Abstract
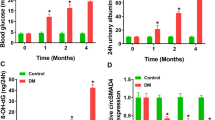
Diabetic nephropathy (DN) threatens the survival quality of patients, with complex pathogenesis. Circular RNA (circRNA) dysregulation occurs in DN development. This work aimed to investigate the role of circ-Luc7l in DN cell models and related molecular mechanisms. The expression of circ-Luc7l, microRNA (miR)-205-5p, and transforming growth factor-beta receptor 1 (Tgfbr1) was examined by real-time quantitative PCR (RT-qPCR). Cell viability and proliferation were detected by Cell Counting Kit-8 (CCK-8) assay and EdU assay. The expression of extracellular matrix (ECM)-related markers and Tgrbr1 protein was measured by Western blot. The binding between miR-205-5p and circ-Luc7l or Tgfbr1 was validated by dual-luciferase reporter assay, RNA immunoprecipitation (RIP) assay, or RNA pull-down assay. Experimental animal models were established to elucidate the function of circ-Luc7l in vivo. Circ-Luc7l expression was notably enhanced in high glucose (HG)-treated mesangial cells. Knockdown of circ-Luc7l attenuated HG-induced cell proliferation, inflammation, and ECM accumulation in vitro and relieved inflammation and ECM accumulation of kidneys of diabetic mice in vivo. Circ-Luc7l targeted miR-205-5p, and miR-205-5p inhibition rescued the depletion effects of circ-Luc7l knockdown on cell proliferation, inflammation, and ECM accumulation. MiR-205-5p bound to Tgfbr1 whose expression was negatively regulated by circ-Luc7l. Tgfbr1 overexpression also rescued the depletion effects of circ-Luc7l knockdown on cell proliferation, inflammation, and ECM accumulation. In HG conditions, increased circ-Luc7l upregulated Tgfbr1 expression via targeting miR-205-5p to induce DN progression.








Similar content being viewed by others
Data Availability
Not applicable.
References
Anders HJ, Huber TB, Isermann B, Schiffer M (2018) CKD in diabetes: diabetic kidney disease versus nondiabetic kidney disease. Nat Rev Nephrol 14:361–377. https://doi.org/10.1038/s41581-018-0001-y
Badal SS, Danesh FR (2014) New insights into molecular mechanisms of diabetic kidney disease. Am J Kidney Dis 63:S63–S83. https://doi.org/10.1053/j.ajkd.2013.10.047
Bolha L, Ravnik-Glavac M, Glavac D (2017) Circular RNAs: biogenesis, function, and a role as possible cancer biomarkers. Int J Genomics 2017:6218353. https://doi.org/10.1155/2017/6218353
Chen B, Li Y, Liu Y, Xu Z (2019) circLRP6 regulates high glucose-induced proliferation, oxidative stress, ECM accumulation, and inflammation in mesangial cells. J Cell Physiol 234:21249–21259. https://doi.org/10.1002/jcp.28730
Dounousi E, Duni A, Leivaditis K, Vaios V, Eleftheriadis T, Liakopoulos V (2015) Improvements in the management of diabetic nephropathy. Rev Diabet Stud 12:119–133. https://doi.org/10.1900/RDS.2015.12.119
Du L, Wang X, Yin Y, Zhang Y, Jia J, Lu B, Xue W, Qu C, Qi J (2021) Identification of a potentially functional circRNA-miRNA-mRNA ceRNA regulatory network in bladder cancer by analysis of microarray data. Transl Androl Urol 10:24–36. https://doi.org/10.21037/tau-20-660
Ge X, Xi L, Wang Q, Li H, Xia L, Cang Z, Peng W, Huang S (2020) Circular RNA Circ_0000064 promotes the proliferation and fibrosis of mesangial cells via miR-143 in diabetic nephropathy. Gene 758:144952. https://doi.org/10.1016/j.gene.2020.144952
Hu W, Han Q, Zhao L, Wang L (2019) Circular RNA circRNA_15698 aggravates the extracellular matrix of diabetic nephropathy mesangial cells via miR-185/TGF-beta1. J Cell Physiol 234:1469–1476. https://doi.org/10.1002/jcp.26959
Kulcheski FR, Christoff AP, Margis R (2016) Circular RNAs are miRNA sponges and can be used as a new class of biomarker. J Biotechnol 238:42–51. https://doi.org/10.1016/j.jbiotec.2016.09.011
Liu J, Wang J, Fu W, Wang X, Chen H, Wu X, Lao G, Wu Y, Hu M, Yang C, Yan L, Ren M (2021a) MiR-195-5p and miR-205-5p in extracellular vesicles isolated from diabetic foot ulcer wound fluid decrease angiogenesis by inhibiting VEGFA expression. Aging (Albany NY) 13:19805–19821. https://doi.org/10.18632/aging.203393
Liu Z, Wu X, Yu Z, Tang X (2021b) Reconstruction of circRNA-miRNA-mRNA associated ceRNA networks reveal functional circRNAs in intracerebral hemorrhage. Sci Rep 11:11584. https://doi.org/10.1038/s41598-021-91059-9
Marrero MB, Banes-Berceli AK, Stern DM, Eaton DC (2006) Role of the JAK/STAT signaling pathway in diabetic nephropathy. Am J Physiol Renal Physiol 290:F762–F768. https://doi.org/10.1152/ajprenal.00181.2005
Min D, Lyons JG, Bonner J, Twigg SM, Yue DK, McLennan SV (2009) Mesangial cell-derived factors alter monocyte activation and function through inflammatory pathways: possible pathogenic role in diabetic nephropathy. Am J Physiol Renal Physiol 297:F1229–F1237. https://doi.org/10.1152/ajprenal.00074.2009
Mou X, Chenv JW, Zhou DY, Liu K, Chen LJ, Zhou D, Hu YB (2020) A novel identified circular RNA, circ_0000491, aggravates the extracellular matrix of diabetic nephropathy glomerular mesangial cells through suppressing miR101b by targeting TGFbetaRI. Mol Med Rep 22:3785–3794. https://doi.org/10.3892/mmr.2020.11486
Ng WL, Mohd Mohidin TB, Shukla K (2018) Functional role of circular RNAs in cancer development and progression. RNA Biol 15:995–1005. https://doi.org/10.1080/15476286.2018.1486659
Rayego-Mateos S, Morgado-Pascual JL, Opazo-Rios L, Guerrero-Hue M, Garcia-Caballero C, Vazquez-Carballo C, Mas S, Sanz AB, Herencia C, Mezzano S, Gomez-Guerrero C, Moreno JA, Egido J (2020) Pathogenic pathways and therapeutic approaches targeting inflammation in diabetic nephropathy. Int J Mol Sci. https://doi.org/10.3390/ijms21113798
Szostek-Mioduchowska AZ, Lukasik K, Skarzynski DJ, Okuda K (2019) Effect of transforming growth factor-beta1 on alpha-smooth muscle actin and collagen expression in equine endometrial fibroblasts. Theriogenology 124:9–17. https://doi.org/10.1016/j.theriogenology.2018.10.005
Tan A, Li T, Ruan L, Yang J, Luo Y, Li L, Wu X (2021) Knockdown of Malat1 alleviates high-glucose-induced angiogenesis through regulating miR-205-5p/VEGF-A axis. Exp Eye Res 207:108585. https://doi.org/10.1016/j.exer.2021.108585
Tang B, Li W, Ji TT, Li XY, Qu X, Feng L, Bai S (2020) Circ-AKT3 inhibits the accumulation of extracellular matrix of mesangial cells in diabetic nephropathy via modulating miR-296-3p/E-cadherin signals. J Cell Mol Med 24:8779–8788. https://doi.org/10.1111/jcmm.15513
Tay Y, Rinn J, Pandolfi PP (2014) The multilayered complexity of ceRNA crosstalk and competition. Nature 505:344–352. https://doi.org/10.1038/nature12986
Toth-Manikowski S, Atta MG (2015) Diabetic kidney disease: pathophysiology and therapeutic targets. J Diabetes Res 2015:697010. https://doi.org/10.1155/2015/697010
Tung CW, Hsu YC, Shih YH, Chang PJ, Lin CL (2018) Glomerular mesangial cell and podocyte injuries in diabetic nephropathy. Nephrology (Carlton) 23(Suppl 4):32–37. https://doi.org/10.1111/nep.13451
Wu W, Ji P, Zhao F (2020) CircAtlas: an integrated resource of one million highly accurate circular RNAs from 1070 vertebrate transcriptomes. Genome Biol 21:101. https://doi.org/10.1186/s13059-020-02018-y
Xu B, Wang Q, Li W, Xia L, Ge X, Shen L, Cang Z, Peng W, Shao K, Huang S (2020) Circular RNA circEIF4G2 aggravates renal fibrosis in diabetic nephropathy by sponging miR-218. J Cell Mol Med. https://doi.org/10.1111/jcmm.16129
Yan N, Wen L, Peng R, Li H, Liu H, Peng H, Sun Y, Wu T, Chen L, Duan Q, Sun Y, Zhou Q, Wei L, Zhang Z (2016) Naringenin ameliorated kidney injury through Let-7a/TGFBR1 signaling in diabetic nephropathy. J Diabetes Res 2016:8738760. https://doi.org/10.1155/2016/8738760
Yao T, Zha D, Hu C, Wu X (2020) Circ_0000285 promotes podocyte injury through sponging miR-654-3p and activating MAPK6 in diabetic nephropathy. Gene 747:144661. https://doi.org/10.1016/j.gene.2020.144661
Yun J, Ren J, Liu Y, Dai L, Song L, Ma X, Luo S, Song Y (2021) Circ-ACTR2 aggravates the high glucose-induced cell dysfunction of human renal mesangial cells through mediating the miR-205-5p/HMGA2 axis in diabetic nephropathy. Diabetol Metab Syndr 13:72. https://doi.org/10.1186/s13098-021-00692-x
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Contributions
ZF designed and performed the research; DW, FS, JC, DY, SL, and JT analyzed the data; ZF wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no interests to disclose.
Ethical Approval and Consent to Participate
Written informed consents were obtained from all participants and this study was permitted by the Ethics Committee of Yantaishan Hospital.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10528_2024_10694_MOESM1_ESM.tif
Supplementary file1 (TIF 1096 kb)
Fig. S1 Regulation of the circ-Luc7l/miR-205-5p/Tgfbr1 axis in proliferation, inflammation, and ECM accumulation of MC53 cells. A RT-qPCR for circ-Luc7l/miR-205-5p/Tgfbr1 mRNA expression in MC53 cells treated as indicated and then in HG-treated SV40-MES13 cells transfected with si-circ-Luc7l, si-circ-Luc7l+anti-miR-205-5p, or si-circ-Luc7l+Tgfbr1. B Cell viability was ascertained by CCK-8 assay. C Cell proliferation was determined by EdU assay. D, E The release of IL-6 and TNF-α was examined using commercial ELISA kits. F The protein levels of FN, Col I, and α-SMA were determined by Western blot. *P < 0.05, **P < 0.01, and ***P < 0.001.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fang, Z., Wang, D., Sun, F. et al. Circ-Luc7l Absence Attenuates Diabetic Nephropathy Progression by Reducing Mesangial Cell Excessive Proliferation, Inflammation, and Extracellular Matrix Accumulation via Mediating the miR-205-5p/Tgfbr1 Pathway. Biochem Genet (2024). https://doi.org/10.1007/s10528-024-10694-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10528-024-10694-9