Abstract
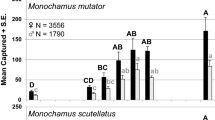
Studies have demonstrated that semiochemical-baited intercept traps differ in their performance for sampling insects, but we have an incomplete understanding of how and why intercept trap design effects vary among insects. This can significantly delay both the development of new and optimization of existing survey and detection tools. The development of a mechanistic understanding of why trap performance varies within and among species would mitigate this delay. The primary objective of this study was to develop methods to characterize and compare the odor plumes associated with intercept traps that differ in their performance for forest Coleoptera. We released CO2 and measured fluctuations of this tracer gas from 175-point locations arranged in a 2-by-3-by-2-m grid cuboid downwind of a standard multiple-funnel, a modified multiple-funnel, a panel, a canopy malaise trap, and a blank control (i.e., no trap) in a greenhouse. Significant differences in trapping efficacy between these different trap designs were observed for Monochamus scutellatus (Say) and Monochamus notatus (Drury) in a field trial. Significant differences were also observed in how CO2 accumulated in time at different positions downwind among these different trap designs. Turbulent dispersion is the dominant force structuring odor plumes and creates intermittency in the odor plume that is important for sustained upwind flight in insects. Methodological and instrumental limitations resulted in the inability to determine instantaneous plume structures and vortex shedding frequencies for different intercept trap designs. Although we observed differences in the odor plumes emanating downwind of the different intercept trap designs, we were unable to reconcile these differences with capture rates of the different trap designs for M. scutellatus and M. notatus.





Similar content being viewed by others
Data accessibility
All data and R code are available on the Dryad Digital Repository (Bouwer et al. 2019).
References
Allison JD, Redak RA (2017) The impact of trap type and design features on survey and detection of bark and woodboring beetles and their associates: a review and meta-analysis. Annu Rev Entomol 62(1):127–146. https://doi.org/10.1146/annurev-ento-010715-023516
Allison JD, Borden JH, Seybold SJ (2004) A review of the chemical ecology of the Cerambycidae (Coleoptera). Chemoecology 14(4):123–150. https://doi.org/10.1007/s00049-004-0277-1
Allison JD, Johnson CW, Meeker JR, Strom BL, Butler SM (2011) Effect of aerosol surface lubricants on the abundance and richness of selected forest insects captured in multiple-funnel and panel traps. J Econ Entomol 104(4):1258–1264. https://doi.org/10.1603/EC11044
Allison JD, McKenney JL, Millar JG, McElfresh JS, Mitchell RF, Hanks LM (2012) Response of the woodborers Monochamus carolinensis and Monochamus titillator (Coleoptera: Cerambycidae) to known cerambycid pheromones in the presence and absence of the host plant volatile α-pinene. Environ Entomol 41(6):1587–1596. https://doi.org/10.1603/EN12185
Allison JD, Bhandari BD, McKenney JL, Millar JG (2014) Design factors that influence the performance of flight intercept traps for the capture of longhorned beetles (Coleoptera: Cerambycidae) from the subfamilies Lamiinae and Cerambycinae. PLoS ONE 9(3):e93203. https://doi.org/10.1371/journal.pone.0093203
Allison JD, Graham EE, Poland TM, Strom BL (2016) Dilution of fluon before trap surface treatment has no effect on Longhorned Beetle (Coleoptera: Cerambycidae) captures. J Econ Entomol 109(3):1215–1219. https://doi.org/10.1093/jee/tow081
Allison JD, Strom B, Sweeney J, Mayo P (2018) Trap deployment along linear transects perpendicular to forest edges: impact on capture of longhorned beetles (Coleoptera: Cerambycidae). J Pest Sci 92:299–308. https://doi.org/10.1007/s10340-018-1008-7
Baker TC, Haynes KF (1989) Field and laboratory electroantennographic measurements of pheromone plume structure correlated with oriental fruit moth behaviour. Physiol Entomol 14(1):1–12. https://doi.org/10.1111/j.1365-3032.1989.tb00931.x
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B 57(1):289–300
Bouwer MC, MacQuarrie CJK, Gil OJ, Slippers B, Allison JD (2019) Data from: impact of intercept trap type on plume structure: a potential mechanism for differential performance of intercept trap designs for longhorned beetles. Dryad Digital Repository
Cardé RT (2016) Moth navigation along pheromone plumes. In: Allison JD, Cardé RT (eds) Pheromone communication in moths, 1st edn. University of California Press, California, pp 173–189
Chénier JVR, Philogene BJR (1989) Evaluation of three trap designs for the capture of conifer-feeding beetles and other forest Coleoptera. Can Entomol 121(2):159–167. https://doi.org/10.4039/Ent121159-2
Cooperband MF, Cardé RT (2006) Comparison of plume structures of carbon dioxide emitted from different mosquito traps. Med Vet Entomol 20(1):1–10. https://doi.org/10.1111/j.1365-2915.2006.00614.x
Czokaljo D, Ross D, Kirsch P (2001) InterceptTM panel trap, a novel trap for monitoring forest Coleoptera. J For Sci 47(2):63–65
de Groot P, Nott RW (2001) Evaluation of traps of six different designs to capture pine sawyer beetles (Coleoptera: Cerambycidae). Agric For Entomol 3(2):107–111. https://doi.org/10.1046/j.1461-9563.2001.00087.x
de Groot P, Nott RW (2003) Response of Monochamus (Col., Cerambycidae) and some Buprestidae to flight intercept traps. J Appl Entomol 127(9–10):548–552. https://doi.org/10.1046/j.1439-0418.2003.00799.x
Dekker T, Takken W, Cardé R (2001) Structure of host odour plumes influences catch of Anopheles gambiae s.s. and Aedes aegypti in a dual choice olfactometer. Physiol Entomol 26(2):124–134. https://doi.org/10.1046/j.1365-3032.2001.00225.x
Dodds KJ, Allison JD, Miller DR, Hanavan RP, Sweeney J (2015) Considering species richness and rarity when selecting optimal survey traps: Comparisons of semiochemical baited flight intercept traps for Cerambycidae in eastern North America. Agric For Entomol 17(1):36–47. https://doi.org/10.1111/afe.12078
Elkinton JS, Cardé RT, Mason CJ (1984) Evaluation of time-averaged dispersion models for estimating pheromone concentration in a deciduous forest. J Chem Ecol 10(7):1081–1108. https://doi.org/10.1007/BF00987515
Fares Y, Sharpe PJH, Magnuson CE (1980) Pheromone dispersion in forests. J Theor Biol 84(2):335–359. https://doi.org/10.1016/S0022-5193(80)80010-5
Farrel JA, Murlis J, Long XZ, Li WEI, Cardé RT (2002) Filament-based atmospheric dispersion model to achieve short time-scale structure of odor plumes. Environ Fluid Mech 2(1):143–169. https://doi.org/10.1023/A:1016283702837
Garcia R, Benet M, Arnau C, Cobo E (2004) Efficiency of the cross-over design: an empirical estimation. Stat Med 23(24):3773–3780. https://doi.org/10.1002/sim.2072
Geier M, Bosch OJ, Boeckh J (1999) Influence of odour plume structure on upwind flight of mosquitoes towards hosts. J Exp Biol 202(12):1639–1648. https://doi.org/10.1242/jeb.055186
Girling RD, Higbee BS, Cardé RT (2013) The plume also rises: trajectories of pheromone plumes issuing from point sources in an orchard canopy at night. J Chem Ecol 39(9):1150–1160. https://doi.org/10.1007/s10886-013-0341-9
Justus KA, Cardé RT (2002) Flight behaviour of males of two moths, Cadra cautella and Pectinophora gossypiella, in homogeneous clouds of pheromone. Physiol Entomol 27(1):67–75. https://doi.org/10.1046/j.1365-3032.2002.00270.x
Kuenen LPS, Cardé RT (1994) Strategies for recontacting a lost pheromone plume: casting and upwind flight in the male gypsy moth. Physiol Entomol 19(1):15–29. https://doi.org/10.1111/j.1365-3032.1994.tb01069.x
Lenth R (2018) Emmeans: estimated marginal means, aka least-squares means. R Package Version 1(2):1
Lewis T, Macaulay EDM (1976) Design and elevation of sex‐attractant traps for pea moth, Cydia nigricana (Steph.) and the effect of plume shape on catches. Ecol Entomol 1(3):175–187
Lindgren BS (1983) A multiple funnel trap for scolytid beetles (Coleoptera). Can Entomol 115(3):299–302. https://doi.org/10.4039/Ent115299-3
Lingafelter SW (2007) Illustrated key to the longhorned woodboring beetles of the Eastern United States. Coleopterists Society, North Potomac
Mafra-Neto A, Cardé RT (1994) Fine-scale structure of pheromone plumes modulates upwind orientation of flying moths. Nature 369(6476):142–144
McCune B, Grace J (2002) Analysis of ecological communities. MjM Software Design, Gleneden Beach
McIntosh RL, Katinic PJ, Allison JD, Borden JH, Downey DL (2001) Comparative efficacy of five types of trap for woodborers in the cerambycidae, buprestidae and siricidae. Agric For Entomol 3(2):113–120. https://doi.org/10.1046/j.1461-9563.2001.00095.x
Miller DR, Crowe CM, Barnes BF, Gandhi KJK, Duerr DA (2013) Attaching lures to multiple-funnel traps targeting saproxylic beetles (Coleoptera) in pine stands: inside or outside funnels? J Econ Entomol 106(1):206–214. https://doi.org/10.1603/EC12254
Morewood WD, Hein KE, Katinic PJ, Borden JH (2002) An improved trap for large wood-boring insects, with special reference to Monochamus scutellatus (Coleoptera: Cerambycidae). Can J For Res 32(3):519–525. https://doi.org/10.1139/x01-224
Murlis J, Jones C (1981) Fine-scale structure of odour plumes in relation to insect orientation to distant pheromone and other attractant sources. Physiol Entomol 6(1):71–86. https://doi.org/10.1111/j.1365-3032.1981.tb00262.x
Murlis J, Elkinton JS, Cardé RT (1992) Odor plumes and how insects use them. Annu Rev Entomol 37(1):505–532. https://doi.org/10.1146/annurev.en.37.010192.002445
Murlis J, Willis MA, Cardé RT (2000) Spatial and temporal structures of pheromone plumes in fields and forests. Physiol Entomol 25(3):211–222. https://doi.org/10.1046/j.1365-3032.2000.00176.x
Pinheiro JC, Bates DM (2000) Mixed-effects models in S and S-PLUS. Springer, New York
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2020). nlme: linear and nonlinear mixed effects models. R package version 3.1-144. https://CRAN.R-project.org/package=nlme
R Development Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Sakamoto H, Haniu H (1990) A study on vortex shedding from spheres in a uniform flow. J Fluid Eng 112(4):386–392. https://doi.org/10.1115/1.2909415
Schal C (1982) Intraspecific vertical stratification as a mate-finding mechanism in tropical cockroaches. Science 215(4538):1405–1407. https://doi.org/10.1126/science.215.4538.1405
Strand T, Lamb B, Thistle H, Allwine E, Peterson H (2009) A simple model for simulation of insect pheromone dispersion within forest canopies. Ecol Model 220(5):640–656. https://doi.org/10.1016/j.ecolmodel.2008.11.018
Thistle HW, Peterson H, Allwine G, Lamb B, Strand T, Holsten EH, Shea PJ (2004) Surrogate pheromone plumes in three forest trunk spaces: composite statistics and case studies. For Sci 50(5):610–625. https://doi.org/10.1093/forestscience/50.5.610
Thistle H, Strom B, Strand T et al (2011) Atmospheric dispersion from a point source in four southern pine thinning scenarios: basic relationships and case studies. Trans ASABE 54:1219–1236. https://doi.org/10.13031/2013.39021
Tukey JW (1949) Comparing individual means in the analysis of variance. Biometrics 5(2):99–114. https://doi.org/10.2307/3001913
Ulyshen MD, Sheehan TN (2019) Trap height considerations for detecting two economically important forest beetle guilds in southeastern US forests. J Pest Sci 92:253–265. https://doi.org/10.1007/s10340-017-0883-7
Vickers NJ (2006) Winging it: moth flight behavior and responses of olfactory neurons are shaped by pheromone plume dynamics. Chem Senses 31(2):155–166. https://doi.org/10.1093/chemse/bjj011
Vickers NJ, Baker TC (1994) Reiterative responses to single strands of odor promote sustained upwind flight and odor source location by moths. Neurobiology 91(13):5756–5760. https://doi.org/10.1073/pnas.91.13.5756
Willis MA, Murlis J, Cardé RT (1994) Effects of pheromone plume structure and visual stimuli on the pheromone-modulated upwind flight of male gypsy moths (Lymantria dispar ) in a Forest. J Insect Behav. https://doi.org/10.1007/BF01989742
Wyatt TD, Phillips ADG, Gregoire JC (1993) Turbulence, trees and semiochemicals: wind-tunnel orientation of the predator, Rhizophagus grandis, to its barkbeetle prey Dendroctonus micans. Physiol Entomol 18(2):204–210. https://doi.org/10.1111/j.1365-3032.1993.tb00469.x
Wyatt TD, Vastiau K, Birch MC (1997) Orientation of flying male Anobium punctatum (Coleoptera: Anobiidae) to sex pheromone: separating effects of visual stimuli and physical barriers to wind. Physiol Entomol 22(2):191–196. https://doi.org/10.1111/j.1365-3032.1997.tb01157.x
Yanega DN, Natural History Survey Division (1996) Field guide to northeastern longhorned beetles (Coleoptera:Cerambycidae). Illinois Natural History Survey, Champaign
Yaws CL (2014) Diffusion coefficient in air—organic compounds. In: Yaws CL (ed) Transport properties of chemicals and hydrocarbons, 2nd edn. Gulf Professional Publishing, Oxford, pp 407–496
Zuur AF, Ieno EN, Walker N, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, New York. https://doi.org/10.1007/978-0-387-87458-6
Acknowledgements
Reginald Nott is thanked for technical support. We thank Patt Ross for access to New North Greenhouses, Brian Strom and Ring Cardé for review of an earlier draft and the National Research Foundation of South Africa (Grant #99644), Natural Resources Canada and USDA—Animal and Plant Health Inspection Service (Grant #15–8130-0395-CA), for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Additional information
Communicated by J.D. Sweeney.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.






Rights and permissions
About this article
Cite this article
Bouwer, M.C., MacQuarrie, C.J.K., Aguirre-Gil, O.J. et al. Impact of intercept trap type on plume structure: a potential mechanism for differential performance of intercept trap designs for Monochamus species. J Pest Sci 93, 993–1005 (2020). https://doi.org/10.1007/s10340-020-01204-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-020-01204-y