Abstract
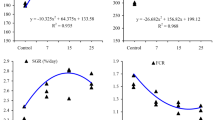
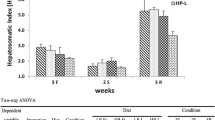
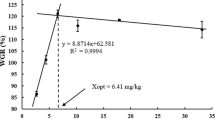
This study was conducted to evaluate the effects of dietary thiamin on the physiological status of the juvenile grouper, Epinephelus coioides. Graded levels of thiamin (0.08, 0.50, 2.12, 3.15, 4.63, 12.37 mg thiamin kg−1 diet) were fed to grouper juveniles (mean weight: 16.97 ± 0.14 g) for 10 weeks. Although fish fed the thiamin-deficient (TD) diet showed no obvious symptoms of thiamin deficiency or increased mortality, those fed the lowest doses of thiamin (0.08 and 0.50 mg thiamin kg−1 diet) had significantly decreased transketolase activity in the liver. In addition, the level of liver thiobarbituric acid reactive substances in fish fed the TD diet was 33–67% higher than that in fish with the thiamin-supplemented diet. There were no significant differences in superoxide dismutase activity between the different groups of fish.



Similar content being viewed by others
References
Amcoff P, Elofsson UOE, Börjeson H et al (2002) Alterations of dopaminergic and serotonergic activity in the brain of sea-run Baltic salmon suffering a thiamine deficiency-related disorder. J Fish Biol 60:1407–1416
AOAC (Association of Official Analytical Chemists) (1990) Thiamin (vitamin B1) in foods. Fluorometric method. Official Methods of Analysis, no. 942.23, 15th edn. AOAC, Arlington, Va.
Ba A, N’Douba V, D’Almeida MA et al (2005) Effects of maternal thiamine deficiencies on the pyramidal and granule cells of the hippocampus of rat pups. Acta Neurobiol Exp (Wars) 65:387–398
Baxter P (2003) Pyridoxine-dependent seizures: a clinical and biochemical conundrum. Biochim Biophys Acta 1647:36–41
Berthon HA, Kuchel PW, Nixon PF (1992) High control coefficient of transketolase in the nonoxidative pentose phosphate pathway of human erythrocytes: NMR, antibody, and computer simulation studiest. Biochemistry 31:12792–12798
Blair PV, Kobayashi R, Edwards III HM et al (1999) Dietary thiamin level influences levels of its diphosphate form and thiamin-dependent enzymic activities of rat liver. J Nutr 129:641–648
Boonyaratpalin M (1997) Nutrient requirements of marine food fish cultured in Southeast Asia. Aquaculture 151:283–313
Calingasan NY, Chun WJ, Park LC et al (1999) Oxidative stress is associated with region-specific neuronal death during thiamine deficiency. J Neuropathol Exp Neurol 58:946–958
Chen HY, Wu FC, Tang SY (1994) Sensitivity of transketolase to the thiamin status of juvenile marine shrimp (Penaeus monodon). Comp Biochem Phys A 109:655–659
Ciccia RM, Langlais PJ (2000) An examination of the synergistic interaction of ethanol and thiamine deficiency in the development of neurological signs and long-term cognitive and memory impairments. Alcohol Clin Exp Res 24:622–634
Cowey CB, Adron JW, Knox D et al. (1975) Studies on the nutrition of marine flatfish. The thiamine requirement of turbot (Scophthalmus maximus). Br J Nutr 34:383–390
Elmadfa I, Majchrzak D, Rust P et al. (2001) The thiamine status of adult humans depends on carbohydrate intake. Int J Vitam Nutr Res 71:217–221
Gibson GE, Ksiezak-Reding H, Sheu K-FR et al (1984) Correlation of enzymatic, metabolic, and behavioral deficits in thiamin deficiency and its reversal. Neurochem Res 9:803–814
Giguère J-F, Butterworth RF (1987) Activities of thiamine-dependent enzymes in two experimental models of thiamine deficiency encephalopathy: 3. transketolase. Neurochem Res 12:305–310
Hashimoto Y, Arai S, Nose T (1970) Thiamine deficiency symptoms experimentally induced in the eel. Nippon Suisan Gakkai Shi 36:791–797
Hazell AS, Pannunzio P, Rama Rao KV et al (2003) Thiamine deficiency results in downregulation of the GLAST glutamate transporter in cultured astrocytes. Glia 43:175–184
Horecker BL (1957) The orcinol reaction for mixtures of pentose and heptulose. In: Colowick SP, Kaplan NO (eds) Methods of enzymology III. Academic Press, New York, pp 105–107
Huang JW, Tian LX, Du ZY et al (2005) Pyridoxine deficiency of grouper, Epinephelus coioides: physiological and biochemical alteration. Fish Physiol Biochem 31:331–337
Langlais PJ, Anderson G, Gu SX et al (1997) Increased cerebral free radical production during thiamine deficiency. Metab Brain Dis 12:137–143
Lehmitz R, Spannhof L (1977) Transketolase activity in thiamine deficiency of the kidney in rainbow trout (Salmo gairdneri) following continous feeding with raw herring. Arch Tierernahr 27:287–295
Lonsdale D (2006) A review of the biochemistry, metabolism and clinical benefits of thiamin(e) and its derivatives. Evid Based Complement Alternat Med 3:49–59
Lukienko PI, Mel’nichenko NG, Zverinskii IV et al (2001) Antioxidant properties of thiamine. Bull Exp Biol Med 130:874–876
Martin PR, Singleton CK, Hiller-Sturmhöfel S (2003) The role of thiamine deficiency in alcoholic brain disease. Alcohol Res Health 27:134–142
Masumoto T, Hardy RW, Casillas E (1987) Comparison of tranketolase activity and thiamin pyrophosphate levels in erythrocytes and liver of rainbow trout (Salmo gairdneri) as indicators of thiamine status. J Nutr 117:1422–1426
Morito CLH, Conrad DH, Hilton JW (1986) The thiamin deficiency signs and requirement of rainbow trout (Salmo gairdneri, Richardson). Fish Physiol Biochem 1:93–104
Mulholland PJ (2006) Susceptibility of the cerebellum to thiamine deficiency. Cerebellum 5:55–63
Murai T, Andrews JW (1978) Thiamin requirement of channel catfish fingerlings. J Nutr 108:176–180
Nakagawasai O, Tadano T, Niijima F et al (2000) Immunohistochemical estimation of rat brain somatostatin on avoidance learning impairment induced by thiamine deficiency. Brain Res Bull 51:47–55
Pannunzio P, Hazell AS, Pannunzio M et al (2000) Thiamine deficiency results in metabolic acidosis and energy failure in cerebellar granule cells: an in vitro model for the study of cell death mechanisms in Wernicke’s encephalopathy. J Neurosci Res 62:286–292
Pekovich SR, Martin PR, Singleton CK (1998) Thiamine deficiency decreases steady-state transketolase and pyruvate dehydrogenase but not α-ketoglutarate dehydrogenase mRNA levels in three human cell types. J Nutr 128:683–687
Rains TM, Emmert JL, Baker DH et al (1997) Minimum thiamin requirement of weanling Sprague-Dawley outbred rats. J Nutr 127:167–170
Shaik Mohamed J, Sivaram V, Christopher Roy TS et al (2003) Dietary vitamin A requirement of juvenile greasy grouper (Epinephelus tauvina). Aquaculture 219:693–701
Shangari N, Bruce WR, Poon R et al (2003) Toxicity of glyoxals-role of oxidative stress, metabolic detoxification and thiamine deficiency. Biochem Soc Trans 31:1390–1393
Sheu K-FR, Calingasan NY, Dienel GA et al (1996). Regional reductions of transketolase in thiamine-deficient rat brain. J Neurochem 67:684–691
Singleton CK, Martin PR (2001) Molecular mechanisms of thiamine utilization. Curr Mol Med 1:197–207
Todd KG, Butterworth RF (1999) Early microglial response in experimental thiamine deficiency: an immunohistochemical analysis. Glia 25:190–198
Warnock LG (1970) A new approach to erythrocyte transketolase measurement. J Nutr 100:1057–1062
Wilson RP (1994) Utilization of dietary carbohydrate by fish. Aquaculture 124:67–80
Wood CM (1993) Ammonia and urea metabolism and excretion. In: Evans DH (ed) The physiology of fishes. CRC Press, Boca Raton, pp 384–385
Zhu W, Mai KS, Wu GT (2002) Thiamin requirement of juvenile abalone, Haliotis discus hannai Ino. Aquaculture 207:331–343
Acknowledgements
This project was supported by the Key Technologies R&D Program during the tenth 5-year plan, China (grant no. 2001DA505D/06). The authors are grateful to Dr. Wei Zhu (Ocean University of China) for providing some references.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, J.W., Tian, L.X., Du, Z.Y. et al. Effects of dietary thiamin on the physiological status of the grouper Epinephelus coioides . Fish Physiol Biochem 33, 167–172 (2007). https://doi.org/10.1007/s10695-007-9127-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-007-9127-8