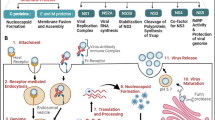
Abstract
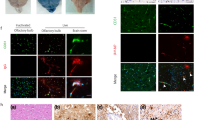
The extent of neuronal cell damage caused by West Nile virus (WNV) infection governs the disease severity ranging from mild, febrile illness to fatal encephalitis. Availability of naturally occurring genetic variants is helpful to study viral factors governing differential pathogenesis. During WNV infection, apoptosis serves as a virulence determinant positively contributing to viral pathogenesis. We investigated the levels of apoptosis induced by a low neurovirulent WNV lineage 5 strain 804994 and a high neurovirulent lineage 1 strain 68856 in human neuroblastoma cells, IMR-32. Our investigations clearly show the correlation between higher multiplication capacities of 68856 with higher levels of cytopathology induced by apoptosis. We observed activation of both the extrinsic and intrinsic apoptotic pathways during WNV infection. Infection with higher neurovirulent strain resulted in higher upregulation of pro-apoptotic proteins including death receptors (DR), adaptor protein, BH3-only regulatory proteins and higher cleavage of initiator caspases of both pathways. These results suggest that the virulence of a WNV strain may correlate with its higher replication fitness and ability to cause more cellular damage.














Similar content being viewed by others
Data availability
The data generated during and analysed during this study is included in the supplementary file, which will be available from the corresponding author on reasonable request.
References
Chancey C, Grinev A, Volkova E, Rios M (2015) The global ecology and epidemiology of west nile virus. BioMed Res Int. https://doi.org/10.1155/2015/376230
Ronca SE, Murray KO, Nolan MS (2019) Cumulative incidence of West Nile virus infection, continental United States, 1999–2016. Emerg Infect Dis 25(2):325–327. https://doi.org/10.3201/eid2502.180765
Statistics & Maps. (2022, October 14). Centers for Disease Control and Prevention [CDC]. Accessed September 9th, 2022, from https://www.cdc.gov/westnile/statsmaps/index.html
Fall G, Di Paola N, Faye M, Dia M, de Freire CCM, Loucoubar C, de Zanotto PMA, Faye O, Sall AA (2017) Biological and phylogenetic characteristics of West African lineages of West Nile virus. PLoS Negl Trop Dis 11(11):1–23. https://doi.org/10.1371/journal.pntd.0006078
Platonov AE, Karan LS, Shopenskaia TA, Fedorova MV, Koliasnikova NM, Rusakova NM, Shishkina LV, Arshba TE, Zhuravlev VI, Govorukhina MV, Valentseva AA, Shipulin GA (2011) Genotyping of West Nile fever virus strains circulating in southern Russia as an epidemiological investigation method: principles and results. Zhurnal Mikrobiologii, Epidemiologii, i Immunobiologii 2:29–37
Bakonyi T, Ferenczi E, Erdélyi K, Kutasi O, Csörgo T, Seidel B, Weissenböck H, Brugger K, Bán E, Nowotny N (2013) Explosive spread of a neuroinvasive lineage 2 West Nile virus in Central Europe, 2008/2009. Vet Microbiol 165(1–2):61–70. https://doi.org/10.1016/j.vetmic.2013.03.005
Bondre VP, Jadi RS, Mishra AC, Yergolkar PN, Arankalle VA (2007) West Nile virus isolates from India: evidence for a distinct genetic lineage. J Gen Virol 88(3):875–884. https://doi.org/10.1099/vir.0.82403-0
Chowdhury P, Khan SA, Dutta P, Topno R, Mahanta J (2014) Characterization of West Nile virus (WNV) isolates from Assam, India: Insights into the circulating WNV in northeastern India. Comp Immunol Microbiol Infect Dis 37(1):39–47. https://doi.org/10.1016/j.cimid.2013.10.006
Frost MJ, Zhang J, Edmonds JH, Prow NA, Gu X, Davis R, Hornitzky C, Arzey KE, Finlaison D, Hick P, Read A, Hobson-Peters J, May FJ, Doggett SL, Haniotis J, Russell RC, Hall RA, Khromykh AA, Kirkland PD (2012) Characterization of virulent West Nile virus Kunjin strain, Australia 2011. Emerging Infect Dis. https://doi.org/10.3201/eid1805.111720
Smithburn KC, Kerr JA, Gatne PB (1954) Neutralizing antibodies against certain viruses in the sera of residents of India. J Immunol 72(4):248LP – 257
Paramasivan SR, Mishra AC, Mourya DT (2003) West Nile virus: The Indian scenario. Indian J Med Res 118:101–108
Khatun T, Chatterjee S (2017) Emergence of West Nile virus in West Bengal, India: a new report. Trans R Soc Trop Med Hyg 111(4):178–184. https://doi.org/10.1093/trstmh/trx033
Tandel K, Sharma S, Dash PK, Shukla J, Parida M (2019) Emergence of human West Nile Virus infection among pediatric population in Madhya Pradesh, India. J Med Virol 91(3):493–497. https://doi.org/10.1002/jmv.25325
Anukumar B, Sapkal GN, Tandale BV, Balasubramanian R, Gangale D (2014) West Nile encephalitis outbreak in Kerala, India, 2011. J Clin Virol 61(1):152–155. https://doi.org/10.1016/j.jcv.2014.06.003
Fiacre L, Pagès N, Albina E, Richardson J, Lecollinet S, Gonzalez G (2020) Molecular determinants of west Nile virus virulence and pathogenesis in vertebrate and invertebrate hosts. Int J Mol Sci 21(23):1–35. https://doi.org/10.3390/ijms21239117
Donadieu E, Lowenski S, Servely JL, Laloy E, Lilin T, Nowotny N, Richardson J, Zientara S, Lecollinet S, Coulpier M (2013) Comparison of the neuropathology induced by two West Nile virus strains. PLoS ONE. https://doi.org/10.1371/journal.pone.0084473
Worwa G, Wheeler SS, Brault AC, Reisen WK (2015) Comparing competitive fitness of West Nile virus strains in avian and mosquito hosts. PLoS ONE. https://doi.org/10.1371/journal.pone.0125668
Gamino V, Pérez-Ramírez E, Gutiérrez-Guzmán AV, Sotelo E, Llorente F, Jiménez-Clavero MÁ, Höfle U (2021) Pathogenesis of two western mediterranean west nile virus lineage 1 isolates in experimentally infected red-legged partridges (Alectoris rufa). Pathogens. https://doi.org/10.3390/pathogens10060748
Beasley DWC, Li L, Suderman MT, Barrett ADT (2002) Mouse neuroinvasive phenotype of West Nile virus strains varies depending upon virus genotype. Virology 296(1):17–23. https://doi.org/10.1006/viro.2002.1372
Barzon L, Pacenti M, Montarsi F, Fornasiero D, Gobbo F, Quaranta E, Monne I, Fusaro A, Volpe A, Sinigaglia A, Riccetti S, Molin ED, Satto S, Lisi V, Gobbi F, Galante S, Feltrin G, Valeriano V, Favero L, Russo F, Capelli G (2022) Rapid spread of a new West Nile virus lineage 1 associated with increased risk of neuroinvasive disease during a large outbreak in northern Italy, 2022: One Health analysis. J Travel Med. https://doi.org/10.1093/jtm/taac125
Swanson PA, McGavern DB (2015) Viral diseases of the central nervous system. Curr Opin Virol 11:44–54. https://doi.org/10.1016/j.coviro.2014.12.009
Shrestha B, Gottlieb D, Diamond MS (2003) Infection and injury of neurons by West NileEncephalitisVirus. J Virol 77(24):13203–13213. https://doi.org/10.1128/jvi.77.24.13203-13213.2003
Guarner J, Shieh WJ, Hunter S, Paddock CD, Morken T, Campbell GL, Marfin AA, Zaki SR (2004) Clinicopathologic study and laboratory diagnosis of 23 cases with West Nile virus encephalomyelitis. Hum Pathol 35(8):983–990. https://doi.org/10.1016/j.humpath.2004.04.008
Cheeran MC-J, Hu S, Sheng WS, Rashid A, Peterson PK, Lokensgard JR (2005) Differential responses of human brain cells to West Nile virus infection. J Neurovirol 11(6):512–524. https://doi.org/10.1080/13550280500384982
Kumar M, Verma S, Nerurkar VR (2010) Pro-inflammatory cytokines derived from West Nile virus (WNV)-infected SK-N-SH cells mediate neuroinflammatory markers and neuronal death. J Neuroinflammation 7(1):73. https://doi.org/10.1186/1742-2094-7-73
Kleinschmidt MC, Michaelis M, Ogbomo H, Doerr HW, Cinatl J (2007) Inhibition of apoptosis prevents West Nile virus induced cell death. BMC Microbiol 7:1–8. https://doi.org/10.1186/1471-2180-7-49
Ghosh Roy S, Datan E, Sadigh B, Lockshin RA, Zakeri Z (2014) Regulation of cell survival and death during Flavivirus infections. World J Biol Chem 5(2):93–105. https://doi.org/10.4331/wjbc.v5.i2.93
Samuel MA, Morrey JD, Diamond MS (2007) Caspase 3-dependent cell death of neurons contributes to the pathogenesis of West Nile virus encephalitis. J Virol 81(6):2614–2623. https://doi.org/10.1128/jvi.02311-06
Peng BH, Wang T (2019) West Nile virus induced cell death in the central nervous system. Pathogens 8(4):215. https://doi.org/10.3390/pathogens8040215
Clarke P, Tyler KL (2009) Apoptosis in animal models of virus-induced disease. Nat Rev Microbiol 7(2):144–155. https://doi.org/10.1038/nrmicro2071
Baer A, Kehn-Hall K (2014) Viral concentration determination through plaque assays: using traditional and novel overlay systems. J Vis Exp 93:1–10. https://doi.org/10.3791/52065
Pavitrakar DV, Ayachit VM, Mundhra S, Bondre VP (2015) Development and characterization of reverse genetics system for the Indian West Nile virus lineage 1 strain 68856. J Virol Methods 226:31–39. https://doi.org/10.1016/j.jviromet.2015.09.008
Kamiloglu S, Sari G, Ozdal T, Capanoglu E (2020) Guidelines for cell viability assays. Food Frontiers 1(3):332–349. https://doi.org/10.1002/fft2.44
Ribble D, Goldstein NB, Norris DA, Shellman YG (2005) A simple technique for quantifying apoptosis in 96-well plates. BMC Biotechnol. https://doi.org/10.1186/1472-6750-5-12
Okamoto T, Suzuki T, Kusakabe S, Tokunaga M, Hirano J, Miyata Y, Matsuura Y (2017) Regulation of apoptosis during flavivirus infection. Viruses. https://doi.org/10.3390/v9090243
Mazar J, Li Y, Rosado A, Phelan P, Kedarinath K, Parks GD, Alexander KA, Westmoreland TJ (2018) Zika virus as an oncolytic treatment of human neuroblastoma cells requires CD24. PLoS ONE. https://doi.org/10.1371/journal.pone.0200358
Brault AC, Huang CYH, Langevin SA, Kinney RM, Bowen RA, Ramey WN, Panella NA, Holmes EC, Powers AM, Miller BR (2007) A single positively selected West Nile viral mutation confers increased virogenesis in American crows. Nat Gene 39(9):1162–1166
Moudy RM, Zhang B, Shi PY, Kramer LD (2009) West Nile virus envelope protein glycosylation is required for efficient viral transmission by Culex vectors. Virology 387(1):222–228. https://doi.org/10.1016/j.virol.2009.01.038
del Carmen Parquet M, Kumatori A, Hasebe F, Morita K, Igarashi A (2001) West Nile virus-induced bax-dependent apoptosis. FEBS Lett 500(1–2):17–24. https://doi.org/10.1016/S0014-5793(01)02573-X
Vaidyanathan R, Scott TW (2006) Apoptosis in mosquito midgut epithelia associated with West Nile virus infection. Apoptosis 11(9):1643–1651. https://doi.org/10.1007/s10495-006-8783-y
Elmore S (2007) Apoptosis: A review of programmed cell death. In Toxicologic Pathology. https://doi.org/10.1080/01926230701320337
Martinez MM, Reif RD, Pappas D (2010) Detection of apoptosis: a review of conventional and novel techniques. Anal Method 2(8):996–1004. https://doi.org/10.1039/C0AY00247J
Sánchez-Osuna M, Garcia-Belinchón M, Iglesias-Guimarais V, Gil-Guinón E, Casañelles E, Yuste VJ (2014) Caspase-activated DNase is necessary and sufficient for oligonucleosomal DNA breakdown, but not for chromatin disassembly during caspase-dependent apoptosis of LN-18 glioblastoma cells. J Biol Chem 289(27):18752–18769. https://doi.org/10.1074/jbc.M114.550020
Chu JJH, Ng ML (2003) The mechanism of cell death during West Nile virus infection is dependent on initial infectious dose. J Gen Virol 84(12):3305–3314. https://doi.org/10.1099/vir.0.19447-0
Munoz-Erazo, L., Natoli, R., Provis, J. M., Madigan, M. C., & King, N. J. C. (2012). Microarray analysis of gene expression in West Nile virus-infected human retinal pigment epithelium. In Molecular Vision (Vol. 18). http://www.molvis.org/molvis/v18/a78
Shrestha B, Diamond MS (2007) Fas ligand interactions contribute to CD8 + T-cell-mediated control of West Nile virus infection in the central nervous system. J Virol 81(21):11749–11757. https://doi.org/10.1128/jvi.01136-07
Clarke P, Leser JS, Quick ED, Dionne KR, Beckham JD, Tyler KL (2014) Death receptor-mediated apoptotic signaling is activated in the brain following infection with West Nile virus in the absence of a peripheral immune response. J Virol 88(2):1080–1089. https://doi.org/10.1128/jvi.02944-13
Venter M, Myers TG, Wilson MA, Kindt TJ, Paweska JT, Burt FJ, Leman PA, Swanepoel R (2005) Gene expression in mice infected with West Nile virus strains of different neurovirulence. Virology 342(1):119–140. https://doi.org/10.1016/j.virol.2005.07.013
Kumar M, Belcaid M, Nerurkar VR (2016) Identification of host genes leading to West Nile virus encephalitis in mice brain using RNA-seq analysis. Sci Rep 6(March):1–14. https://doi.org/10.1038/srep26350
Medigeshi GR, Lancaster AM, Hirsch AJ, Briese T, Lipkin WI, DeFilippis V, Früh K, Mason PW, Nikolich-Zugich J, Nelson JA (2007) West Nile Virus infection activates the unfolded protein response, leading to CHOP induction and apoptosis. J Virol 81(20):10849–10860. https://doi.org/10.1128/jvi.01151-07
Acknowledgements
Authors are thankful to the Director, ICMR-NIV, Pune for continuous support and critical evaluation of the work. We are grateful to the technical and scientific staff of the Encephalitis Group for providing active support and critique during the work. We are also thankful to the Director General, ICMR and CSIR, New Delhi, India and for financial support (project ID: ENC1304).
Funding
Research work was funded by Indian Council of Medical Research (ICMR, NIV ID: ENC1304).
Author information
Authors and Affiliations
Contributions
SM performed all the experiments, data analysis and manuscript writing; VPB conceived concept, planning and supervising of experiments, data analysis and manuscript editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors state that there is no conflict of interest.
Ethical approval
Study was approved by the Institutional Biosafety Committee, ICMR-NIV, Pune (IBSC:2013–39). This study does not involve any animal experiments while the standard antiviral sera was procured from Virus Registry, ICMR-NIV, Pune.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mundhra, S., Bondre, V.P. Higher replication potential of West Nile virus governs apoptosis induction in human neuroblastoma cells. Apoptosis 28, 1113–1127 (2023). https://doi.org/10.1007/s10495-023-01844-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-023-01844-2