Abstract
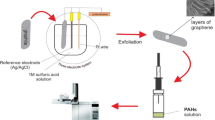
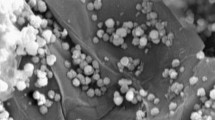
Multiwalled carbon nanotubes were exposed to hydrothermal treatment for obtaining graphene oxide nanoribbons (GONRs). The fabricated graphene oxide nanoribbons have been morphologically and compositionally characterized via FE-SEM, XRD, and FT-IR techniques. The as-synthesized GONRs have been used as sorbent phase for headspace solid-phase microextraction of phthalate esters (PEs) from aqueous solutions. In this regard, the GC–FID analysis route has been used for quantification of PEs. The new SPME fiber shows remarkable analytical figures of merit including broad dynamic linear ranges, low limits of detection, as well as good stability and reasonable relative standard deviations for evaluation of PEs. The linearity of the method for analysis of PEs including DnBP, DnPP, DEHP, DEHA, BBP, and DMP was between the range of 0.05–100, 0.05–100, 0.1–100, 0.1–100, 0.2–100, and 0.5–100 μg L−1, respectively. The limits of detection (based on S/N = 3) and correlation coefficients were found to be in the range of 0.02–0.2 μg L−1 and 0.9907–0.9952, correspondingly. The prepared GONR-coated SPME fiber shows larger extraction yield in comparison to pristine MWNTs and commercial PDMS SPME fibers. Furthermore, real sample analysis showed that there is no significant matrix effect for evaluation of PEs from environmental water samples and proposed method could be used for evaluation and determination of PEs from aqueous samples in a precise and accurate manner. The existence of functional groups, π–π interactions, as well as hydrogen bonding between adsorbent phase and PE analytes could be the reason for observing such a high extraction yield.








Similar content being viewed by others
References
Peñalver A, Pocurull E, Borrull F, Marcé RM (2001) J Chromatogr A 922:377–384. https://doi.org/10.1016/S0021-9673(01)00920-7
Sajid M, Basheer C, Alsharaa A, Narasimhan K, Buhmeida A, Al Qahtani M, Al-Ahwal MS (2016) Anal Chim Acta 924:35–44
Gao D-W, Wen Z-D (2016) Sci Total Environ 541:986–1001. https://doi.org/10.1016/j.scitotenv.2015.09.148
Amanzadeh H, Yamini Y, Moradi M, Asl YA (2016) J Chromatogr A 1465:38–46. https://doi.org/10.1016/j.chroma.2016.08.068
Cheng L, Pan S, Ding C, He J, Wang C (2017) J Chromatogr A 1511:85–91. https://doi.org/10.1016/j.chroma.2017.07.012
Feng Z, Huang C, Guo Y, Tong P, Zhang L (2019) Anal Chim Acta 1084:43–52. https://doi.org/10.1016/j.aca.2019.08.015
Sun M, Feng J, Qiu H, Fan L, Li L, Luo C (2013) J Chromatogr A 1300:173–179. https://doi.org/10.1016/j.chroma.2013.04.061
Feng J, Sun M, Bu Y, Luo C (2015) J Sep Sci 38:128–133
Du L, Ma L, Qiao Y, Lu Y, Xiao D (2016) Food Chem 197:1200–1206. https://doi.org/10.1016/j.foodchem.2015.11.082
Carnol L, Schummer C, Moris G (2017) Food Anal Methods 10:298–309
Zhang Y, Yang Y, Li Y, Zhang M, Wang X, Du X (2015) Anal Chim Acta 876:55–62
Xie Q, Sun D, Han Y, Jia L, Hou B, Liu S, Li D (2016) J Sep Sci 39:857–863
Chi J, Gao J (2015) Chemosphere 119:59–64
Vavrouš A, Pavloušková J, Ševčík V, Vrbík K, Čabala R (2016) J Chromatogr A 1456:196–204
Pang Y-H, Yue Q, Huang Y-Y, Yang C, Shen X-F (2020) Talanta 206:120194
Guiñez M, Martinez LD, Fernandez L, Cerutti S (2017) Microchem J 131:1–8
Montevecchi G, Masino F, Zanasi L, Antonelli A (2017) Food Chem 221:1354–1360
González-Sálamo J, González-Curbelo MÁ, Socas-Rodríguez B, Hernández-Borges J, Rodríguez-Delgado MÁ (2018) Chemosphere 201:254–261
Wang J, Huang S, Wang P, Yang Y (2016) Food Control 67:278–284
Sun M, Feng J, Bu Y, Wang X, Duan H, Luo C (2015) Talanta 134:200–205
Wang F, Li J, Wu J-F, Zhao G-C (2018) Chromatographia 81:799–807
Spietelun A, Kloskowski A, Chrzanowski W, Namieśnik J (2012) Chem Rev 113:1667–1685
Song X-L, Chen Y, Yuan J-P, Qin Y-J, Zhao R-S, Wang X (2016) J Chromatogr A 1468:17–22
Zhang S, Du Z, Li G (2011) Anal Chem 83:7531–7541
Makkliang F, Kanatharana P, Thavarungkul P, Thammakhet C (2015) Food Chem 166:275–282
Ghaemmaghami M, Yamini Y, Amanzadeh H, Hosseini Monjezi B (2018) Chem Commun 54:507–510. https://doi.org/10.1039/C7CC08273H
Liu X, Sun Z, Chen G, Zhang W, Cai Y, Kong R, Wang X, Suo Y, You J (2015) J Chromatogr A 1409:46–52
Du J, Wang F, Wang Z, Wang X, Du X (2019) Anal Methods 11:1237–1247
Dargahi R, Ebrahimzadeh H, Alizadeh R (2018) Microchim Acta 185:150
Tamayo F, Turiel E, Martín-Esteban A (2007) J Chromatogr A 1152:32–40
Luo Z, Cheng G, Li X, Wang L, Shu H, Cui X, Chang C, Zeng A, Fu Q (2019) J Sep Sci 3352:3362
Esfandiarnejad R, Sereshti H, Farahani A (2019) Anal Bioanal Chem 411(16):3631–3640
Kamalabadi M, Mohammadi A, Alizadeh N (2016) Talanta 156:147–153
Zakerian R, Bahar S (2017) J Sep Sci 40:4439–4445
Crucello J, Miron LF, Ferreira VH, Nan H, Marques MO, Ritschel PS, Zanus MC, Anderson JL, Poppi RJ, Hantao LW (2018) Anal Bioanal Chem 410:4749–4762
Trujillo-Rodríguez MJ, Nan H, Anderson JL (2018) J Chromatogr A 1540:11–20
Cheng H, Song Y, Bian Y, Wang F, Ji R, He W, Gu C, Ouyang G, Jiang X (2018) Microchim Acta 185:56
Patil V, Dennis RV, Rout TK, Banerjee S, Yadav GD (2014) RSC Adv 4:49264–49272
Asadian E, Shahrokhian S, Jokar E (2014) Sens Actuators B Chem 196:582–588
Dimiev AM, Khannanov A, Vakhitov I, Kiiamov A, Shukhina K, Tour JM (2018) ACS Nano 12:3985–3993. https://doi.org/10.1021/acsnano.8b01617
Wu Z-L, Li C-K, Yu J-G, Chen X-Q (2017) Sens Actuators B Chem 239:544–552. https://doi.org/10.1016/j.snb.2016.08.062
Higginbotham AL, Kosynkin DV, Sinitskii A, Sun Z, Tour JM (2010) ACS Nano 4:2059–2069. https://doi.org/10.1021/nn100118m
Mehdinia A, Bashour F, Roohi F, Jabbari A, Saleh A (2012) J Sep Sci 35:563–570
Peijnenburg WJGM, Struijs J (2006) Ecotoxicol Environ Saf 63:204–215. https://doi.org/10.1016/j.ecoenv.2005.07.023
Wang X, Lou X, Zhang N, Ding G, Chen Z, Xu P, Wu L, Cai J, Han J, Qiu X (2015) Environ Toxicol Chem 34:2205–2212
Banitaba MH, Davarani SS, Pourahadi A (2013) J Chromatogr A 1283:1–8. https://doi.org/10.1016/j.chroma.2013.01.092
Jafari M, Ebrahimzadeh H, Banitaba MH, Davarani SSH (2014) J Sep Sci 37:3142–3149
Asadollahzadeh H, Noroozian E, Maghsoudi S (2010) Anal Chim Acta 669:32–38. https://doi.org/10.1016/j.aca.2010.04.029
Hou X, Guo Y, Liang X, Wang X, Wang L, Wang L, Liu X (2016) Talanta 153:392–400. https://doi.org/10.1016/j.talanta.2016.03.034
Holadová K, Prokůpková G, Hajšlová J, Poustka J (2007) Anal Chim Acta 582:24–33
Gorji S, Biparva P, Bahram M, Nematzadeh G (2019) Talanta 194:859–869
Tian T, Wang F, Zhao G-C (2020) Microchem J 153:104510
Sukree W, Sooksawat D, Kanatharana P, Thavarungkul P, Thammakhet-Buranachai C (2020) J Environ Sci Health B 55:60–68
Mehrani Z, Ebrahimzadeh H, Moradi E (2019) J Chromatogr A 1600:87–94
Acknowledgements
The authors gratefully thankful of financial support from Arak Branch, Islamic Azad University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kazemi, M., Niazi, A. & Yazdanipour, A. Solid-Phase Microextraction of Phthalate Esters from Aqueous Media by Functionalized Carbon Nanotubes (Graphene Oxide Nanoribbons) and Determination by GC–FID. Chromatographia 84, 559–569 (2021). https://doi.org/10.1007/s10337-021-04032-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-021-04032-z