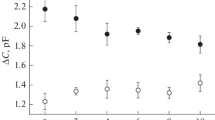
We studied the effects of ionophores on activity of Na+,Cl–(HCO −3 )-ATPase. The most significant effect on the activity of this enzyme was produced by protonophore 2,4-dinitrophenol. The effect of this drug largely depended on the cation and anion composition and pH of the incubation medium and its pH. Activity of Na+,Cl–(HCO −3 )-ATPase increased at neutral and weakly alkaline pH and decreased at pH below 6.5-6.7. In control animals (without histamine injection) with very weak or absent Na+,Cl–(HCO −3 )-ATPase activity, the observed effect of the protonophore on ATPase activity was also virtually absent. The stimulatory effect of other ionophores (monensine, valinomycin, and A23187) was significantly weaker and depended on pH of the incubation medium, its cationic and anionic composition, and concentration of these ionophores.
Similar content being viewed by others
References
Metelsky ST. Inherent metabolic diseases and absorption (sodium dependent) of nutrients in the gut: is there a link between them. Patogenez. 2012;10(4):4-15. Russian.
Shubin VS, Yurkiv VA. Properties of Na+/HCO −3 (Cl-)-stimulated ATPase activity in rabbit small intestinal mucosa. Biokhimiya. 1995;60(6):864-873. Russian.
Yurkiv VA1, Melikhov VI, Shubin VS. Ca2+ ions regulate activity of Na+,Cl–(HCO −3 )-ATPase in the mucosa of rabbit small intestine. Bull. Exp. Biol. Med. 2015;158(3):326-328.
Chen TY, Hwang TC. CLC-0 and CFTR: chloride channels evolved from transporters. Physiol. Rev. 2008;88(2):351-387.
Donowitz M, Li X. Regulatory binding partners and complexes of NHE3. Physiol. Rev. 2007;87(3):825-872.
Field M. Intestinal ion transport and the pathophysiology of diarrhea. J. Clin. Invest. 2003;111(7):931-943.
Hall RA, Ostedgaard LS, Premont RT, Blitzer JT, Rahman N, Welsh MJ, Lefkowitz RJ. A C-terminal motif found in the beta2-adrenergic receptor, P2Y1 receptor and cystic fibrosis transmembrane conductance regulator determines binding to the Na+/H+ exchanger regulatory factor family of PDZ proteins. Proc. Natl Acad. Sci. USA. 1998;95(15):8496-8501.
Miller C. CFTR: break a pump, make a channel. Proc. Natl Acad. Sci. USA. 2010;107(3):959-960.
Park M, Ko SB, Choi JY, Muallem G, Thomas PJ, Pushkin A, Lee MS, Kim JY, Lee MG, Muallem S, Kurtz I. The cystic fi brosis transmembrane conductance regulator interacts with and regulates the activity of the HCO −3 salvage transporter human Na+ ‐ HCO −3 cotransport isoform 3. J. Biol. Chem. 2002;277(52):50,503-50,509.
Parker MD, Boron WF. The divergence, actions, roles, and relatives of sodium-coupled bicarbonate transporters. Physiol. Rev. 2013;93(2):803-959.
Romero M.F, Fulton C.M, Boron W.F. The SLC4 family of HCO −3 transporters. Pflugers Arch. 2004;447(5):495–509.
Short DB, Trotter KW, Reczek D, Kreda SM, Bretscher A, Boucher RC, Stutts MJ, Milgram SL. An apical PDZ protein anchors the cystic fibrosis transmembrane conductance regulator to the cytoskeleton. J. Biol. Chem. 1998;273(31):19,797-19,801.
Sun F, Hug MJ, Lewarchik CM, Yun CH, Bradbury NA, Frizzell RA. E3KARP mediates the association of ezrin and protein kinase A with the cystic fibrosis transmembrane conductance regulator in airway cells. J. Biol. Chem. 2000;275(38):29 539-29 546.
Wang YZ, Cooke HJ, Su HC, Fertel R. Histamine augments colonic secretion in guinea pig distal colon. Am. J. Physiol. 1990;258(3, Pt 1):G432-G439.
Wingo CS, Smolka AJ. Function and structure of H-K-ATPase in the kidney. Am. J. Physiol. 1995;269(1, Pt 2):F1-F16.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 161, No. 6, pp. 715-718, June, 2016
Rights and permissions
About this article
Cite this article
Yurkiv, V.A., Melikhov, V.I. & Shubin, V.S. Effect of Ionophores on Activity of Na+,Cl–(HCO −3 )-ATPase. Bull Exp Biol Med 161, 763–766 (2016). https://doi.org/10.1007/s10517-016-3504-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-016-3504-1