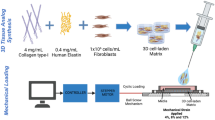
Abstract
Ligament wound healing involves the proliferation of a dense and disorganized fibrous matrix that slowly remodels into scar tissue at the injury site. This remodeling process does not fully restore the highly aligned collagen network that exists in native tissue, and consequently repaired ligament has decreased strength and durability. In order to identify treatments that stimulate collagen alignment and strengthen ligament repair, there is a need to develop in vitro models to study fibroblast activation during ligament wound healing. The objective of this study was to measure gene expression and matrix protein accumulation in fibroblast-collagen gels that were subjected to different static stress conditions (stress-free, biaxial stress, and uniaxial stress) for three time points (1, 2 or 3 weeks). By comparing our in vitro results to prior in vivo studies, we found that stress-free gels had time-dependent changes in gene expression (col3a1, TnC) corresponding to early scar formation, and biaxial stress gels had protein levels (collagen type III, decorin) corresponding to early scar formation. This is the first study to conduct a targeted evaluation of ligament healing biomarkers in fibroblast-collagen gels, and the results suggest that biomimetic in-vitro models of early scar formation should be initially cultured under biaxial stress conditions.








Similar content being viewed by others
References
Akhouayri, O., M.-H. Lafage-Proust, A. Rattner, N. Laroche, A. Caillot-Augusseau, C. Alexandre, and L. Vico. Effects of static or dynamic mechanical stresses on osteoblast phenotype expression in three-dimensional contractile collagen gels. J. Cell. Biochem. 76:217–230, 2000.
Bellows, C. G., A. H. Melcher, and J. E. Aubin. Contraction and organization of collagen gels by cells cultured from periodontal ligament, gingiva and bone suggest functional differences between cell types. J. Cell Sci. 50:299–314, 1981.
Bloemen, V., T. Schoenmaker, T. J. De Vries, and V. Everts. Direct cell-cell contact between periodontal ligament fibroblasts and osteoclast precursors synergistically increases the expression of genes related to osteoclastogenesis. J. Cell. Physiol. 222:565–573, 2010.
Boykiw, R., P. Sciore, C. Reno, L. Marchuk, C. B. Frank, and D. A. Hart. Altered levels of extracellular matrix molecule mRNA in healing rabbit ligaments. Matrix Biol. 17:371–378, 1998.
Brune, T., A. Borel, T. W. Gilbert, J. P. Franceschi, S. F. Badylak, and P. Sommer. In vitro comparison of human fibroblasts from intact and ruptured ACL for use in tissue engineering. Eur. Cell. Mater. 14:71–78, 2007.
Burgess, M. L., W. E. Carver, L. Terracio, S. P. Wilson, M. A. Wilson, and T. K. Borg. Integrin-Mediated Collagen Gel Contraction by Cardiac Fibroblasts Effects of Angiotensin II. Circ. Res. 74:291–298, 1994.
Canović, E. P., D. T. Seidl, S. R. Polio, A. A. Oberai, P. E. Barbone, D. Stamenović, and M. L. Smith. Biomechanical imaging of cell stiffness and prestress with subcellular resolution. Biomech. Model. Mechanobiol. 13:665–678, 2014.
Chamberlain, C. S., E. M. Crowley, H. Kobayashi, K. W. Eliceiri, and R. Vanderby. Quantification of collagen organization and extracellular matrix factors within the healing ligament. Microanal. Microsc. 17:779–787, 2011.
Chamberlain, C. S., E. Crowley, and R. Vanderby. The spatio-temporal dynamics of ligament healing. Wound Repair Regen. 17:206–215, 2009.
Chen, L. H., M. Warner, L. Fingerhut, and D. Makuc. Injury episodes and circumstances: National Health Interview Survey, 1997–2007. Vital Health Stat. 10. 1–55, 2009.
Chevallay, B., N. Abdul-Malak, and D. Herbage. Mouse fibroblasts in long-term culture within collagen three-dimensional scaffolds: Influence of crosslinking with diphenylphosphoryl azide on matrix reorganization, growth, and biosynthetic and proteolytic activities. J. Biomed. Mater. Res. 49:448–459, 2000.
Chiquet-Ehrisrnann, R., M. Tarmheimer, M. Koch, A. Brunner, J. Spring, D. Martin, S. Baumgartner, and M. Chiquet. Tenascin-C expression by fibroblasts is elevated in stressed collagen gels. J. Cell Biol. 127:2093–2101, 1994.
Chockalingam, P. S., S. S. Glasson, and L. S. Lohmander. Tenascin-C levels in synovial fluid are elevated after injury to the human and canine joint and correlate with markers of inflammation and matrix degradation. Osteoarthr. Cartil. 21:339–345, 2013.
Clements, D. N., S. D. Carter, J. F. Innes, W. E. R. Ollier, and P. J. R. Day. Gene expression profiling of normal and ruptured canine anterior cruciate ligaments. Osteoarthr. Cartil. 16:195–203, 2008.
Fernández, P., P. A. Pullarkat, and A. Ott. A master relation defines the nonlinear viscoelasticity of single fibroblasts. Biophys. J. 90:3796–3805, 2006.
Foolen, J., S. L. Wunderli, S. Loerakker, J. G. Snedeker. Tissue alignment enhances remodeling potential of tendon-derived cells - Lessons from a novel microtissue model of tendon scarring. Matrix Biol. 65:14–29, 2018.
Frank, C. B., D. A. Hart, and N. G. Shrive. Molecular biology and biomechanics of normal and healing ligaments—a review. Osteoarthr. Cartil. 7:130–140, 1999.
Frank, C., S. L. Y. Woo, D. Amiel, F. Harwood, M. Gomez, and W. Akeson. Medial collateral ligament healing: a multidisciplinary assessment in rabbits. Am. J. Sports Med. 11:379–389, 1983.
Gentleman, E., G. A. Livesay, K. C. Dee, and E. A. Nauman. Development of ligament-like structural organization and properties in cell-seeded collagen scaffolds in vitro. Ann. Biomed. Eng. 34:726–736, 2006.
Gomez, M. A., S. L. Woo, M. Inoue, D. Amiel, F. L. Harwood, and L. Kitabayashi. Medial collateral ligament healing subsequent to different treatment regimens. J. Appl. Physiol. 66:245–252, 1989.
Hagerty, P., A. Lee, S. Calve, C. A. Lee, M. Vidal, and K. Baar. The effect of growth factors on both collagen synthesis and tensile strength of engineered human ligaments. Biomaterials 33:6355–6361, 2012.
Haslauer, C. M., B. L. Proffen, V. M. Johnson, and M. M. Murray. Expression of modulators of extracellular matrix structure after anterior cruciate ligament injury. Wound Repair Regen. 22:103–110, 2014.
Hauser, R. A. Ligament injury and healing: a review of current clinical diagnostics and therapeutics. Open Rehabil. J. 6:1–20, 2013.
Henshaw, D. R., E. Attia, M. Bhargava, and J. A. Hannafin. Canine ACL fibroblast integrin expression and cell alignment in response to cyclic tensile strain in three-dimensional collagen gels. J. Orthop. Res. 24:481–490, 2006.
Jensen, E. C. Quantitative analysis of histological staining and fluorescence using ImageJ. Anat. Rec. 296:378–381, 2013.
Jhun, C.-S., M. C. Evans, V. H. Barocas, and R. T. Tranquillo. Planar biaxial mechanical behavior of bioartificial tissues possessing prescribed fiber alignment. J. Biomech. Eng. 131:81006, 2009.
Junqueira, L. C. U., G. Bignolas, and R. R. Brentani. Picrosirius staining plus polarization microscopy, a specific method for collagen detection in tissue sections. Histochem. J. 11:447–455, 1979.
Kharaz, Y. A., S. R. Tew, M. Peffers, E. G. Canty-Laird, and E. Comerford. Proteomic differences between native and tissue-engineered tendon and ligament. Proteomics 16:1547–1556, 2016.
Loghmani, M. T., and S. J. Warden. Instrument-assisted cross-fiber massage accelerates knee ligament healing. J. Orthop. Sport. Phys. Ther. 39:506–514, 2009.
Martinez, D. A., A. C. Vailas, R. Vanderby, and R. E. Grindeland. Temporal extracellular matrix adaptations in ligament during wound healing and hindlimb unloading. Am. J. Physiol. Integr. Comp. Physiol. 293:R1552–R1560, 2007.
Monici, M. Cell and tissue autofluorescence research and diagnostic applications. Biotechnol. Annu. Rev. 11:227–256, 2005.
Morrill, E. E., A. N. Tulepbergenov, C. J. Stender, R. Lamichhane, R. J. Brown, and T. J. Lujan. A validated software application to measure fiber organization in soft tissue. Biomech. Model. Mechanobiol. 15:1467–1478, 2016.
Murphy, P. G., B. J. Loitz, C. B. Frank, and D. A. Hart. Influence of exogenous growth factors on the synthesis and secretion of collagen types I and III by explants of expression of normal and healing rabbit ligaments. Biochem. Cell Biol. 72:403–409, 1994.
Nguyen, D. T., T. H. Ramwadhdoebe, C. P. Van Der Hart, L. Blankevoort, P. P. Tak, and C. N. Van Dijk. Intrinsic healing response of the human anterior cruciate ligament: an histological study of reattached ACL remnants. J. Orthop. Res. 32:296–301, 2014.
Niyibizi, C., K. Kavalkovich, T. Yamaji, and S. L. Woo. Type V collagen is increased during rabbit medial collateral ligament healing. Knee Surg. Sports Traumatol. Arthrosc. 8:281–285, 2000.
Nöth, U., K. Schupp, A. Heymer, S. Kall, F. Jakob, N. Schütze, B. Baumann, T. Barthel, J. Eulert, and C. Hendrich. Anterior cruciate ligament constructs fabricated from human mesenchymal stem cells in a collagen type I hydrogel. Cytotherapy 7:447–455, 2005.
Pascher, A., A. F. Steinert, G. D. Palmer, O. Betz, J.-N. Gouze, E. Gouze, C. Pilapil, S. C. Ghivizzani, C. H. Evans, and M. M. Murray. Enhanced repair of the anterior cruciate ligament by in situ gene transfer: evaluation in an in vitro model. Mol. Ther. 10:327–336, 2004.
Plaas, A. H., S. Wong-Palms, T. Koob, D. Hernandez, L. Marchuk, and C. B. Frank. Proteoglycan metabolism during repair of the ruptured medial collateral ligament in skeletally mature rabbits. Arch. Biochem. Biophys. 374:35–41, 2000.
Pu, X., and J. T. Oxford. Proteomic analysis of engineered cartilage. Methods Mol. Biol. 1340:263–278, 2015.
Saddiq, Z. A., J. C. Barbenel, and M. H. Grant. The mechanical strength of collagen gels containing glycosaminoglycans and populated with fibroblasts. J. Biomed. Mater. Res. Part A 89:697–706, 2009.
Schain, A. J., R. A. Hill, and J. Grutzendler. Label-free in vivo imaging of myelinated axons in health and disease with spectral confocal reflectance microscopy. Nat. Med. 20:443–449, 2014.
Schreck, P. J., L. R. Kitabayashi, D. Amiel, W. H. Akeson, and V. L. Woods. Integrin display increases in the wounded rabbit medial collateral ligament but not the wounded anterior cruciate ligament. J. Orthop. Res. 13:174–183, 1995.
Singer, A. J., and R. A. Clark. Cutaneous wound healing. N. Engl. J. Med. 341:738–746, 1999.
Stender, C. J., E. Rust, P. T. Martin, E. E. Neumann, R. J. Brown, and T. J. Lujan. Modeling the effect of collagen fibril alignment on ligament mechanical behavior. Biomech. Model. Mechanobiol. 17:543–557, 2018.
Stoll, C., T. John, M. Endres, C. Rosen, C. Kaps, B. Kohl, M. Sittinger, W. Ertel, and G. Schulze-Tanzil. Extracellular matrix expression of human tenocytes in three-dimensional air-liquid and PLGA cultures compared with tendon tissue: implications for tendon tissue engineering. J. Orthop. Res. 28:1170–1177, 2010.
van Rijn, R. M., A. G. van Os, R. M. D. Bernsen, P. A. Luijsterburg, B. W. Koes, and S. M. A. Bierma-Zeinstra. What is the clinical course of acute ankle sprains? A systematic literature review. Am. J. Med. 121(324–331):e7, 2008.
Acknowledgments
Authors wish to thank John Everingham for designing and assembling the cruciform, Laura Bond for statistical analysis, and Peter Martin for analysis of Alcian blue stained images. Authors acknowledge support by the Institutional Development Award (IDeA) Program from the National Institute of General Medical Sciences of the National Institutes of Health under Grants #P20GM103408 and P20GM109095. We also acknowledge support from The Biomolecular Research Center at Boise State with funding from the National Science Foundation, Grants #0619793 and #0923535; the MJ Murdock Charitable Trust; Lori and Duane Steuckle, and the Idaho State Board of Education. Authors have no competing financial interests.
Conflict of interest
Authors have no competing financial interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Jennifer West oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Frahs, S.M., Oxford, J.T., Neumann, E.E. et al. Extracellular Matrix Expression and Production in Fibroblast-Collagen Gels: Towards an In Vitro Model for Ligament Wound Healing. Ann Biomed Eng 46, 1882–1895 (2018). https://doi.org/10.1007/s10439-018-2064-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-018-2064-0