Abstract
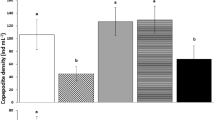
Studies considering broodstock conditioning of Ruditapes decussatus mostly used flagellates and diatoms for feeding, mainly Isochrysis galbana and Chaetoceros calcitrans, respectively. The present study evaluated the effect of different microalgal diets on gonadic maturation of R. decussatus including three microalgae species (C. calcitrans, Tetraselmis suecica, and Nannochloropsis oculata) which were used to prepare six diets: three monospecific and three bispecific mixed diets at a proportion of 1:1. Clams were daily fed at a ratio of 1% dry weight of algae/live weight of clam and unfed treatment was used as control. Results showed that clams fed C. calcitrans alone or mixed with T. suecica attained earlier full maturity with the highest response to spawning induction (after 34 conditioning days) and largest proportion in the partial spawning phase for both, followed by T. suecica alone after 48 days. In contrast, N. oculata alone or mixed diets demonstrated poor performance for gonadal maturation. Unfed treatment did not show any sign of maturation. The results indicated that T. suecica has high conditioning potential like C. calcitrans and their mixture gave the highest benefit. Therefore, T. suecica can be recommended for R. decussatus broodstock conditioning.



Similar content being viewed by others
References
Aldrich JC, Crowley M (1986) Condition and variability in Mytilus edulis (L.) from different habitats in Ireland. Aquaculture 52:273–286. https://doi.org/10.1016/0044-8486(86)90370-4
Babinchak J, Ukeles R (1979) Epifluorescence microscopy, a technique for the study of feeding in Crassostrea virginica veliger larvae. Mar Biol 51:69–76. https://doi.org/10.1007/BF00389032
Beninger PG, Lucas A (1984) Seasonal variations in condition index, reproductive activity, and gross biochemical composition of two species of adult clam reared in a common habitat: Tapes decussatus L. (Jereys) and Tapes philippinarum (Adams and Reeve). J Exp Mar Biol Ecol 79:19–37
Berntsson KM, Jonsson PR, Wangberg SA, Carlsson AS (1997) Effects of broodstock diets on fatty acid composition, survival and growth rates in larvae of the European flat oyster, Ostrea edulis. Aquaculture 154(2):139–153. https://doi.org/10.1016/S0044-8486(97)00041-0
Chessa LA, Paesanti F, Pais A, Scardi M, Serra S, Vitale L (2005) Perspective for development of low impact aquaculture in western Mediterranean lagoon: the case of the carpet clam Tapes decussatus. Aquac Int 13:147–155. https://doi.org/10.1007/s10499-004-9022-6
Coutteau P (1996) Microalgae. In: Lavens P, Sorgeloos PŽ (eds) Manual on the production and use of live food for aquaculture. FAO fisheries technical paper no. 361. FAO, Rome, pp 7–48
Dare P,J, Edwards D,B (1975) Seasonal changes in flesh and weight and biochemical composition of mussels (Mytilus edulis L.) in the Conwy Estuary, North Wales. J Exp Mar Biol Ecol 18:89–97. https://doi.org/10.1016/0022-0981(75)90066-0
Delgado M, Pérez-Camacho A (2002) Efectos de la rácion de alimento en el desarrollo gonadal de la almeja Ruditapes decussatus (L.). Bol Inst Esp Oceanogr 18(1–4):293–300
Delgado M, Perez Camacho A (2003) Study of gonadal development in Ruditapes decussatus (mollasca bivalvia), using image analysis techniques: influence of food ration and energy balance. J Shellfish Res 22:435–441
Delgado M, Pérez-Camacho A (2005) Histological study of the gonadal development of Ruditapes decussatus (L.): (Mollusca: Bivalvia) and its relationship with available food. Scienta Marina 69:87–97
Delgado M, Pérez-Camacho A (2007a) Influence of temperature on gonadal development of Ruditapes philippinarum (Adams and Reeve, 1850) with special reference to ingested food and energy balance. Aquaculture 264:398–407. https://doi.org/10.1016/j.aquaculture.2006.11.009
Delgado M, Pérez-Camacho A (2007b) Comparative study of gonadal development of Ruditapes philippinarum (Adam and Reeve) and Ruditapes decussatus (L.) (Mollusca: Bivalvia): influence of temperature. Sci Mar 71:471–484
Delgado M, Pérez-Camacho A, Labarta U, Fernández-Reiriz MJ (2004) The role of lipids in the gonadal development of the clam Ruditapes decussatus (L.). Aquaculture 241:395–411. https://doi.org/10.1016/j.aquaculture.2004.07.018
Dhont J, Lavens P (1996) Tank production and use of on grown Artemia. In: Lavens P, Sorgeloos PŽ (eds) Manual on the production and use of live food for aquaculture. FAO fisheries technical paper no. 361. FAO, Rome, pp 164–195
DuBois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356. https://doi.org/10.1021/ac60111a017
Epifanio C (1979) Comparison of yeast and algal diets for bivalve molluscs. Aquaculture 16:187–192. https://doi.org/10.1016/0044-8486(79)90106-6
Fernández-Reiriz M,J, Labarta U, Albentosa M, Pe’rez-Camacho A (1999) Lipid profile and growth of the clam spat, Ruditapes decussatus (L), fed with microalgal diets and cornstarch. Comp Biochem Physiol Part B 124:309–318. https://doi.org/10.1016/S0305-0491(99)00129-7
Folch J, Lees M, Sloane Stanley GH (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226:497–509
Guerra Díaz, A., Lodeiros Seijo, C., Baptista Gaspar, M., da Costa González, F (2011) Razor clams: biology, aquaculture and fisheries. Consellería do Mar, Xunta de Galicia, Santiago de Compostela
Guillard RRL (1975) Culture of phytoplankton for feeding marine invertebrates. In: Smith WL, Chanley MH (eds) Culture of marine invertebrate Animals. Plenum Press, New York, pp 26–60. https://doi.org/10.1007/978-1-4615-8714-9_3
Hamida L, Medhioub MN, Cochard JC, Le Pennec M (2004) Evaluation of the effects of serotonin (5-HT) on oocyte competence in Ruditapes decussatus (Bivalvia, Veneridae). Aquaculture 239:413–420. https://doi.org/10.1016/j.aquaculture.2004.05.010
Holland, D.A (1978) Lipid reserves and energy metabolism in the larvae of benthic marine invertebrates. In: Malins DC, Sargent JR (eds) Biochemistry and biophysical perspectives in marine biology, vol. 4. Academic Press: 85–123
Helm MM, Bourne N (2004) Hatchery culture of bivalves. A practical manual. FAO, Roma
Helm MM, Holland DL, Stephenson RR (1973) The effect of supplementary algal feeding of a hatchery breeding stock of Ostrea edulis L. on larval vigour. J Mar Biol Assoc U K 53:673–684. https://doi.org/10.1017/S0025315400058872
Iglesias JIP, Camacho C, Navarro E, Labarta U, Beiras R, Hawkins AJS, Widdows J (1996) Microgeographic variability in feeding, absorption and condition of mussels (Mytilus galloprovincialis Lmk): a transplant experiment. J Shellfish Res 15:673–680
Joaquim S., Matias D., Moreno O. (2008) Cultivo de bivalvos en maternidade. Instituto de Investigación y Formación Agraria y Pesquera. Consejería de Agricultura y Pescas. Junta de Andalucia
Liu W, Alabi AO, Pearce CM (2008) Fertilization and embryonic development in the basket cockle, Clinocardium nuttallii. J Shellfish Res 27:393–397. https://doi.org/10.2983/0730-8000(2008)27[393:FAEDIT]2.0.CO;2
Lora-Vilchis MC, Maeda-Martı’nez AN (1997) Ingestion and digestion index of catarina scallop Argopecten ventricosus-circularis, Sowerby II, 1842, veliger larvae with ten microalgae species. Aquac Res 28:905–910
Marasigan ET, Laureta LV (2001) Broodstock maintenance and early gonadal maturation of Pholas orientalis (Bivalvia: Pholadidae). J Shellfish Res 20:1095–1099
Martinez G (1991) Seasonal variation in biochemical composition of three size classes of Chilean scallop Argopecten purpuratus Lamarck, 1819. Veliger 34:335–343
Martínez-Fernández E, Acosta-Salmóna H, Rangel-Dávalosa C (2004) Ingestion and digestion of 10 species of microalgae by winged pearl oyster Pteria sterna (Gould, 1851) larvae. Aquaculture 230:417–423. https://doi.org/10.1016/S0044-8486(03)00416-2
Marshall R, McKinley S, Pearce CM (2010) Effects of nutrition on larval growth and survival in bivalves. Rev Aquac 2:33–55
Matias D, Joaquim S, Leitão A, Massapina C (2009) Effect of geographic origin, temperature, and timing of broodstock collection on conditioning, spawning success, and larval viability of Ruditapes decussatus (Linné, 1758). Aquac Int 17:257–271. https://doi.org/10.1007/s10499-008-9197-3
Matias D, Joaquim S, Ramos M, Sobral P, Leitão A (2011) Biochemical compounds dynamics during larval development of the carpet-shell clam (Linnaeus, 1758): effects of mono-specific diets and starvation. Helgol Mar Res 65:369–379. https://doi.org/10.1007/s10152-010-0230-3
Matias D, Joaquim S, Matias AM, Moura P, Teixeira de Sousa J, Sobral P, Leitão A (2013) The reproductive cycle of the European clam Ruditapes decussatus (L., 1758) in two Portuguese populations: Implications for management and aquaculture programs. Aquaculture 406:51–62. https://doi.org/10.1016/j.aquaculture2013.04.030
Matias, D., Ben-Hamadou, R., Joaquim, S., Matias, A., M., Sobral, P., Leitão., A. 2015. The influence of different microalgal diets on European clam (Ruditapes decussatus, Linnaeus, 1758) larvae culture performances. Aquac Res 46: 2527–2543. DOI: https://doi.org/10.1111/are.12417
Matias D, Joaquim S, Matias AM, Leitão A (2016) Reproductive effort of the European clam Ruditapes decussatus (Linnaeus, 1758): influence of different diets and temperatures. Invertebr Reprod Dev 60:49–58. https://doi.org/10.1080/07924259.2015.1126537
Mladineo I, Peharda M, Orhanović S, Bolotin J, Pavela-Vrančić M, Treursić B (2007) The reproductive cycle, condition index and biochemical composition of the horse-bearded mussel Modiolus barbatus. Helgol Mar Res 61:183–192. https://doi.org/10.1007/s10152-007-0065-8
Ojea J, Pazos AJ, Martínez D, Novoa S, Sánchez JL, Abad M (2004) Seasonal variation in weight and biochemical composition of the tissues of Ruditapes decussatus in relation to the gametogenic cycle. Aquaculture 238:451–468. https://doi.org/10.1016/j.aquaculture.2004.05.022
Ojea J, Pazos AP, Martínez D, Novoa S, García-Martínez P, Sánchez JL, Abad M (2008) Effects of temperature regime on broodstock conditioning of Ruditapes decussatus. J Shellfish Res 27:1093–1100. https://doi.org/10.2983/0730-8000-27.5.1093
Payne MF, Rippingale RJ (2000) Evaluation of diets for culture of the calanoid copepod Gladioferens imparipes. Aquaculture 187:85–96. https://doi.org/10.1016/S0044-8486(99)00391-9
Pérez-Camacho A, Delgado M, Fernández-Reiriz MJ, Labarta U (2003) Energy balance, gonad development and biochemical composition in the clam Ruditapes decussatus. Mar Ecol Prog Ser 258:133–145. https://doi.org/10.3354/meps258133
Pita IM, Clara SL, Prieto E, Moreno O (2011) The effect of diet on gonadal development of the smooth Venus clam Callista chione (Mollusca: Bivalvia). J Shellfish Res 30:295–301. https://doi.org/10.2983/035.030.0215
Pronker A, Nevejan N, Peene F, Geijsen P, Sorgeloos P (2008) Hatchery broodstock conditioning of the blue mussel Mytilus edulis (Linnaeus 1758). Part I. Impact of different micro-algae mixtures on broodstock performance. Aquac Int 16:297–307. https://doi.org/10.1007/s10499-007-9143-9
Serdar S, Lök A (2009) Gametogenic cycle and biochemical composition of the transplanted carpet shell clam Tapes decussatus, Linnaeus 1758 in Sufa (Homa) Lagoon, Izmir, Turkey. Aquaculture 293:81–88. https://doi.org/10.1016/j.aquaculture.2009.03.052
Sobral P, Widdows J (2000) Effects of increasing current velocity, turbidity and particle size selection on the feeding activity and scope for growth of Ruditapes decussatus from Ria Formosa, southern Portugal. J Exp Mar Biol Ecol 245:111–125. https://doi.org/10.1016/S0022-0981(99)00154-9
Utting SD, Millican PF (1997) Techniques for the hatchery conditioning of bivalve broodstocks and the subsequent effect on egg quality and larval viability. Aquaculture 155:45–54. https://doi.org/10.1016/S0044-8486(97)00108-7
Walne PR (1976) Experiments on the culture in the sea of the butterfish Venerupis decussata L. Aquaculture 8:371–381. https://doi.org/10.1016/0044-8486(76)90119-8
Acknowledgments
The authors would like to thank the Science and Technology Development fund (STDF), Egypt, for funding the current research through the US-Egypt project # 1971 entitled “Biomarker Assisted Selection for Disease-Resistance in Clams for Establishing Egyptian and Improving American Bivalve Aquaculture”. Special thanks to Prof. Somaya Mahfouz, Head of Invertebrates Laboratory, Aquaculture Division, for her continuous encouragement and support throughout the project duration.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abbas, A.S.A., El-Wazzan, E., Khafage, A.R. et al. Influence of different microalgal diets on gonadal development of the carpet shell clam Ruditapes decussatus broodstock. Aquacult Int 26, 1297–1309 (2018). https://doi.org/10.1007/s10499-018-0284-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-018-0284-9