Abstract
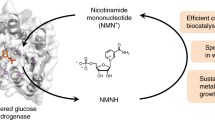
Nicotinamide mononucleotide (NMN) or Nicotinamide-1-ium-1-β-D-ribofuranoside 5′-phosphate is a nucleotide that can be converted into nicotinamide adenine dinucleotide (NAD) in human cells. NMN has recently attracted great attention because of its potential as an anti-aging drug, leading to great efforts for its effective manufacture. The chemical synthesis of NMN is a challenging task since it is an isomeric compound with a complicated structure. The majority of biological synthetic routes for NMN is through the intermediate phosphoribosyl diphosphate (PRPP), which is further converted to NMN by nicotinamide phosphoribosyltransferase (Nampt). There are various routes for the synthesis of PRPP from simple starting materials such as ribose, adenosine, and xylose, but all of these require the expensive phosphate donor adenosine triphosphate (ATP). Thus, an ATP regeneration system can be included, leading to diminished ATP consumption during the catalytic process. The regulations of enzymes that are not directly involved in the synthesis of NMN are also critical for the production of NMN. The aim of this review is to present an overview of the biological production of NMN with respect to the critical enzymes, reaction conditions, and productivity.






Similar content being viewed by others
References
Anderson RM, Bitterman KJ, Wood JG, Medvedik O, Sinclair DA (2003) Nicotinamide and PNC1 govern lifespan extension by calorie restriction in. Saccharomyces cerevisiae Nature 423:181–185. doi:https://doi.org/10.1038/nature01578
Andersson CE, Mowbray SL (2002) Activation of ribokinase by monovalent cations. J Mol Biol 315:409–419. doi:https://doi.org/10.1006/jmbi.2001.5248
Andexer JN, Richter M (2015) Emerging enzymes for ATP regeneration in. biocatalytic processes Chembiochem 16:380–386. doi:https://doi.org/10.1002/cbic.201402550
Arnvig K, Hove-Jensen B, Switzer RL (1990) Purification and properties of phosphoribosyl-diphosphate synthetase from Bacillus subtilis Eur J Biochem 192:195–200 doi:https://doi.org/10.1111/j.1432-1033.1990.tb19214.x
Bashor C, Denu JM, Brennan RG, Ullman B (2002) Kinetic mechanism of adenine phosphoribosyltransferase from. Leishmania donovani Biochemistry 41:4020–4031. doi:https://doi.org/10.1021/bi0158730
Becker MA, Smith PR, Taylor W, Mustafi R, Switzer RL (1995) The genetic and functional basis of purine nucleotide feedback-resistant phosphoribosylpyrophosphate synthetase superactivity. J Clin Invest 96:2133–2141. doi:https://doi.org/10.1172/JCI118267
Burgos ES, Ho MC, Almo SC, Schramm VL (2009) A phosphoenzyme mimic, overlapping catalytic sites and reaction coordinate motion for human NAMPT. Proc Natl Acad Sci U S A 106:13748–13753. doi:https://doi.org/10.1073/pnas.0903898106
Burgos ES, Schramm VL (2008) Weak coupling of ATP hydrolysis to the chemical equilibrium of human. nicotinamide phosphoribosyltransferase Biochemistry 47:11086–11096. doi:https://doi.org/10.1021/bi801198m
Cao H, Li C, Zhao J, Wang F, Tan T, Liu L (2018) Enzymatic production of glutathione coupling with an ATP regeneration system based on polyphosphate kinase. Appl Biochem Biotechnol 185:385–395. https://doi.org/10.1007/s12010-017-2664-4
Chini CCS, Tarrago MG, Chini EN (2017) NAD and the aging process: role in life, death and everything in between. Mol Cell Endocrinol 455:62–74. doi:https://doi.org/10.1016/j.mce.2016.11.003
Chuvikovsky DV et al (2006) Ribokinase from E. coli: expression, purification, and substrate specificity. Bioorg Med Chem 14:6327–6332. doi:https://doi.org/10.1016/j.bmc.2006.05.057
Curtin NJ, Szabo C (2020) Poly(ADP-ribose) polymerase inhibition: past, present and future. Nat Rev Drug Discov 19:711–736. doi:https://doi.org/10.1038/s41573-020-0076-6
Eriksen TA, Kadziola A, Bentsen AK, Harlow KW, Larsen S (2000) Structural basis for the function of Bacillus subtilis phosphoribosyl-pyrophosphate synthetase. Nat Struct Biol 7:303–308. doi:https://doi.org/10.1038/74069
Fu R, Zhang Q (2018) Nicotinamide phosphoribosyltransferase (NAMPT) mutant and use thereof US patent 20180230443A1
Gallo CM, Smith DL Jr, Smith JS (2004) Nicotinamide clearance by Pnc1 directly regulates Sir2-mediated silencing and longevity. Mol Cell Biol 24:1301–1312. doi:https://doi.org/10.1128/mcb.24.3.1301-1312.2004
Grozio A et al (2019) Slc12a8 is a nicotinamide mononucleotide transporter. Nat Metab 1:47–57. doi:https://doi.org/10.1038/s42255-018-0009-4
Hove-Jensen B, Andersen KR, Kilstrup M, Martinussen J, Switzer RL, Willemoes M (2017) Phosphoribosyl diphosphate (PRPP): biosynthesis, enzymology, utilization, and metabolic significance microbiol Mol Biol Rev 81 doi:https://doi.org/10.1128/MMBR.00040-16
Huyet J et al (2018) Structural insights into the forward and reverse enzymatic reactions in human adenine. phosphoribosyltransferase Cell Chem Biol 25:666–676 e664. doi:https://doi.org/10.1016/j.chembiol.2018.02.011
Irie J et al (2020) Effect of oral administration of nicotinamide mononucleotide on clinical parameters and nicotinamide metabolite levels in healthy Japanese. men Endocr J 67:153–160. doi:https://doi.org/10.1507/endocrj.EJ19-0313
Kadziola A, Jepsen CH, Johansson E, McGuire J, Larsen S, Hove-Jensen B (2005) Novel class III phosphoribosyl diphosphate synthase: structure and properties of the tetrameric, phosphate-activated, non-allosterically inhibited enzyme from Methanocaldococcus jannaschii J Mol Biol 354:815–828 doi:https://doi.org/10.1016/j.jmb.2005.10.001
Krath BN, Eriksen TA, Poulsen TS, Hove-Jensen B (1999) Cloning and sequencing of cDNAs specifying a novel class of phosphoribosyl diphosphate synthase in Arabidopsis thaliana. Biochim Biophys Acta 1430:403–408. doi:https://doi.org/10.1016/s0167-4838(99)00022-9
Krath BN, Hove-Jensen B (2001) Class II recombinant phosphoribosyl diphosphate synthase from spinach. Phosphate independence and diphosphoryl donor specificity. J Biol Chem 276:17851–17856. doi:https://doi.org/10.1074/jbc.M010172200
Lee J, Churchil H, Choi W, Lynch JE, Roberts FE, Volante RP, Paul JR (1999) A chemical synthesis of nicotinamide adenine dinucleotide (NAD+) Chem Commun:729–730 doi:https://doi.org/10.1039/A809930H
Li S, Lu Y, Peng B, Ding J (2007) Crystal structure of human phosphoribosylpyrophosphate synthetase 1 reveals a novel allosteric site. Biochem J 401:39–47. doi:https://doi.org/10.1042/BJ20061066
Liu L et al (2018) Quantitative analysis of NAD synthesis-breakdown lfuxes. Cell Metab 27:1067–1080
Marinescu GC, Popescu RG, Stoian G, Dinischiotu A (2018) beta-nicotinamide mononucleotide (NMN) production in Escherichia coli Sci Rep 8:12278 doi:https://doi.org/10.1038/s41598-018-30792-0
Motomura K, Hirota R, Okada M, Ikeda T, Ishida T, Kuroda A (2014) A new subfamily of polyphosphate kinase 2 (class III PPK2) catalyzes both nucleoside monophosphate phosphorylation and nucleoside diphosphate phosphorylation. Appl Environ Microbiol 80:2602–2608. doi:https://doi.org/10.1128/AEM.03971-13
Nosal JM, Switzer RL, Becker MA (1993) Overexpression, purification, and characterization of recombinant human 5-phosphoribosyl-1-pyrophosphate synthetase isozymes I and II. J Biol Chem 268:10168–10175
Panel M, Ghaleh B, Morin D (2018) Mitochondria and aging: a role for the mitochondrial transition pore? Aging Cell 17:e12793. doi:https://doi.org/10.1111/acel.12793
Park J, Gupta RS (2008) Adenosine kinase and ribokinase–the RK family of proteins. Cell Mol Life Sci 65:2875–2896. doi:https://doi.org/10.1007/s00018-008-8123-1
Revollo JR, Grimm AA, Imai S (2007) The regulation of nicotinamide adenine dinucleotide biosynthesis by Nampt/PBEF/visfatin in mammals. Curr Opin Gastroenterol 23:164–170. doi:https://doi.org/10.1097/MOG.0b013e32801b3c8f
Roessler BJ, Nosal JM, Smith PR, Heidler SA, Palella TD, Switzer RL, Becker MA (1993) Human X-linked phosphoribosylpyrophosphate synthetase superactivity is associated with distinct point mutations in the PRPS1 gene. J Biol Chem 268:26476–26481
Sarver AE, Wang CC (2002) The adenine phosphoribosyltransferase from Giardia lamblia has a unique reaction mechanism and unusual substrate binding properties. J Biol Chem 277:39973–39980. doi:https://doi.org/10.1074/jbc.M205595200
Selles Vidal L, Kelly CL, Mordaka PM, Heap JT (2018) Review of NAD(P)H-dependent oxidoreductases: properties, engineering and application Biochim. Biophys Acta Proteins Proteom 1866:327–347. doi:https://doi.org/10.1016/j.bbapap.2017.11.005
Shoji S, Yamaji T, Makino H, Ishii J, Kondo A (2020) Metabolic design for selective production of nicotinamide mononucleotide from glucose and nicotinamide. Metab Eng. https://doi.org/10.1016/j.ymben.2020.11.008
Sinclair DA, Ear PH (2015) Biological production of nad precursors and analogs WO Patent 2015069860A1
Takanori A (2018) Method for Producing Nicotinamide Mononucleotide and Transformant Used in Said Method WO Patent 2019065876A1
Todisco S, Agrimi G, Castegna A, Palmieri F (2006) Identification of the mitochondrial NAD + transporter in Saccharomyces cerevisiae. J Biol Chem 281:1524–1531. doi:https://doi.org/10.1074/jbc.M510425200
Wang T, Zhang X, Bheda P, Revollo JR, Imai S, Wolberger C (2006) Structure of Nampt/PBEF/visfatin, a mammalian NAD + biosynthetic enzyme. Nat Struct Mol Biol 13:661–662. doi:https://doi.org/10.1038/nsmb1114
Willemoes M, Hove-Jensen B (1997) Binding of divalent magnesium by Escherichia coli phosphoribosyl diphosphate synthetase. Biochemistry 36:5078–5083. https://doi.org/10.1021/bi962610a
Yoshino J, Baur JA, Imai SI (2018) NAD(+) Intermediates: The Biology and Therapeutic Potential of NMN and NR Cell Metab 27:513–528 doi:https://doi.org/10.1016/j.cmet.2017.11.002
Zakataeva NP, Romanenkov DV, Skripnikova VS, Vitushkina MV, Livshits VA, Kivero AD, Novikova AE (2012) Wild-type and feedback-resistant phosphoribosyl pyrophosphate synthetases from Bacillus amyloliquefaciens: purification, characterization, and application to increase purine nucleoside production. Appl Microbiol Biotechnol 93:2023–2033. doi:https://doi.org/10.1007/s00253-011-3687-3
Zhang X, Wu H, Huang B, Li Z, Ye Q (2017) One-pot synthesis of glutathione by a two-enzyme cascade using a thermophilic ATP regeneration system. J Biotechnol 241:163–169. doi:https://doi.org/10.1016/j.jbiotec.2016.11.034
Funding
This work was supported by the Fundamental Research Funds for the Provincial Universities of Zhejiang (RF-C2020002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shen, Q., Zhang, SJ., Xue, YZ. et al. Biological synthesis of nicotinamide mononucleotide. Biotechnol Lett 43, 2199–2208 (2021). https://doi.org/10.1007/s10529-021-03191-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-021-03191-1