Abstract
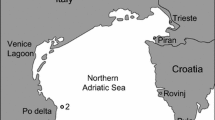
Long-term population data of marine invaders are rarely collected although it provides fundamental knowledge of the invasion dynamics that is important for evaluating the impacts, interactions and range expansion of the invader and for management purposes. During a 6-year monitoring period, we studied the dynamics and population demographic characteristics of the newly introduced mud crab Rhithropanopeus harrisii. The overall abundance of R. harrisii appeared to follow the boom and bust pattern with a rapid initial abundance increase and subsequent decline. The recruitment and growth of juveniles were correlated with the temperature during the larval period indicating that the increase in water temperature caused by climate change could have a positive effect on the recruitment and have potentially facilitated the establishment of R. harrisii in the northern Baltic Sea. The changes in the survival of reproductive females influence most the growth rate of the studied population. Hence, native predators feeding on benthic fauna such as fish could regulate the population growth of R. harrisii in the study area by reducing female survival. Although the population size seems to be stabilized at the monitoring locations, R. harrisii continues to expand its distribution range, and the rapid initial population increase is likely occurring at newly invaded sites.





Similar content being viewed by others
References
Angilletta MJ, Steury JTD, Sears M (2004) Temperature, growth rate, and body size in ectotherms: fitting pieces of a life-history puzzle. Integr Comp Biol 44:498–509
Bacevičius E, Gasiunaite ZR (2008) Two crab species-Chinese mitten crab (Eriocheir sinensis Edw.) and mud crab (Rhithropanopeus harrisii (Gould) ssp. tridentatus (Maitland) in the Lithuanian coastal waters, Baltic Sea. Transit Waters Bull 2:63–68. https://doi.org/10.1285/i1825229Xv2n2p63
Barnes RD (1980) Invertebrate zoology. Saunders College, Philadelphia
Bauer LJ, Miller TJ (2010) Temperature-, salinity-, and size-dependent winter mortality of juvenile blue crabs (Callinectes sapidus). Estuar Coasts 33:668–677. https://doi.org/10.1007/sl2237-010-9277-2
Blakeslee A (2015) Parasites and genetics in marine invertebrate introductions: signatures of diversity declines across systems. In: Canning-Clode J (ed) Biological invasion in changing ecosystems- vectors, ecological impacts, management and predictions. De Gruyter Open, Warsaw, pp 138–182
Bonsdorff E (2006) Zoobenthic diversity-gradients in the Baltic Sea: continuous post-glacial succession in a stressed ecosystem. J Exp Mar Bio Ecol 330:383–391. https://doi.org/10.1016/j.jembe.2005.12.041
Boyle T, Keith D, Pfau R (2010) Occurrence, reproduction, and population genetics of the estuarine Mud Crab, Rhithropanopeus harrisii (Gould) (Decapoda, Panopidae) in Texas freshwater reservoirs. Crustaceana 83:493–505. https://doi.org/10.1163/001121610X492148
Branch G (1975) Intraspecific competition in petella cochlear born. J Anim Ecol 44:263–281
Burnaford JL, Henderson SY, Pernet B (2011) Assemblage shift following population collapse of a non-indigenous bivalve in an urban lagoon. Mar Biol 158:1915–1927. https://doi.org/10.1007/s00227-011-1703-x
Canning-Clode J, Carlton JT (2017) Refining and expanding global climate change scenarios in the sea: poleward creep complexities, range termini, and setbacks and surges. Divers Distrib 23:463–473. https://doi.org/10.1111/ddi.12551
Canning-Clode J, Fowler AE, Byers JE et al (2011) “Caribbean creep” chills out: climate change and marine invasive species. PLoS One 6:2–6. https://doi.org/10.1371/journal.pone.0029657
Caswell H (2000) Matrix population models, 2nd edn. Sinauer Associates, Sunderland
Chamberlain N (1962) Ecological studies of the larval development of Rhithropanopeus harrisii (Xanthidae, Brachyura). Chesap Bay Inst Tech Rep 28:1–47
Coale A, Trussell J (1996) The development and use of demographic models. Popul Stud (NY) 50:469–484. https://doi.org/10.1080/0032472031000149576
Costlow JJ, Bookhout CG, Monroe RT (1966) Studies on the larval development of the crab, Rhithropanopeus harrisii (Gould). The effect of salinity and temperature on larval development. Physiol Zool 39:81–100
Cunningham SR, Darnell MZ (2015) Temperature-dependent growth and molting in early juvenile blue crabs Callinectes sapidus. J Shellfish Res 34:505–510. https://doi.org/10.2983/035.034.0246
Czerniejewski P (2009) Some aspects of population biology of the mud crab, Rhithropanopeus harrisii (Gould, 1841) in the Odra estuary, Poland. Oceanol Hydrobiol Stud. https://doi.org/10.2478/v10009-009-0043-3
D’Incao F, Martins ST (1998) Occurence of Rhithropanopeus harrisii (Gould 1841) in the southern coast of Brazil (Decapoda, Xanthidae). Nauplius Rio Gd 6:191–194
de Rivera C, Ruiz G, Hines A, Jivoff P (2005) Biotic resistance to invasion: native predator limits abundance and distribution of an introduced crab. Ecology 86:3364–3376
Donahue MJ (2004) Size-dependent competition in a gregarious porcelain crab Petrolisthes cinctipes (Anomura: Porcellanidae). Mar Ecol Prog Ser 267:219–231. https://doi.org/10.3354/meps267219
Dorgelo J (1976) Salt tolerance in Crustacea and the influence of temperature upon it. Biol Rev 51:255–290. https://doi.org/10.1111/j.1469-185X.1976.tb01127.x
Forsström T, Fowler AE, Manninen I, Vesakoski O (2015) An introduced species meets the local fauna: predatory behavior of the crab Rhithropanopeus harrisii in the Northern Baltic Sea. Biol Invasions. https://doi.org/10.1007/s10530-015-0909-0
Forward RB (2009) Larval biology of the crab Rhithropanopeus harrisii (Gould): A synthesis. Biol Bull 216:243–256. https://doi.org/10.2307/25548158
Fowler AE, Forsström T, von Numers M, Vesakoski O (2013) The North American mud crab Rhithropanopeus harrisii (Gould, 1841) in newly colonized Northern Baltic Sea: Distribution and ecology. Aquat Invasions. https://doi.org/10.3391/ai.2013.8.1.10
Gíslason OS, Jonasson JP, Palsson S et al (2017) Population density and growth of the newly introduced Atlantic rock crab Cancer irroratus Say, 1817 (Decapoda, Brachyura) in Iceland: a four-year mark-recapture study. Mar Biol Res 13:198–209. https://doi.org/10.1080/17451000.2016.1240875
Gissler C (1884) The Crab parasite, Sacculina. Am Nat 18:225–229
Gonçalves F, Ribeiro R, Soares AM V (1995) Laboratory study of effects of temperature and salinity on survival and larval development of a population of Rhithropanopeus harrisii from the Mondego River estuary, Portugal. Mar Biol 121:639–645
Grosholz E (2002) Ecological and evolutionary consequences of coastal invasions. Trends Ecol Evol 17:22–27
Grosholz ED, Ruiz GM (2003) Biological invasions drive size increases in marine and estuarine invertebrates. Ecol Lett 6:700–705. https://doi.org/10.1046/j.1461-0248.2003.00495.x
Hegele-Drywa J, Normant M (2014) Non-native crab Rhithropanopeus harrisii (Gould, 1984)—a new component of the benthic communities in the Gulf of Gdańsk (southern Baltic Sea). Oceanologia 56:125–139. https://doi.org/10.5697/oc.56-1.125
Hegele-Drywa J, Normant M, Szwarc B, Podłuska A (2014) Population structure, morphometry and individual condition of the non-native crab Rhithropanopeus harrisii (Gould, 1841), a recent coloniser of the Gulf of Gdańsk (southern Baltic Sea). Oceanologia 56:805–824. https://doi.org/10.5697/oc.56-4.805
HELCOM (2015) Balsam project-work package 4: testing monitoring methods for non-indigenous species in baltic ports
Holopainen R, Lehtiniemi M, Meier HEM et al (2016) Impacts of changing climate on the non-indigenous invertebrates in the northern Baltic Sea by end of the twenty-first century. Biol Invasions 18:3015–3032. https://doi.org/10.1007/s10530-016-1197-z
Hunt CE, Behrens Yamada S (2003) Biotic resistance experienced by an invasive crustacean in a temperate estuary. Biol Invasions 5:33–43. https://doi.org/10.1023/A:1024011226799
Iseda M, Otani M, Kimura T (2007) First record of an introduced Crab Rhithropanopeus harrisii (Crustacea: Brachyura: Panopeidae) in Japan. Jpn J Benthol 62:39–44
Jackson MC, Ruiz-Navarro A, Britton JR (2015) Population density modifies the ecological impacts of invasive species. Oikos 124:880–887. https://doi.org/10.1111/oik.01661
Jensen GC, Mcdonald PS, Armstrong DA (2007) Biotic resistance to green crab, Carcinus maenas, in California bays. Mar Biol 151:2231–2243. https://doi.org/10.1007/s00227-007-0658-4
Jormalainen V, Gagnon K, Sjöroos J, Rothäusler E (2016) The invasive mud crab enforces a major shift in a rocky littoral invertebrate community of the Baltic Sea. Biol Invasions 18:1409–1419. https://doi.org/10.1007/s10530-016-1090-9
Kelley AL, de Rivera CE, Grosholz ED et al (2015) Thermogeographic variation in body size of Carcinus maenas, the European green crab. Mar Biol 162:1625–1635. https://doi.org/10.1007/s00227-015-2698-5
Kornis MS, Carlson J, Lehrer-Brey G, Vander Zanden MJ (2014) Experimental evidence that ecological effects of an invasive fish are reduced at high densities. Oecologia 175:325–334. https://doi.org/10.1007/s00442-014-2899-5
Kotta J, Ojaveer H (2012) Rapid establishment of the alien crab Rhithropanopeus harrisii (Gould) in the Gulf of Riga. Est J Ecol 61:293–298. https://doi.org/10.3176/eco.2012.4.04
Kottelat M, Freyhof J (2007) Handbook of European freshwater fishes. Cornol and Freyhof, Switzerland and Berlin
Laughlin RB, French W (1989a) Differences in responses to factorial combinations of temperature and salinity by zoeae from two geographically isolated populations of the mud crab Rhithropanopeus harrisii. Mar Biol 102:387–395
Laughlin RB, French W (1989b) Interactions between temperature and salinity during brooding on subsequent zoeal development of the mud crab Rhithropanopeus harrisii. Mar Biol 102:377–386
Lehtiniemi M, Ojaveer H, David M et al (2015) Dose of truth-Monitoring marine non-indigenous species to serve legislative requirements. Mar Policy 54:26–35. https://doi.org/10.1016/j.marpol.2014.12.015
Leppäkoski E, Helminen H, Hänninen J, Tallqvist M (1999) Aquatic biodiversity under anthropogenic stress: an insight from the Archipelago Sea (SW Finland). Biodivers Conserv 8:55–70. https://doi.org/10.1023/A:1008805007339
Leppäranta M, Myrberg K (2009) Physical oceanography of the Baltic Sea. Springer, Berlin
Mäkinen K, Vuorinen I, Hänninen J (2017) Climate-induced hydrography change favours small-bodied zooplankton in a coastal ecosystem. Hydrobiologia 792:83–96. https://doi.org/10.1007/s10750-016-3046-6
Meier HEM (2006) Baltic Sea climate in the twenty-first century: a dynamical downscaling approach using two global models and two emission scenarios. Clim Dyn 27:39–68
Mizzan L, Zanella L (1996) First record of Rhithropanopeus harrisii (Gould, 1841) (Crustacea, Decapoda, Xanthidae) in the Italian waters. Bolletino del Mus Civ di Stor Nat di Venezia 46:109–122
Moksnes PO (2004) Interference competition for space in nursery habitats: density-dependent effects on growth and dispersal in juvenile shore crabs Carcinus maenas. Mar Ecol Prog Ser 281:181–191. https://doi.org/10.3354/meps281181
O’Connor NJ (2014) Invasion dynamics on a temperate rocky shore: from early invasion to establishment of a marine invader. Biol Invasions 16:73–87. https://doi.org/10.1007/s10530-013-0504-1
Parker JD, Torchin ME, Hufbauer RA et al (2013) Do invasive species perform better in their new ranges? Ecology 94:985–994
Petersen C (2006) Range expansion in the northeast Pacific by an estuary mud crab—a molecular study. Biol Invasions 8:565–576. https://doi.org/10.1007/s10530-005-0160-1
Puntila R (2016) Trophic interactions and impacts of non-indigenous species in Baltic Sea coastal ecosystems. University of Helsinki, Helsinki
Ricciardi A (2003) Predicting the impacts of an introduced species from its invasion history: an empirical approach applied to zebra mussel invasions. Freshw Biol 48:972–981. https://doi.org/10.1046/j.1365-2427.2003.01071.x
Roche DR, Torchin ME (2007) Established population of the North American Harris mud crab, Rhithropanopeus harrisii (Gould 1841) (Crustacea: Brachyura: Xanthidae) in the Panama Canal. Aquat Invasions 2:155–161. https://doi.org/10.3391/ai.2007.2.3.1
Roche DG, Torchin ME, Leung B, Binning SA (2009) Localized invasion of the North American Harris mud crab, Rhithropanopeus harrisii, in the Panama Canal: implications for eradication and spread. Biol Invasions 11:983–993. https://doi.org/10.1007/s10530-008-9310-6
Rodrigues MA, Incao FD (2015) Abundância e relações biométricas do caranguejo invasor Rhithropanopeus harrisii (Crustacea, Decapoda) no estuário da Lagoa dos Patos, Rio Grande do Sul, Brasil. Bol do Mus Biol Mello Leitão 37:219–232
Rodríguez G, Suárez H (2001) Anthropogenic dispersal of decapod crustaceans in aquatic environments. Interciencia 26:282–288
Rome MS, Young-Williams AC, Davis GR, Hines AH (2005) Linking temperature and salinity tolerance to winter mortality of Chesapeake Bay blue crabs (Callinectes sapidus). J Exp Mar Bio Ecol 319:129–145. https://doi.org/10.1016/j.jembe.2004.06.014
Ryan EP, Naturalist AM, Jul N (1956) Observations on the life histories and the distribution of the Xanthidae (Mud Crabs) of Chesapeake Bay. Am Midl Nat 56:138–162
Ryer CH, Ottmar M, Spencer M et al (2016) Temperature-dependent growth of early juvenile southern tanner crab Chionoecetes bairdi: implications for cold pool effects and climate change in the Southeastern Bering Sea. J Shellfish Res 35:259–267. https://doi.org/10.2983/035.035.0128
Sargent LW, Lodge DM (2014) Evolution of invasive traits in nonindigenous species: increased survival and faster growth in invasive populations of rusty crayfish (Orconectes rusticus). Evol Appl 7:949–961. https://doi.org/10.1111/eva.12198
Simberloff D, Gibbons L (2004) Now you see them, now you don’t!—population crashes of established introduced species. Biol Invasions 6:161–172. https://doi.org/10.1023/B:BINV.0000022133.49752.46
Stoner AW, Copeman LA, Ottmar ML (2013) Molting, growth, and energetics of newly-settled blue king crab: effects of temperature and comparisons with red king crab. J Exp Mar Bio Ecol 442:10–21. https://doi.org/10.1016/j.jembe.2013.02.002
Strayer DL, Malcom HM (2006) Long-term demography of a zebra mussel (Dreissena polymorpha) population. Freshw Biol 51:117–130. https://doi.org/10.1111/j.1365-2427.2005.01482.x
Strayer DL, Eviner VT, Jeschke JM, Pace ML (2006) Understanding the long-term effects of species invasions. Trends Ecol Evol 21:645–651. https://doi.org/10.1016/j.tree.2006.07.007
Stubben C, Milligan B (2007) Estimating and analyzing demographic models using popbio package in R. J Stat Softw 22:1–23. https://doi.org/10.18637/jss.v022.i11
Torchin ME, Lafferty KD, Kuris AM (2001) Release from parasites as natural enemies: increased performance of a globally introduced marine crab. Biol Invasions 3:333–345. https://doi.org/10.1023/A:1015855019360
Torchin ME, Lafferty KD, Dobson AP et al (2003) Introduced species and their missing parasites. Nature 421:628–630. https://doi.org/10.1038/nature01346
Turoboyski K (1973) Biology and ecology of the crab Rhithropanopeus harrisii ssp. tridentatus. Mar Biol 23:303–313. https://doi.org/10.1007/BF00389338
Van Engel W, Dillon W, Zwernet D, Eldridge D (1966) Loxothylacus panopaei (Cirripedia, Sacculinidae) an introduced parasite on a xanthid crab in Chesapeake Bay, USA. Crustaceana 10:110–112
Virnstein RW (1977) The importance of predation by crabs and fishes on benthic infauna in Chesapeake Bay. Ecology 58:1199–1217
Williams A (1984) Shrimps, lobsters, and crabs of the Atlantic coast of the eastern United States, Maine to Florida. Smithsonian Institution Press, Washington
Yuan Q, Wang Q, Zhang T et al (2017) Effects of water temperature on growth, feeding and molting of juvenile Chinese mitten crab Eriocheir sinensis. Aquaculture 468:169–174. https://doi.org/10.1016/j.aquaculture.2016.10.007
Acknowledgements
This study was financed by the Finnish Foundation for Nature Conservation from Rafael Kuuskoski memorial fund. A Research Coordination Network Grant funded by the Global Invasions Network, with support from the National Science Foundation, was awarded to AEF for this research. We would like to thank Juho Yli-Rosti and Salla Mikkola that have helped to collect the population data. We would also like to thank Veijo Jormalainen, Maiju Lehtiniemi, Jonne Kotta and three anonymous reviewers for the comments that greatly improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Forsström, T., Vesakoski, O., Riipinen, K. et al. Post-invasion demography and persistence of a novel functional species in an estuarine system. Biol Invasions 20, 3331–3345 (2018). https://doi.org/10.1007/s10530-018-1777-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-018-1777-1