Abstract
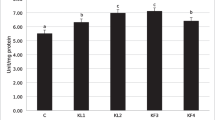
This study investigated the dietary thiamine (vitamin B1) requirement of Pacific white shrimp (Penaeus vannamei) for optimal growth performance, feed utilization, and innate immunity. Six test diets were formulated to contain thiamine at 3.32, 20.8, 38.7, 60.2, 80.7, and 108 mg/kg of diet (designated as T0, T20, T40, T60, T80, and T100, respectively). A total of 360 shrimp (6.03 ± 0.03 g) were randomly assigned into 24 acryl tanks (240 L) to be quadruplicated per treatment, and feeding was done in six equal portions daily for 58 days. The T20 diet fed group exhibited significantly higher growth performance compared to shrimp fed T0 and T100 diets. Feed conversion ratio and protein efficiency ratio were not significantly different among experimental groups. A significantly higher phenoloxidase activity was exhibited in T20 dietary group than T0 and T100 groups. Lysozyme activity was significantly enhanced in T20, T40, T60, and T80 groups than in T0 group and antiprotease activity was not significantly affected. Antioxidant parameters: superoxide dismutase and glutathione peroxidase activities were significantly elevated in T20 dietary treatment group. Significantly upregulated proPO gene expression was observed in T20 and T40 dietary treatment groups, although penaidine 3a and insulin-like growth factor-1 relative gene expressions were not significantly different. Glucose, total cholesterol, triglyceride, and total protein levels in hemolymph were not significantly different. Intestinal histology observations did not show significant differences in villi heights. A piecewise regression of weight gain percentage (WG%) estimates the optimal dietary thiamine requirement of P. vannamei at 19.7 mg/kg of diet.



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Amparyup P, Charoensapsri W, Tassanakajon A (2013) Prophenoloxidase system and its role in shrimp immune responses against major pathogens. Fish Shellfish Immunol 34:990–1001. https://doi.org/10.1016/j.fsi.2012.08.019
AOAC (2005) Official method of Analysis, 18th edn. Association of Officiating Analytical Chemists, Washington DC method 935.14 and 992.24
Bachère E, Destoumieux D, Bulet P (2000) Penaeidins, antimicrobial peptides of shrimp: a comparison with other effectors of innate immunity. Aquaculture 191:71–88. https://doi.org/10.1016/S0044-8486(00)00419-1
Bettendorff L, Lakaye B, Kohn G, Wins P (2014) Thiamine triphosphate: a ubiquitous molecule in search of a physiological role. Metab Brain Dis 29:1069–1082. https://doi.org/10.1007/s11011-014-9509-4
Boonyaratpalin M (1998) Nutrition of Penaeus merguiensis and Penaeus idicus. Rev Fish Sci 6(1–2):69–78. https://doi.org/10.1080/10641269891314203
Campa-Córdova AI, Hernández-Saavedra NY, De Philippis R, Ascencio F (2002) Generation of superoxide anion and SOD activity in haemocytes and muscle of American white shrimp (Litopenaeus vannamei) as a response to β-glucan and sulphated polysaccharide. Fish Shellfish Immunol 12:353–366. https://doi.org/10.1006/fsim.2001.0377
Chen HY, Wu FC, Tang SY (1991) Thiamine requirement of juvenile shrimp (Penaeus monodon). J Nutr 121:1984–1989. https://doi.org/10.1093/jn/121.12.1984
Chen YY, Chen JC, Tseng KC, Lin YC, Huang CL (2015) Activation of immunity, immune response, antioxidant ability, and resistance against Vibrio alginolyticus in white shrimp Litopenaeus vannamei decrease under long-term culture at low pH. Fish Shellfish Immunol 46:192–199. https://doi.org/10.1016/j.fsi.2015.05.055
Conklin D (1989) Vitamin requirements of juvenile penaeid shrimp. Actes de colloques Ifremer Tahiti French Polynesia, pp 287–308. Retrieved February 15, 2023, from https://archimer.ifremer.fr/doc/00000/1469/
De Caterina R, Madonna R (2004) Nutrients and gene expression. In: Simopoulos AP, Ordovas JM (eds) World Review of Nutrition and Dietetics: volume 93. Karger, Basel, pp 99–133
Delafontaine P, Song YH, Li Y (2004) Expression, regulation, and function of IGF-1, IGF-1R, and IGF-1 binding proteins in blood vessels. Arterioscler Thromb Vasc Biol 24:435–444. https://doi.org/10.1161/01.ATV.0000105902.89459.09
Depeint F, Bruce WR, Shangari N, Mehta R, O’Brien PJ (2006) Mitochondrial function and toxicity: role of the B vitamin family on mitochondrial energy metabolism. Chem Biol Interact 163:94–112. https://doi.org/10.1016/j.cbi.2006.04.014
Deshimaru O, Kuroki K (1979) Requirement of prawn for dietary thiamine, pyridoxine, and choline chloride. Bull Jpn Soc Sci Fish 45:363–367. https://doi.org/10.2331/suisan.45.363
Ellis AE (1990) Serum antiprotease in fish. In: Stolen JS, Fletcher TC, Anderson DP, Roberson WB, Muiswinkel WB (eds) Techniques in fish immunology. NJ: SOS Publication Fair Haven, pp 95–99
FAO (2022) The State of World Fisheries and Aquaculture 2022. Towards Blue Transformation FAO, Rome. https://doi.org/10.4060/cc0461en
Fattal-Valevski A (2011) Thiamine (vitamin B1). Evid Based Complementary Altern Med 16:12–20. https://doi.org/10.1177/1533210110392941
Folch J, Lees M, Stanley GS (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226:497–509
Grunclová L, Fouquier H, Hypša V, Kopáček P (2003) Lysozyme from the gut of the soft tick Ornithodoros moubata: the sequence, phylogeny and post-feeding regulation. Dev Comp Immunol 27:651–660. https://doi.org/10.1016/S0145-305X(03)00052-1
Hansen AC, Waagbø R, Hemre GI (2015) New B vitamin recommendations in fish when fed plant-based diets. Aquac Nutr 21:507–527. https://doi.org/10.1111/anu.12342
Harder AM, Ardren WR, Evans AN, Futia MH, Kraft CE, Marsden JE, Richter CA, Rinchard J, Tillitt DE, Christie MR (2018) Thiamine deficiency in fishes: causes, consequences, and potential solutions. Rev Fish Biol Fish 28:865–886. https://doi.org/10.1007/s11160-018-9538-x
Hasanthi M, Lee KJ (2023) Dietary niacin requirement of Pacific white shrimp (Litopenaeus vannamei). Aquaculture 739169. https://doi.org/10.1016/j.aquaculture.2022.739169
Hellio C, Bado-Nilles A, Gagnaire B, Renault T, Thomas-Guyon H (2007) Demonstration of a true phenoloxidase activity and activation of a ProPO cascade in Pacific oyster, Crassostrea gigas (Thunberg) in vitro. Fish Shellfish Immunol 22:433–440. https://doi.org/10.1016/j.fsi.2006.06.014
Hernández-López J, Gollas-Galván T, Vargas-Albores F (1996) Activation of the prophenoloxidase system of the brown shrimp Penaeus californiensis Holmes. Comp Biochem Physiol Part-C: Toxicol Pharmacol 113:61–66. https://doi.org/10.1016/0742-8413(95)02033-0
Holmblad T, Söderhäll K (1999) Cell adhesion molecules and antioxidative enzymes in a crustacean, possible role in immunity. Aquaculture 172:111–123. https://doi.org/10.1016/S0044-8486(98)00446-3
Huang HH, Feng L, Liu Y, Jiang J, Jiang WD, Hu K, Li SH, Zhou XQ (2011) Effects of dietary thiamine supplement on growth, body composition and intestinal enzyme activities of juvenile Jian carp (Cyprinus carpio var. Jian). Aquac Nutr 17:233–240. https://doi.org/10.1111/j.1365-2095.2010.00756.x
Huang JW, Tian LX, Du ZY, Yang HJ, Liu YJ (2007) Effects of dietary thiamine on the physiological status of the grouper Epinephelus coioides. Fish Physiol Biochem 33:167–172. https://doi.org/10.1007/s10695-007-9127-8
Huang X, Xia M, Wang H, Jin M, Wang T, Zhou Q (2015) Dietary thiamine could improve growth performance, feed utilization and non-specific immune response for juvenile Pacific white shrimp (Litopenaeus vannamei). Aquac Nutr 21:364–372. https://doi.org/10.1111/anu.12167
Hultmark D, Steiner H, Rasmuson T, Boman HG (1980) Insect immunity. Purification and properties of three inducible bactericidal proteins from hemolymph of immunized pupae of Hyalophora cecropia. Eur J Biochem 106:7–16. https://doi.org/10.1111/j.1432-1033.1980.tb05991.x
Ichikawa K, Shib Y, Yamazaki M, Serizawa N (1997) Thiamine increases expression of yeast gene. Biosci Biotechnol Biochem 61:1221–1224. https://doi.org/10.1271/bbb.61.1221
Jiang M, Huang F, Zhao Z, Wen H, Wu F, Liu W, Wang W (2014) Dietary thiamine requirement of juvenile grass carp, Ctenopharyngodon idella. J World Aquacult Soc 45:461–468. https://doi.org/10.1111/jwas.12132
Jones KS, Parkington DA, Cox LJ, Koulman A (2021) Erythrocyte transketolase activity coefficient (ETKAC) assay protocol for the assessment of thiamine status. Ann N Y Acad Sci 1498:77–84. https://doi.org/10.1111/nyas.14547
Li E, Wang X, Chen K, Xu C, Qin JG, Chen L (2017) Physiological change and nutritional requirement of Pacific white shrimp Litopenaeus vannamei at low salinity. Rev Aquac 9:57–75. https://doi.org/10.1111/raq.12104
Li E, Xu C, Wang X, Wang S, Zhao Q, Zhang M, Qin JG, Chen L (2018) Gut microbiota and its modulation for healthy farming of Pacific white shrimp Litopenaeus vannamei. Rev Fish Sci Aquac 26:381–399. https://doi.org/10.1080/23308249.2018.1440530
Li XY, Huang HH, Hu K, Liu Y, Jiang WD, Jiang J, Li SH, Feng L, Zhou XQ (2014) The effects of dietary thiamin on oxidative damage and antioxidant defence of juvenile fish. Fish Physiol Biochem 40:673–687. https://doi.org/10.1007/s10695-013-9875-6
Lim C, Yildirim-Aksoy M, Barros MM, Klesius P (2011) Thiamine requirement of Nile tilapia, Oreochromis niloticus. J World Aquac Soc 42:824–833. https://doi.org/10.1111/j.1749-7345.2011.00531.x
Liu CH, Tseng MC, Cheng W (2007) Identification and cloning of the antioxidant enzyme, glutathione peroxidase, of white shrimp, Litopenaeus vannamei, and its expression following Vibrio alginolyticus infection. Fish Shellfish Immunol 23:34–45. https://doi.org/10.1016/j.fsi.2006.09.002
Lonsdale D (2015) Thiamine and magnesium deficiencies: keys to disease. Med hypotheses 84:129–134. https://doi.org/10.1016/j.mehy.2014.12.004
Lukienko PI, Mel'Nichenko NG, Zverinskii IV, Zabrodskaya SV (2000) Antioxidant properties of thiamine. Bull Exp Biol Med 130:874–876. https://doi.org/10.1007/BF02682257
Magnadóttir B, Jónsdóttir H, Helgason S, Björnsson B, Jørgensen TØ, Pilström L (1999) Humoral immune parameters in Atlantic cod (Gadus morhua L.): I. The effects of environmental temperature. Comp Biochem Physiol - B Biochem Mol Biol 122:173–180. https://doi.org/10.1016/S0305-0491(98)10156-6
Mahmood S, Dani,HM, Mahmood A (1984) Effect of dietary thiamine deficiency on intestinal functions in rats. Am J Clin Nutr 40:226-234. https://doi.org/10.1093/ajcn/40.2.226
Martin PR (2001) Molecular mechanisms of thiamine utilization. Curr Mol Med 1:197–207. https://doi.org/10.2174/1566524013363870
Martin PR, Singleton CK, Hiller-Sturmhöfel S (2003) The role of thiamine deficiency in alcoholic brain disease. Alcohol Res Health 27:134–142
McDowell LR (1989) Vitamins in animal and human nutrition. Iowa State University Press, Ames IA, p 763
Morris PC, Davies SJ (1995) Thiamine supplementation of diets containing varied lipid: carbohydrate ratio given to gilthead seabream (Sparus aurata L.). J Anim Sci 61:597–603. https://doi.org/10.1017/S1357729800014193
NRC (National Research Council) (2011) Nutrient Requirements of Fish and Shrimp. The National Academies Press, Washington DC. https://doi.org/10.17226/13039
Okumura T (2007) Effects of lipopolysaccharide on gene expression of antimicrobial peptides (penaeidins and crustin), serine proteinase and prophenoloxidase in haemocytes of the Pacific white shrimp, Litopenaeus vannamei. Fish Shellfish Immunol 22:68–76. https://doi.org/10.1016/j.fsi.2006.03.013
Paul B, Das S, Girj S, Mohanty S (2010) Water soluble Vitamins in Aquaculture Nutrition. Fish Chimes 30:13–16
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res 29:45–45. https://doi.org/10.1093/nar/29.9.e45
Ren Q, Sun RR, Zhao XF, Wang JX (2009) A selenium-dependent glutathione peroxidase (Se-GPx) and two glutathione S-transferases (GSTs) from Chinese shrimp (Fenneropenaeus chinensis). Comp Biochem Physiol Part - C: Toxicol Pharmacol 149:613–623. https://doi.org/10.1016/j.cbpc.2009.01.007
Sandau K, Pfeilschifter J, Brüne B (1998) Nitrosative and oxidative stress induced heme oxygenase-1 accumulation in rat mesangial cells. Eur J Pharmacol 342:77–84. https://doi.org/10.1016/S0014-2999(97)01321-6
Sannino DR, Kraft CE, Edwards KA, Angert ER (2018) Thiaminease I provides a growth advantage by salvaging precursors from environmental thiamine and its analogs in Burkholderia thailandensis. Appl Environ Microbiol 84:e01268–e01218. https://doi.org/10.1128/AEM.01268-18
Saurabh S, Sahoo PK (2008) Lysozyme: an important defence molecule of fish innate immune system. Aquac Res 39:223–239. https://doi.org/10.1111/j.1365-2109.2007.01883.x
Shearer KD (2000) Experimental design, statistical analysis and modelling of dietary nutrient requirement studies for fish: a critical review. Aquac Nutr 6:91–102. https://doi.org/10.1046/j.1365-2095.2000.00134.x
Söderhäll K, Cerenius L (1998) Role of the prophenoloxidase-activating system in invertebrate immunity. Curr Opin Immunol 10:23–28. https://doi.org/10.1016/S0952-7915(98)80026-5
Strickland JDH, Parsons, TR (1972). A practical handbook of seawater analysis.
Sun M, Li XF, Ge YP, Zhang L, Liu B, Liu WB (2022) Dietary thiamine requirement and its effects on glycolipid metabolism in oriental river prawn (Macrobrachium nipponense). Aquaculture 550:737824. https://doi.org/10.1016/j.aquaculture.2021.737824
Thamizhvanan S, Nafeez Ahmed A, Vinoth Kumar D, Vimal S, Majeed SA, Taju G, Hauton C, Sahul Hameed AS (2021) Silencing of prophenoloxidase (proPO) gene in freshwater prawn, Macrobrachium rosenbergii, makes them susceptible to white spot syndrome virus (WSSV). J Fish Dis 44:573–584. https://doi.org/10.1111/jfd.13297
Whitfield KC, Bourassa MW, Adamolekun B, BergeronG BL, Brown KH, Cox L, Fattal-Valevski A, Fischer PR, Frank EL, Hiffler L, Hlaing LM, Jefferds ME, Kapner H, Kounnavong S, Mousavi MPS, Roth DE, Tsaloglou MN, Wieringa F, Combs GF Jr (2018) Thiamine deficiency disorders: diagnosis, prevalence, and a roadmap for global control programs. Ann N Y Acad Sci 1430:3–43. https://doi.org/10.1111/nyas.13919
Xiang X, Zhou XQ, Chen GF, Wu P, Zheng ZL (2016) Effect of graded levels of dietary thiamine on the growth performance, body composition and haemato-biochemical parameters of juvenile Sclizothorax prenanti. Aquac Nutr 22:691–697. https://doi.org/10.1111/anu.12291
Xu C, Li YY, Brown PB, Liu WB, Gao LL, Ding ZR, Li XF, Xie DZ (2022) Interactions between dietary carbohydrate and thiamine: implications on the growth performance and intestinal mitochondrial biogenesis and function of Megalobrama amblycephala. Br J Nutr 127:321–334. https://doi.org/10.1017/S000711452100101X
Xun P, Lin H, Wang R, Huang Z, Zhou C, Yu W, Huang Q, Tan L, Wang Y, Wang J (2019) Effects of dietary vitamin B1 on growth performance, intestinal digestion and absorption, intestinal microflora and immune response of juvenile golden pompano (Trachinotus ovatus). Aquaculture 506:75–83. https://doi.org/10.1016/j.aquaculture.2019.03.017
Yang G, Yang L, Zhao Z, Wang J, Zhang X (2012) Signature miRNAs involved in the innate immunity of invertebrates. PLoS One 7:e39015. https://doi.org/10.1371/journal.pone.0039015
Zehra S, Khan MA (2018) Dietary thiamin requirement of fingerling Channa punctatus (Bloch) based on growth, protein gain, liver thiamin storage, RNA/DNA ratio and biochemical composition. Aquac Nutr 24:1015–1023. https://doi.org/10.1111/anu.12638
Zhao J, Qiu L, Ning X, Chen A, Wu H, Li C (2010) Cloning and characterization of an invertebrate type lysozyme from Venerupis philippinarum. Comp Biochem Physiol - B Biochem Mol Biol 156:56–60. https://doi.org/10.1016/j.cbpb.2010.02.001
Zhu W, Mai K, Wu G (2002) Thiamine requirement of juvenile abalone, Haliotis discus hannai Ino. Aquaculture 207:331–343. https://doi.org/10.1016/S0044-8486(01)00749-9
Zhou D, Wang C, Zheng J, Zhao J, Wei S, Xiong Y, Limbu SM, Kong Y, Cao F, Ding Z (2022) Dietary thiamine modulates carbohydrate metabolism, antioxidant status, and alleviates hypoxia stress in oriental river prawn Macrobrachium nipponense (de Haan). Fish Shellfish Immunol 131:42–53. https://doi.org/10.1016/j.fsi.2022.09.059
Acknowledgements
This research was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIR) (No. 2021R1A2C2008384) and Basic Science Research Program through the NRF funded by the Ministry of Education (2019R1A6A1A03033553).
Funding
This research was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIR) (No. 2021R1A2C2008384) and Basic Science Research Program through the NRF funded by the Ministry of Education (2019R1A6A1A03033553).
Author information
Authors and Affiliations
Contributions
Udith Wijemanna: Investigation, Formal analysis, Writing – original draft.
Kyeong-Jun Lee: Conceptualization, Methodology, Supervision, Writing – review & editing.
The final manuscript was approved by all the authors.
Corresponding author
Ethics declarations
Animal and human rights statement
Experimental protocols followed the guidelines of the Animal Care and Use Committee of Jeju National University, South Korea. All protocols of animal work were approved by the Jeju National University, South Korea. This study does not contain any studies with human participation.
Competing interests
The authors declare no competing interests.
Additional information
Handling editor: Laurence Mercier
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wijemanna, U., Lee, KJ. Dietary thiamine requirement and its effects on growth and innate immunity of Pacific white shrimp (Penaeus vannamei). Aquacult Int (2023). https://doi.org/10.1007/s10499-023-01308-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10499-023-01308-z