JM (2011) Functional endothelial progenitor cells from cryopreserved umbilical cord blood. Cell Transplant 20:515–522. doi:10.3727/096368910X532729
Llevadot J et al (2001) HMG-CoA reductase inhibitor mobilizes bone marrow-derived endothelial progenitor cells. J Clin Invest 108:399–405. doi:10.1172/Jci13131
Martin-Ramirez J, Hofman M, van den Biggelaar M, Hebbel RP, Voorberg J (2012) Establishment of outgrowth endothelial cells from peripheral blood. Nat Protoc 7:1709–1715. doi:10.1038/nprot.2012.093
Medina RJ, O’Neill CL, Sweeney M, Guduric-Fuchs J, Gardiner TA, Simpson DA, Stitt AW (2010) Molecular analysis of endothelial progenitor cell (EPC) subtypes reveals two distinct cell populations with different identities. BMC Med Genomics. doi:10.1186/1755-8794-3-18
Peichev M et al (2000) Expression of VEGFR-2 and AC133 by circulating human CD34(+) cells identifies a population of functional endothelial precursors. Blood 95:952–958
Pelus LM (2008) Peripheral blood stem cell mobilization: new regimens, new cells, where do we stand. Curr Opin Hematol 15:285–292. doi:10.1097/MOH.0b013e328302f43a
Petit I et al (2002) G-CSF induces stem cell mobilization by decreasing bone marrow SDF-1 and up-regulating CXCR4. Nat Immunol 3:687–694. doi:10.1038/ni813
Pitchford SC, Furze RC, Jones CP, Wengner AM, Rankin SM (2009) Differential mobilization of subsets of progenitor cells from the bone marrow. Cell Stem Cell 4:62–72. doi:10.1016/j.stem.2008.10.017
Pusic I, DiPersio JF (2010) Update on clinical experience with AMD3100, an SDF-1/CXCL12-CXCR4 inhibitor, in mobilization of hematopoietic stem and progenitor cells. Curr Opin Hematol 17:319–326. doi:10.1097/MOH.0b013e328338b7d5
Resch T, Pircher A, Kahler C, Pratschke J, Hilbe W (2012) Endothelial progenitor cells: current issues on characterization and challenging clinical applications. Stem Cell Rev Rep 8:926–939. doi:10.1007/s12015-011-9332-9
Richardson MR, Yoder MC (2011) Endothelial progenitor cells: quo Vadis? J Mol Cell Cardiol 50:266–272. doi:10.1016/j.yjmcc.2010.07.009
Sen S, McDonald SP, Coates PT, Bonder CS (2011) Endothelial progenitor cells: novel biomarker and promising cell therapy for cardiovascular disease. Clin Sci (Lond) 120:263–283. doi:10.1042/CS20100429
Shepherd RM, Capoccia BJ, Devine SM, DiPersio J, Trinkaus KM, Ingram D, Link DC (2006) Angiogenic cells can be rapidly mobilized and efficiently harvested from the blood following treatment with AMD3100. Blood 108:3662–3667. doi:10.1182/blood-2006-06-030577
Timmermans F, Plum J, Yoder MC, Ingram DA, Vandekerckhove B, Case J (2009) Endothelial progenitor cells: identity defined? J Cell Mol Med 13:87–102. doi:10.1111/j.1582-4934.2008.00598.x
Toshner M et al (2009) Evidence of dysfunction of endothelial progenitors in pulmonary arterial hypertension. Am J Respir Crit Care Med 180:780–787. doi:10.1164/rccm.200810-1662OC
Tura O et al (2010) Granulocyte colony-stimulating factor (G-CSF) depresses angiogenesis in vivo and in vitro: implications for sourcing cells for vascular regeneration therapy. J Throm Haemost JTH 8:1614–1623. doi:10.1111/j.1538-7836.2010.03900.x
Xue S, Zhang HT, Zhang P, Luo J, Chen ZZ, Jang XD, Xu RX (2010) Functional endothelial progenitor cells derived from adipose tissue show beneficial effect on cell therapy of traumatic brain injury. Neurosci Lett 473:186–191. doi:10.1016/j.neulet.2010.02.035
Yoshioka T et al (2006) Granulocyte colony-stimulating factor (G-CSF) accelerates reendothelialization and reduces neointimal formation after vascular injury in mice. Cardiovasc Res 70:61–69. doi:10.1016/j.cardiores.2005.12.013
Yuan Y, Altalhi WA, Ng JJ, Courtman DW (2013) Derivation of human peripheral blood derived endothelial progenitor cells and the role of osteopontin surface modification and eNOS transfection. Biomaterials 34:7292–7301. doi:10.1016/j.biomaterials.2013.06.003
Acknowledgments
This study was supported by the National Natural Science Foundation of China (No. 31370984, No. 81301560) and the China Postdoctoral Science Foundation (No. 2012M521698).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fu, WL., Xiang, Z., Huang, FG. et al. Combination of granulocyte colony-stimulating factor and CXCR4 antagonist AMD3100 for effective harvest of endothelial progenitor cells from peripheral blood and in vitro formation of primitive endothelial networks. Cell Tissue Bank 17, 161–169 (2016). https://doi.org/10.1007/s10561-015-9527-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-015-9527-4