Abstract
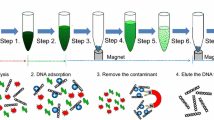
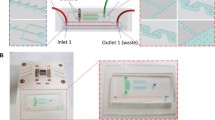
Nucleic acids serve as biomarkers of disease and it is highly desirable to develop approaches to extract small number of such genomic extracts from human bodily fluids. Magnetic particles-based nucleic acid extraction is widely used for concentration of small amount of samples and is followed by DNA amplification in specific assays. However, approaches to integrate such magnetic particles based capture with micro and nanofluidic based assays are still lacking. In this report, we demonstrate a magnetophoretic-based approach for target-specific DNA extraction and concentration within a microfluidic device. This device features a large chamber for reducing flow velocity and an array of μ-magnets for enhancing magnetic flux density. With this strategy, the device is able to collect up to 95 % of the magnetic particles from the fluidic flow and to concentrate these magnetic particles in a collection region. Then an enzymatic reaction is used to detach the DNA from the magnetic particles within the microfluidic device, making the DNA available for subsequent analysis. Concentrations of over 1000-fold for 90 bp dsDNA molecules is demonstrated. This strategy can bridge the gap between detection of low concentration analytes from clinical samples and a range of micro and nanofluidic sensors and devices including nanopores, nano-cantilevers, and nanowires.




Similar content being viewed by others
References
S. M. Azimi et al., A magnetic bead-based DNA extraction and purification microfluidic device. Microfluid. Nanofluid. 11, 157–165 (2011)
A. Cerf et al., Single DNA molecule patterning for high-throughput epigenetic mapping. Anal. Chem. 83, 8073–8077 (2011)
P. M. Das, R. Singal, DNA methylation and cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 22, 4632–4642 (2004)
T. P. Forbes, S. P. Forry, Microfluidic magnetophoretic separations of immunomagnetically labeled rare mammalian cells. Lab Chip 12, 1471–1479 (2012)
L. M. Gonzalez et al., Controlling Magnetotactic Bacteria through an Integrated Nanofabricated Metallic Island And Optical Microscope Approach. Sci Rep. 4, 4104 (2014)
S. Jahr et al., DNA fragments in the blood plasma of cancer patients: quantitations and evidence for their origin from apoptotic and necrotic cells. Cancer Res. 61, 1659–1665 (2001)
R. Kandimalla, A. A. van Tilborg, E. C. Zwarthoff, DNA methylation-based biomarkers in bladder cancer. Nat Rev Urol 10, 327–335 (2013)
K. Kiianitsa, N. Maizels, Ultrasensitive isolation, identification and quantification of DNA-protein adducts by ELISA-Based RADAR assay. Nucleic Acids Res. 42, e108 (2014)
J. B. Kisiel et al., Stool DNA testing for the detection of pancreatic cancer assessment of methylation marker candidates. Cancer 118, 2623–2631 (2012)
P. W. Laird, The power and the promise of DNA methylation markers. Nat. Rev. Cancer 3, 253–266 (2003)
R. Mariella Jr., Sample preparation: the weak link in microfluidics-based biodetection. Biomed. Microdevices 10, 777–784 (2008)
A. Murrell, V. K. Rakyan, S. Beck, From genome to epigenome. Hum. Mol. Genet. 14, R3–R10 (2005)
B. Nagy, Z. Ban, Z. Papp, The DNA isolation method has effect on allele drop-out and on the results of fluorescent PCR and DNA fragment analysis. Clin. Chim. Acta 360, 128–132 (2005)
A. Niemz, T. M. Ferguson, D. S. Boyle, Point-of-care nucleic acid testing for infectious diseases. Trends Biotechnol. 29, 240–250 (2011)
J. A. Osborn, Demagnetizing factors of the General Ellipsoid. Phys. Rev. 67, 351–357 (1945)
L. A. Sasso et al., Automated microfluidic processing platform for multiplexed magnetic bead immunoassays. Microfluid. Nanofluid. 13, 603–612 (2012)
J. C. Sheehan, G. L. Boshart, P. A. Cruickshank, Convenient Synthesis of Water-Soluble Carbodiimides. J Org Chem 26, 2525–2528 (1961)
J. Shim et al., Detection and quantification of methylation in DNA using solid-state nanopores. Scientific Reports 3, 1389 (2013)
J. Shim et al., Nanopore-based assay for detection of methylation in double-stranded DNA fragments. ACS Nano 9, 290–300 (2015)
M. M. Sirdah, Superparamagnetic-bead based method: An effective DNA Extraction from dried blood spots (DBS) for diagnostic PCR. Journal of clinical and diagnostic research: JCDR 8, FC01–FC04 (2014)
H. W. Wu et al., An integrated microfluidic system for isolation, counting, and sorting of hematopoietic stem cells. Biomicrofluidics 4(2), 024112 (2010)
H. Z. Zou et al., High detection rates of colorectal neoplasia by stool DNA Testing with a novel digital melt curve assay. Gastroenterology 136, 459–470 (2009)
Acknowledgments
The authors would like to acknowledge funding support from National Institute of Health (R21 CA155863), Oxford Nanopore Technologies U.K., and financial support from Mayo-Illinois Alliance for Technology Based Healthcare (http://mayoillinois.org/). Finally, authors would like to thank Dr. Gregory Damhorst for experimental advice on qPCR.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(PDF 615 kb)
Rights and permissions
About this article
Cite this article
Shim, S., Shim, J., Taylor, W.R. et al. Magnetophoretic-based microfluidic device for DNA Concentration. Biomed Microdevices 18, 28 (2016). https://doi.org/10.1007/s10544-016-0051-5
Published:
DOI: https://doi.org/10.1007/s10544-016-0051-5