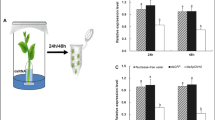
Abstract
Aphids are a major group of crop pests, which directly feed on phloem and indirectly transmit plant virus. The entomopathogenic fungi are essential members of green pest control strategies, and insecticidal RNAi is considered as a new generation of biopesticides. However, the effects of combining entomopathogenic fungus and RNAi-based biopesticides for aphid control are unknown. Here, the responsive genes of Acyrthosiphon pisum were screened from the transcriptomes upon spraying of Beauveria bassiana Bb07 strain (hereafter referred to Bb07). Subsequently, six candidate genes, Ap4714, Ap15782, Ap20844, Ap14741, Ap15105 and Ap16968, were targeted by spray-based RNAi, which produced a range of 20.0, 58.0, 44.0, 27.0, 52.0 and 24.0% gene silencing, respectively. Silencing of single-target (Ap4714, Ap15782, and Ap20844) resulted in increased virulence of Bb07 to Ac. pisum. Importantly, the mortality was increased to 82.1, 76.7, and 60.7% within 7 days to Ac. pisum when double-targets (Ap4714 + Ap15782, Ap4714 + Ap20844 and Ap15782 + Ap20844) were silenced in together with application of Bb07, respectively. This high mortality was also observed in Myzus persicae and Aphis citricidus by co-spray of the combination of dsAp4714 + dsAp15782 together with Bb07. In together, this study demonstrates the potential of a combination of RNAi- and entomopathogenic fungus-based technologies to control aphids, thereby reducing application of chemical pesticides.






Similar content being viewed by others
References
Alavo TBC, Sermann H, Bochow H (2002) Virulence of strains of the entomopathogenic fungus Verticillium lecaniito aphids: strain improvement. Arch Phytopath Pflanz 34:379–398. https://doi.org/10.1080/716061669
Amobonye A, Bhagwat P, Pandey A, Singh S, Pillai S (2020) Biotechnological potential of Beauveria bassiana as a source of novel biocatalysts and metabolites. Crit Rev Biotechnol 40:1019–1034. https://doi.org/10.1080/07388551.2020.1805403
Bass C, Puinean AM, Zimmer CT et al (2014) The evolution of insecticide resistance in the peach potato aphid, Myzus persicae. Insect Biochem Molec 51:41–51. https://doi.org/10.1016/j.ibmb.2014.05.003
Ding BY, Niu JZ, Shang F, Yang L, Zhang W, Smagghe G, Wang JJ (2020) Parental silencing of a horizontally transferred carotenoid desaturase gene causes a reduction of red pigment and fitness in the pea aphid. Pest Manag Sci 76:2423–2433. https://doi.org/10.1002/ps.5783
Feng MG, Johnson JB, Kish LP (1990) Virulence of Verticillium lecanii and an aphid-derived isolate of Beauveria bassiana (Fungi: Hyphomycetes) for six species of cereal-infesting aphids (Homoptera: Aphididae). Environ Entomol 19:815–820. https://doi.org/10.1093/ee/19.3.815
Florea L, Song L, Salzberg SL (2013) Thousands of exon skipping events differentiate among splicing patterns in sixteen human tissues Fes1000Res 2:188 https://doi.org/10.12688/f1000research.2-188.v2
Guo M, Nanda S, Chen S et al (2021) Oral RNAi toxicity assay suggests clathrin heavy chain as a promising molecular target for controlling the 28‐spotted potato ladybird, Henosepilachna vigintioctopunctata Pest Manage Sci in press https://doi.org/10.1002/ps.6594
Hajek AE, Butler L (2000) Predicting the host range of entomopathogenic fungi. In: Follett PA, Duan JJ (eds) Nontarget effects of biological control Springer US, Boston, MA, pp:263–276 https://doi.org/10.1007/978-1-4615-4577-4_15
Hou CX, Qin GX, Liu T, Geng T, Gao K, Pan ZH, Qian HY, Guo XJ (2014) Transcriptome analysis of silkworm, Bombyx mori, during early response to Beauveria bassiana challenges. PLoS ONE 9:e91189. https://doi.org/10.1371/journal.pone.0091189
Hou QL, Xu LJ, Liu GY et al (2019) Plant-mediated gene silencing of an essential olfactory-related Gqα gene enhances resistance to grain aphid in common wheat in greenhouse and field. Pest Manag Sci 75:1718–1725. https://doi.org/10.1002/ps.5292
Hu J, Xia YX (2019) Increased virulence in the locust-specific fungal pathogen Metarhizium acridum expressing dsRNAs targeting the host F1F0-ATPase subunit genes. Pest Manag Sci 75:180–186. https://doi.org/10.1002/ps.5085
Kanost MR, Jiang HB (2015) Clip-domain serine proteases as immune factors in insect hemolymph. Curr Opin Insect Sci 11:47–55. https://doi.org/10.1016/j.cois.2015.09.003
Kary NE, Alizadeh Z (2017) Effects of sub-culturing on genetic and physiological parameters in different Beauveria bassiana isolates. J Invertebr Pathol 145:62–67. https://doi.org/10.1016/j.jip.2017.03.008
Laity JH, Lee BM, Wright PE (2001) Zinc finger proteins: new insights into structural and functional diversity. Curr Opin Struc Biol 11:39–46. https://doi.org/10.1016/S0959-440X(00)00167-6
Lee JB, Kim HS, Park YJ (2017) Down-regulation of a chitin synthase a gene by RNA interference enhances pathogenicity of Beauveria bassiana ANU1 against Spodoptera exigua (HÜBNER). Arch Insect Biochem 94:e21371. https://doi.org/10.1002/arch.21371
Lee WW, Shin TY, Bae SM, Woo SD (2015) Screening and evaluation of entomopathogenic fungi against the green peach aphid, Myzus persicae, using multiple tools. J Asia-Pac Entomol 18:607–615. https://doi.org/10.1016/j.aspen.2015.07.012
Lopez DC, Zhu-Salzman K, Ek-Ramos MJ, Sword GA (2014) The entomopathogenic fungal endophytes Purpureocillium lilacinum (formerly Paecilomyces lilacinus) and Beauveria bassiana negatively affect cotton aphid reproduction under both greenhouse and field conditions. PLoS ONE 9:e103891. https://doi.org/10.1371/journal.pone.0103891
Lu HL, St Leger RJ (2016) Insect immunity to entomopathogenic fungi. Adv Genet 94:251–285. https://doi.org/10.1016/bs.adgen.2015.11.002
Ma L, Chen F, Wang W, Xu L, Lu ZQ (2020) Identification of two clip domain serine proteases involved in the pea aphid’s defense against bacterial and fungal infection. Insect Sci 27:735–744. https://doi.org/10.1111/1744-7917.12673
Mitter N, Worrall EA, Robinson KE et al (2017) Clay nanosheets for topical delivery of RNAi for sustained protection against plant viruses. Nat Plants 3:16207. https://doi.org/10.1038/nplants.2016.207
Naessens E, Dubreuil G, Giordanengo P, Baron OL, Minet-Kebdani N, Keller H, Coustau C (2015) A secreted MIF cytokine enables aphid feeding and represses plant immune responses. Curr Biol 25:1898–1903. https://doi.org/10.1016/j.cub.2015.05.047
Nault LR (1997) Arthropod transmission of plant viruses: a new synthesis. Ann Entomol Soc Am 90:521–541. https://doi.org/10.1093/aesa/90.5.521
Nazir T, Basit A, Hanan A, Majeed MZ, Qiu D (2018) In vitro pathogenicity of some entomopathogenic fungal strains against green peach aphid Myzus persicae (Homoptera: Aphididae). Agronomy-Basel 9:7. https://doi.org/10.3390/agronomy9010007
Niu J, Taning CNT, Christiaens O, Smagghe G, Wang JJ (2018) Rethink RNAi in insect pest control: challenges and perspectives. Adv Insect Physiol 55:1–17. https://doi.org/10.1016/bs.aiip.2018.07.003
Niu JZ, Yang WJ, Tian Y et al (2019) Topical dsRNA delivery induces gene silencing and Mortality in the pea aphid. Pest Manag Sci 75:2873–2881. https://doi.org/10.1002/ps.5457
Pan H, Yang X, Romeis J, Siegfried BD, Zhou X (2020) Dietary RNAi toxicity assay exhibits differential responses to ingested dsRNAs among lady beetles. Pest Manag Sci 76:3606–3614. https://doi.org/10.1002/ps.5894
Peng GX, Jin K, Liu YC, Xia YX (2015) Enhancing the utilization of host trehalose by fungal trehalase improves the virulence of fungal insecticide. Appl Microbiol Biot 99:8611–8618. https://doi.org/10.1007/s00253-015-6767-y
Qu S, Wang SB (2018) Interaction of entomopathogenic fungi with the host immune system. Dev Comp Immunol 83:96–103. https://doi.org/10.1016/j.dci.2018.01.010
Rakhra G, Rakhra G (2021) Zinc finger proteins: insights into the transcriptional and post transcriptional regulation of immune response. Mol Biol Rep 48:5735–5743. https://doi.org/10.1007/s11033-021-06556-x
Rispe C, Kutsukake M, Doublet V, Hudaverdian S, Legeai F, Simon CJ, Tagu D, Fukatsu T (2008) Large gene family expansion and variable selective pressures for cathepsin B in aphids. Mol Biol Evol 25:5–17. https://doi.org/10.1093/molbev/msm222
Shang F, Ding BY, Ye C et al (2020) Evaluation of a cuticle protein gene as a potential RNAi target in aphids. Pest Manag Sci 76:134–140. https://doi.org/10.1002/ps.5599
Simon JC, Peccoud J (2018) Rapid evolution of aphid pests in agricultural environments. Curr Opin Insect Sci 26:17–24. https://doi.org/10.1016/j.cois.2017.12.009
Singh S, Bhatnagar S, Choudhary S, Nirwan B, Sharma K (2018) Fungi as biocontrol agent: an alternate to chemicals. In: Gehlot P, Singh J (eds) Fungi and their role in sustainable development: current perspectives. Springer, Singapore. https://doi.org/10.1007/978-981-13-0393-7_2
Skinner M, Parker BL, Kim JS (2014) Role of entomopathogenic fungi in integrated pest management In: Abrol DP (eds) Integrated pest management: current concepts and ecological perspective Academic Press, pp169–191 https://doi.org/10.1016/B978-0-12-398529-3.00011-7
Sparks TC, Nauen R (2015) IRAC: mode of action classification and insecticide resistance management. Pestic Biochem Physiol 121:122–128. https://doi.org/10.1016/j.pestbp.2014.11.014
Taning CNT, Gui S, De Schutter K, Jahan M, Castellanos NL. Christiaens O, Smagghe G (2021) A sequence complementarity-based approach for evaluating off-target transcript knockdown in Bombus terrestris, following ingestion of pest-specific dsRNA J Pest Sci, 94:487-503 https://doi.org/10.1007/s10340-020-01273-z
Urban P, Lautier T, Pompon D, Truan G (2018) Ligand access channels in cytochrome P450 enzymes: a review. Int J Mol Sci 19:1617. https://doi.org/10.3390/ijms19061617
Wang Q, Xu L (2012) Beauvericin, a bioactive compound produced by fungi: a short review. Molecules 17:2367–2377. https://doi.org/10.3390/molecules17032367
Wang LJ, Lu LH, He YR, Xie MQ (2010) Observation on the infection process of Beauveria bassiana on cuticle of the red imported fire ant, Solenopsis invicta Buren (Hymenoptera: Formicidae), using scanning electron microscopy. J Acta Entomologica Sinica 53:118–124. https://doi.org/10.1016/S1002-0721(10)60377-8
Yan S, Qian J, Cai C, Ma ZZ, Li JH, Yin MZ, Ren BY, Shen J (2020) Spray method application of transdermal dsRNA delivery system for efficient gene silencing and pest control on soybean aphid Aphis glycines. J Pest Sci 93:449–459. https://doi.org/10.1007/s10340-019-01157-x
Yang LZ, Keyhani NO, Tang GR, Tian C, Lu RP, Wang X, Pei Y, Fan YH (2014) Expression of a toll signalling regulator serpin in a mycoinsecticide for increased virulence. Appl Environ Microbiol 80:4531–4539. https://doi.org/10.1128/AEM.01197-14
Ye C, An X, Jiang YD et al (2019) Induction of RNAi core machinery’s gene expression by exogenous dsRNA and the effects of pre-exposure to dsRNA on the gene silencing efficiency in the pea aphid (Acyrthosiphon pisum). Front Physiol 9:1906. https://doi.org/10.3389/fphys.2018.01906
Ye C, Wang ZW, Sheng YL, Wang ZG, Smagghe G, Christiaens O, Niu JZ, Wang JJ (2021) GNBP1 as a potential RNAi target to enhance the virulence of Beauveria bassiana for aphid control. J Pest Sci in Press. https://doi.org/10.1007/s10340-021-01388-x
Zhao XD, Zhang BW, Fu LJ, Li QL, Lin Y, Yu XQ (2020) Possible insecticidal mechanism of cry41-related toxin against Myzus persicae by enhancing cathepsin B activity. J Agric Food Chem 68:4607–4615. https://doi.org/10.1021/acs.jafc.0c01020
Zheng Y, Hu YS, Yan S, Zhou H, Song DL, Yin MZ, Shen J (2019) A polymer/detergent formulation improves dsRNA penetration through the body wall and RNAi-induced mortality in the soybean aphid Aphis glycines. Pest Manag Sci 75:1993–1999. https://doi.org/10.1002/ps.5313
Zhu KY, Palli SR (2020) Mechanisms, applications, and challenges of insect RNA interference. Annu Rev Entomol 65:293–311. https://doi.org/10.1146/annurev-ento-011019-025224
Zimmermann G (2007) Review on safety of the entomopathogenic fungi Beauveria bassiana and Beauveria brongniartii. Biocontrol Sci Techn 17:553–596. https://doi.org/10.1080/09583150701309006
Funding
This research was supported by the National Natural Science Foundation of China-Major International (Regional) Joint Research Project (32020103010); 111 Project (B18044), China; and China Agriculture Research System of MOF and MARA, and the Foundation Project of Southwest University (SWU019033).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any conflicts of interest.
Consent to participate
Consent was given by all of the participants included in the study.
Consent for publication
All of the authors consent to the publication of this manuscript in Journal of Pest Science.
Ethical approval
This study does not contain any experiments using any animal species that require ethical approval.
Additional information
Communicated by Su Wang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.Fig. S1 Number of differentially expressed genes (DEGs) of aphids post 36 h , and 72 h sprayed with B. bassiana (Bb07). 0 h vs 36 h indicates the DEGs between 0 h and 36 h; 0 h vs 72 h indicates the DEGs between 0 h and 72 h; Co-regulated DEGs indicates the co-regulated DEGs between 0 h vs 36 h and 0 h vs 72 h; Co-upregulated DEGs indicates the co-upregulated DEGs between 0 h vs 36 h and 0 h vs 72 h; Co-downregulated DEGs indicates the co-downregulated DEGs between 0 h vs 36 h and 0 h vs 72h

Rights and permissions
About this article
Cite this article
Zhang, J., Ye, C., Wang, ZG. et al. DsRNAs spray enhanced the virulence of entomopathogenic fungi Beauveria bassiana in aphid control. J Pest Sci 96, 241–251 (2023). https://doi.org/10.1007/s10340-022-01508-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-022-01508-1