Abstract
Purpose
Epidemiological evidence on the risk factors for uterine/endometrial cancer in breast cancer (BCa) survivors is limited and inconsistent. Therefore, we critically reviewed and summarized available evidence related to the risk factors for uterine/endometrial cancer in BCa survivors.
Methods
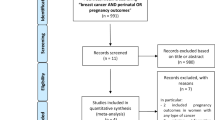
We conducted a literature search through PubMed, Web of Science Core Collection/Cited Reference Search, as well as through manual searches of the bibliographies of the articles identified in electronic searches. We included in this review studies that were published up to November 30, 2018 that were accessible in full-text format and were published in English.
Results
Of the 27 eligible studies, 96% had > 700 participants, 74% were prospective cohorts, 70% originated outside of the US, 44% reported as having pre-/postmenopausal women, and 26% reported having racially heterogeneous populations. Risk factors positively associated with uterine/endometrial cancer risk among BCa survivors included age at BCa diagnosis > 50 years, African American race, greater BMI/weight gain, and Tamoxifen treatment. For other lifestyle, reproductive and clinical factors, associations were either not significant (parity) or inconsistent (HRT use, menopausal status, smoking status) or had limited evidence (alcohol intake, family history of cancer, age at first birth, oral contraceptive use, age at menopause, comorbidities).
Conclusion
We identified several methodological concerns and limitations across epidemiological studies on potential risk factors for uterine/endometrial cancer in BCa survivors, including lack of details on uterine/endometrial cancer case ascertainment, varying and imprecise definitions of important covariates, insufficient adjustment for potential confounders, and small numbers of uterine/endometrial cancer cases in the overall as well as stratified analyses. Based on the available evidence, older age and higher body weight measures appear to be a shared risk factor for uterine/endometrial cancer in the general population as well as in BCa survivors. In addition, there is suggestive evidence that African American BCa survivors have a higher risk of uterine/endometrial cancer as compared to their White counterparts. There is also evidence that Tamoxifen contributes to uterine/endometrial cancer in BCa survivors. Given limitations of existing studies, more thorough investigation of these associations is warranted to identify additional preventive strategies needed for BCa survivors to reduce uterine/endometrial cancer risk and improve overall survival.





Similar content being viewed by others
Abbreviations
- ACS:
-
American Cancer Society
- BCa:
-
Breast cancer
- CI:
-
Confidence interval
- HR:
-
Hazard ratios
- OR:
-
Odds ratio
- RR:
-
Relative risk
- SEER:
-
Surveillance, epidemiology, and end results
- SIR:
-
Standardized incidence ratio
- US:
-
United States
References
American Cancer Society ACS (2018) Cancer Facts & Figures, Atlanta
Surveillance E, and End Results Program (2018) Female Breast Cancer—Cancer Stat Facts. https://seer.cancer.gov/statfacts/html/breast.html.
Ricceri F, Fasanelli F, Giraudo MT, Sieri S, Tumino R, Mattiello A, Vagliano L, Masala G, Quiros JR, Travier N, Sanchez MJ, Larranaga N, Chirlaque MD, Ardanaz E, Tjonneland A, Olsen A, Overvad K, Chang-Claude J, Kaaks R, Boeing H, Clavel-Chapelon F, Kvaskoff M, Dossus L, Trichopoulou A, Benetou V, Adarakis G, Bueno-de-Mesquita HB, Peeters PH, Sund M, Andersson A, Borgquist S, Butt S, Weiderpass E, Skeie G, Khaw KT, Travis RC, Rinaldi S, Romieu I, Gunter M, Kadi M, Riboli E, Vineis P, Sacerdote C (2015) Risk of second primary malignancies in women with breast cancer: results from the European prospective investigation into cancer and nutrition (EPIC). Int J Cancer 137(4):940–948
Jung HK, Park S, Kim NW, Lee JE, Kim Z, Han SW, Hur SM, Kim SY, Lim CW, Lee MH, Lee J (2017) Development of second primary cancer in Korean breast cancer survivors. Ann Surg Treat Res 93(6):287–292
Molina-Montes E, Pollan M, Payer T, Molina E, Davila-Arias C, Sanchez MJ (2013) Risk of second primary cancer among women with breast cancer: a population-based study in Granada (Spain). Gynecol Oncol 130(2):340–345
Corso G, Veronesi P, Santomauro GI, Maisonneuve P, Morigi C, Peruzzotti G, Intra M, Sacchini V, Galimberti V (2018) Multiple primary non-breast tumors in breast cancer survivors. J Cancer Res Clin Oncol 144(5):979–986
Nsouli-Maktabi HH, Henson DE, Younes N, Young HA, Cleary SD (2011) Second primary breast, endometrial, and ovarian cancers in Black and White breast cancer survivors over a 35-year time span: effect of age. Breast Cancer Res Treat 129(3):963–969
Casey MJ, Crotzer D (2019) Cancer, Endometrial. In: StatPearls (ed) StatPearls Publishing LLC, Treasure Island (FL)
Li Z, Wu Q, Song J, Zhang Y, Zhu S, Sun S (2018) Risk of second primary female genital malignancies in women with breast cancer: a SEER analysis. Horm Cancer 9(3):197–204
Ngo C, Brugier C, Plancher C, de la Rochefordiere A, Alran S, Feron JG, Malhaire C, Scholl S, Sastre X, Rouzier R, Fourchotte V (2014) Clinico-pathology and prognosis of endometrial cancer in patients previously treated for breast cancer, with or without tamoxifen: a comparative study in 363 patients. Eur J Surg Oncol 40(10):1237–1244
Molina-Montes E, Requena M, Sanchez-Cantalejo E, Fernandez MF, Arroyo-Morales M, Espin J, Arrebola JP, Sanchez MJ (2015) Risk of second cancers cancer after a first primary breast cancer: a systematic review and meta-analysis. Gynecol Oncol 136(1):158–171
Trentham-Dietz A, Newcomb PA, Nichols HB, Hampton JM (2007) Breast cancer risk factors and second primary malignancies among women with breast cancer. Breast Cancer Res Treat 105(2):195–207
Druesne-Pecollo N, Touvier M, Barrandon E, Chan DS, Norat T, Zelek L, Hercberg S, Latino-Martel P (2012) Excess body weight and second primary cancer risk after breast cancer: a systematic review and meta-analysis of prospective studies. Breast Cancer Res Treat 135(3):647–654
Segev Y, Rosen B, Lubinski J, Gronwald J, Lynch HT, Moller P, Kim-Sing C, Ghadirian P, Karlan B, Eng C, Gilchrist D, Neuhausen SL, Eisen A, Friedman E, Euhus D, Ping S, Narod SA (2015) Risk factors for endometrial cancer among women with a BRCA1 or BRCA2 mutation: a case control study. Fam Cancer 14(3):383–391
Lee YC, Milne RL, Lheureux S, Friedlander M, McLachlan SA, Martin KL, Bernardini MQ, Smith C, Picken S, Nesci S, Hopper JL, Phillips KA (2017) Risk of uterine cancer for BRCA1 and BRCA2 mutation carriers. Eur J Cancer 84:114–120
Bland AE, Calingaert B, Secord AA, Lee PS, Valea FA, Berchuck A, Soper JT, Havrilesky L (2009) Relationship between tamoxifen use and high risk endometrial cancer histologic types. Gynecol Oncol 112(1):150–154
Dong C, Chen L (2014) Second malignancies after breast cancer: the impact of adjuvant therapy. Mol Clin Oncol 2(3):331–336
Kirova YM, De Rycke Y, Gambotti L, Pierga JY, Asselain B, Fourquet A, Institut Curie Breast Cancer Study G (2008) Second malignancies after breast cancer: the impact of different treatment modalities. Br J Cancer 98(5):870–874
Torres D, Myers JA, Eshraghi LW, Riley EC, Soliman PT, Milam MR (2015) Risk factors for the development of uterine cancer in breast cancer survivors: an army of women study. Ann Surg Oncol 22(6):1974–1979
Liu J, Jiang W, Mao K, An Y, Su F, Kim BY, Liu Q, Jacobs LK (2015) Elevated risks of subsequent endometrial cancer development among breast cancer survivors with different hormone receptor status: a SEER analysis. Breast Cancer Res Treat 150(2):439–445
Raymond JS, Hogue CJR (2006) Multiple primary tumours in women following breast cancer, 1973–2000. Br J Cancer 94(11):1745–1750. https://doi.org/10.1038/sj.bjc.6603172
Partners SRaLRSbE (2019) DistillerSR forest plot generator. Evidence Partners, Canada
Chen JY, Kuo SJ, Liaw YP, Avital I, Stojadinovic A, Man YG, Mannion C, Wang J, Chou MC, Tsai HD, Chen ST, Hsiao YH (2014) Endometrial cancer incidence in breast cancer patients correlating with age and duration of tamoxifen use: a population based study. J Cancer 5(2):151–155
Chen MC, Lee KD, Lu CH, Wang TY, Huang SH, Chen CY (2018) The bidirectional association among female hormone-related cancers: breast, ovary, and uterine corpus. Cancer Med 7(6):2299–2306
Cortesi L, De Matteis E, Rashid I, Cirilli C, Proietto M, Rivasi F, Federico M (2009) Distribution of second primary malignancies suggests a bidirectional effect between breast and endometrial cancer: a population-based study. Int J Gynecol Cancer 19(8):1358–1363
Liu X, Zhao F, Hu L, Sun Y (2015) Value of detection of serum human epididymis secretory protein 4 and carbohydrate antigen 125 in diagnosis of early endometrial cancer of different pathological subtypes. Onco Targets Ther 8:1239–1243
Lee KD, Chen SC, Chan CH, Lu CH, Chen CC, Lin JT, Chen MF, Huang SH, Yeh CM, Chen MC (2008) Increased risk for second primary malignancies in women with breast cancer diagnosed at young age: a population-based study in Taiwan. Cancer Epidemiol Biomark Prev 17(10):2647–2655
Mellemkjaer L, Friis S, Olsen JH, Scelo G, Hemminki K, Tracey E, Andersen A, Brewster DH, Pukkala E, McBride ML, Kliewer EV, Tonita JM, Kee-Seng C, Pompe-Kirn V, Martos C, Jonasson JG, Boffetta P, Brennan P (2006) Risk of second cancer among women with breast cancer. Int J Cancer 118(9):2285–2292
Kamigaki Y, Kawakami K (2011) Risk of second cancer after initial treatment of breast cancer: an osaka cancer registry database study. Oncol Lett 2(5):963–973
Andersson M, Jensen MB, Engholm G, Henrik Storm H (2008) Risk of second primary cancer among patients with early operable breast cancer registered or randomised in Danish Breast Cancer Cooperative Group (DBCG) protocols of the 77, 82 and 89 programmes during 1977–2001. Acta Oncol 47(4):755–764
Schaapveld M, Visser O, Louwman MJ, de Vries EG, Willemse PH, Otter R, van der Graaf WT, Coebergh JW, van Leeuwen FE (2008) Risk of new primary nonbreast cancers after breast cancer treatment: a Dutch population—based study. J Clin Oncol 26(8):1239–1246
Swerdlow AJ, Jones ME (2005) Tamoxifen treatment for breast cancer and risk of endometrial cancer: a case–control study. J Natl Cancer Inst 97(5):375–384
Soerjomataram I, Louwman WJ, de Vries E, Lemmens VEPP, Klokman WJ, Coebergh JWW (2005) Primary malignancy after primary female breast cancer in the South of the Netherlands, 1972–2001. Breast Cancer Res Treat 93(1):91–95
Chen JY, Kuo SJ, Liaw YP, Avital I, Stojadinovic A, Man YG, Mannion C, Wang J, Chou MC, Tsai HD, Chen ST (2014) Endometrial cancer incidence in breast cancer patients correlating with age and duration of tamoxifen use: a population based study. J Cancer 5(2):e151
Bergman L, Beelen ML, Gallee MP, Hollema H, Benraadt J, van Leeuwen FE (2000) Risk and prognosis of endometrial cancer after tamoxifen for breast cancer comprehensive cancer centres' ALERT Group Assessment of liver and endometrial cancer risk following tamoxifen. Lancet 356(9233):881–887
Mignotte H, Lasset C, Bonadona V, Lesur A, Luporsi E, Rodier JF, Cutuli B, Lasry S, Mauriac L, Granon C, Kerr C, Giard S, Hill C, de Lafontan B, de Gislain C, D'Anjou J, Fondrinier E, Lefeuvre C, Parache RM, Chauvin F (1998) Iatrogenic risks of endometrial carcinoma after treatment for breast cancer in a large French case-control study federation nationale des centres de lutte contre le cancer (FNCLCC). Int J Cancer 76(3):325–330
Tanaka H, Tsukuma H, Koyama H, Kinoshita Y, Kinoshita N, Oshima A (2001) Second primary cancers following breast cancer in the Japanese female population. Jpn J Cancer Res 92(1):1–8
Chlebowski RT, Schottinger JE, Shi J, Chung J, Haque R (2015) Aromatase inhibitors, tamoxifen, and endometrial cancer in breast cancer survivors. Cancer 121(13):2147–2155
Hajian-Tilaki K (2011) Sample size estimation in epidemiologic studies. Caspian J Intern Med 2(4):289–298
Peduzzi P, Concato J, Kemper E, Holford TR, Feinstein AR (1996) A simulation study of the number of events per variable in logistic regression analysis. J Clin Epidemiol 49(12):1373–1379
Noordzij M, Tripepi G, Dekker FW, Zoccali C, Tanck MW, Jager KJ (2010) Sample size calculations: basic principles and common pitfalls. Nephrol Dial Transpl 25(5):1388–1393
Mellemkjaer L, Christensen J, Frederiksen K, Pukkala E, Weiderpass E, Bray F, Friis S, Andersson M, Olsen JH (2011) Risk of primary non-breast cancer after female breast cancer by age at diagnosis. Cancer Epidemiol Biomark Prev 20(8):1784–1792
Brown LM, Chen BE, Pfeiffer RM, Schairer C, Hall P, Storm H, Pukkala E et al (2007) Risk of second non-hematological malignancies among 376,825 breast cancer survivors. Breast Cancer Res Treat 106(3):439–451
Lee J, Park S, Kim S, Kim J, Ryu J, Park HS, Kim SI, Park BW (2015) Characteristics and survival of breast cancer patients with multiple synchronous or metachronous primary cancers. Yonsei Med J 56(5):1213–1220
Bray F, Ferlay J (2019) Chapter 5: data comparability and quality. World Health Organization, IARC. https://ci5.iarc.fr/CI5-XI/Pages/Chapter5.aspx.
Association ICR (2017) Map of cancer registries. https://www.registri-tumori.it/cms/pagine/mappa-dei-registri-di-popolazione.
Antoni S, Soerjomataram I, Møller B, Bray F, Ferlay J (2016) An assessment of GLOBOCAN methods for deriving national estimates of cancer incidence. Bull World Health Organ 94(3):174
Kao WH, Hong JH, See LC, Yu HP, Hsu JT, Chou IJ, Chou WC, Chiou MJ, Wang CC, Kuo CF (2018) Validity of cancer diagnosis in the National Health Insurance database compared with the linked National Cancer Registry in Taiwan. Pharmacoepidemiol Drug Saf 27(10):1060–1066
Parikh-Patel A, Allen M, Wright WE (2003) Validation of self-reported cancers in the California Teachers Study. Am J Epidemiol 157(6):539–545
Navarro C, Chirlaque MD, Tormo MJ, Pérez-Flores D, Rodríguez-Barranco M, Sánchez-Villegas A, Agudo A, Pera G, Amiano P, Dorronsoro M, Larrañaga N, Quirós JR, Ardanaz E, Barricarte A, Martínez C, Sánchez MJ, Berenguer A, González CA (2006) Validity of self reported diagnoses of cancer in a major Spanish prospective cohort study. J Epidemiol Community Health 60(7):593–599. https://doi.org/10.1136/jech.2005.039131
Oncology ASoC (2012) Uterine cancer—risk factors and prevention. https://www.cancer.net/cancer-types/uterine-cancer/risk-factors-and-prevention.
Society AC (2019) Endometrial cancer risk factors.
Marcheselli R, Marcheselli L, Cortesi L, Bari A, Cirilli C, Pozzi S, Ferri P, Napolitano M, Federico M, Sacchi S (2015) Risk of second primary malignancy in breast cancer survivors: a nested population-based case–control study. J Breast Cancer 18(4):378–385
Phipps AI, Ichikawa L, Bowles EJ, Carney PA, Kerlikowske K, Miglioretti DL, Buist DS (2010) Defining menopausal status in epidemiologic studies: a comparison of multiple approaches and their effects on breast cancer rates. Maturitas 67(1):60–66
Trentham-Dietz A, Sprague BL, Hampton JM, Miglioretti DL, Nelson HD, Titus LJ, Egan KM, Remington PL, Newcomb PA (2014) Modification of breast cancer risk according to age and menopausal status: a combined analysis of five population-based case-control studies. Breast Cancer Res Treat 145(1):165–175
Eden JA (2013) Menopausal status, adipose tissue, and breast cancer risk: impact of estrogen replacement therapy. Horm Mol Biol Clin Investig 14(2):57–63
Bonotto M, Puglisi F (2015) Early breast cancer in pre-menopausal women and endocrine treatment: which factors impact on decision-making process? Recent Prog Med 106(8):364–369
Sr J (2014) Menopausal hormone therapy and breast cancer. J Steroid Biochem Mol Biol 142:52–61
Ali AT (2013) Risk factors for endometrial cancer. Ceska Gynekol 78(5):448–459
Plagens-Rotman K, Żak E, Pięta B (2016) Odds ratio analysis in women with endometrial cancer. Prz Menopauzalny 15(1):12–19. https://doi.org/10.5114/pm.2016.58767
Setiawan VW, Pike MC, Kolonel LN, Nomura AM, Goodman MT, Henderson BE (2006) Racial/ethnic differences in endometrial cancer risk: the Multiethnic Cohort Study. Am J Epidemiol 165(3):262–270
Allard JE, Maxwell GL (2009) Race disparities between black and white women in the incidence, treatment, and prognosis of endometrial cancer. Cancer Control 16(1):53–56
Cote ML, Alhajj T, Ruterbusch JJ, Bernstein L, Brinton LA, Blot WJ, Chen C, Gass M, Gaussoin S, Henderson B, Lee E, Horn-Ross PL, Kolonel LN, Kaunitz A, Liang X, Nicholson WK, Park AB, Petruzella S, Rebbeck TR, Setiawan VW, Signorello LB, Simon MS, Weiss NS, Wentzensen N, Yang HP, Zeleniuch-Jacquotte A, Olson SH (2015) Risk factors for endometrial cancer in black and white women: a pooled analysis from the epidemiology of Endometrial Cancer Consortium (E2C2). Cancer Causes Control 26(2):287–296
Babatunde OA, Adams SA, Eberth JM, Wirth MD, Choi SK, Hebert JR (2016) Racial disparities in endometrial cancer mortality-to-incidence ratios among blacks and whites in South Carolina. Cancer Causes Control 27(4):503–511
Raglan O, Kalliala I, Markozannes G, Cividini S, Gunter MJ, Nautiyal J, Gabra H, Paraskevaidis E, Martin-Hirsch P, Tsilidis KK, Kyrgiou M (2018) Risk factors for endometrial cancer: an umbrella review of the literature. Int J Cancer 145(7):1719–1730
Aune D, Navarro Rosenblatt DA, Chan DS, Vingeliene S, Abar L, Vieira AR, Greenwood DC, Bandera EV, Norat T (2015) Anthropometric factors and endometrial cancer risk: a systematic review and dose-response meta-analysis of prospective studies. Ann Oncol 26(8):1635–1648
Arnold M, Pandeya N, Byrnes G, Renehan PAG, Stevens GA, Ezzati PM, Ferlay J, Miranda JJ, Romieu I, Dikshit R, Forman D, Soerjomataram I (2015) Global burden of cancer attributable to high body-mass index in 2012: a population-based study. Lancet Oncol 16(1):36–46
Nevadunsky NS, Van Arsdale A, Strickler HD, Moadel A, Kaur G, Levitt J, Girda E, Goldfinger M, Goldberg GL, Einstein MH (2014) Obesity and age at diagnosis of endometrial cancer. Obstet Gynecol 124(2 Pt 1):300–306
Birmann BM, Barnard ME, Bertrand KA, Bao Y, Crous-Bou M, Wolpin BM, De Vivo I, Tworoger SS (2016) Nurses’ health study contributions on the epidemiology of less common cancers: endometrial, ovarian, pancreatic, and hematologic. Am J Public Health 106(9):1608–1615. https://doi.org/10.2105/ajph.2016.303337
Jenabi E, Poorolajal J (2015) The effect of body mass index on endometrial cancer: a meta-analysis. Public Health 129(7):872–880
Zhou B, Yang L, Sun Q, Cong R, Gu H, Tang N, Zhu H, Wang B (2008) Cigarette smoking and the risk of endometrial cancer: a meta-analysis. Am J Med 121(6):501–508.e503
Fedirko V, Jenab M, Rinaldi S, Biessy C, Allen NE, Dossus L, Onland-Moret NC, Schutze M, Tjonneland A, Hansen L, Overvad K, Clavel-Chapelon F, Chabbert-Buffet N, Kaaks R, Lukanova A, Bergmann MM, Boeing H, Trichopoulou A, Oustoglou E, Barbitsioti A, Saieva C, Tagliabue G, Galasso R, Tumino R, Sacerdote C, Peeters PH, Bueno-de-Mesquita HB, Weiderpass E, Gram IT, Sanchez S, Duell EJ, Molina-Montes E, Arriola L, Chirlaque MD, Ardanaz E, Manjer J, Lundin E, Idahl A, Khaw KT, Romaguera-Bosch D, Wark PA, Norat T, Romieu I (2013) Alcohol drinking and endometrial cancer risk in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Ann Epidemiol 23(2):93–98
Zhou Q, Guo P, Li H, Chen XD (2017) Does alcohol consumption modify the risk of endometrial cancer? A dose-response meta-analysis of prospective studies. Arch Gynecol Obstet 295(2):467–479
Friberg E, Orsini N, Mantzoros CS, Wolk A (2010) Alcohol intake and endometrial cancer risk: a meta-analysis of prospective studies. Br J Cancer 103(1):127–131
Win AK, Reece JC, Ryan S (2015) Family history and risk of endometrial cancer: a systematic review and meta-analysis. Obstet Gynecol 125(1):89–98
Hackshaw A, Roughton M, Forsyth S, Monson K, Reczko K, Sainsbury R, Baum M (2011) Long-term benefits of 5 years of tamoxifen: 10-year follow-up of a large randomized trial in women at least 50 years of age with early breast cancer. J Clin Oncol 29(13):1657–1663
Gray RG, Rea D, Handley K, Bowden SJ, Perry P, Earl HM, Poole CJ, Bates T, Chetiyawardana S, Dewar JA, Fernando IN, Grieve R, Nicoll J, Rayter Z, Robinson A, Salman A, Yarnold J, Bathers S, Marshall A, Lee M, Group obotaC (2013) aTTom: long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years in 6,953 women with early breast cancer. J Clin Oncol 31:5–5
Acknowledgements
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
Not applicable.
Research involving human participants and/or animals
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wijayabahu, A.T., Egan, K.M. & Yaghjyan, L. Uterine cancer in breast cancer survivors: a systematic review. Breast Cancer Res Treat 180, 1–19 (2020). https://doi.org/10.1007/s10549-019-05516-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-019-05516-1