Abstract
How the migration behavior of long-lived birds develops throughout their life is still a research question in ornithology that is largely unanswered. Here, we analyzed GPS data of 70 juvenile Black Storks during the first 3 years of their migration to determine the influence of age and experience on migration patterns. We focused on the choice of the migration flyway, as well as the movements in the wintering area, and the changes in departure times with increasing age. Migratory routes of German Black Storks were divided into the western (82% of all tracked individuals) and eastern flyways (16%). The central migratory flyway through Italy or Greece was taken only by two individuals and never repeatedly. In addition, we found that the wintering area decreased by almost 60% during their first 3 years of life. In addition, spring departure dates from the wintering areas also became earlier with increasing age. However, we did not find changes in departures from the breeding area in autumn. Thus, our study shows that age and migration experiences have a considerable influence on essential migratory decisions in Black Storks. Further, it also demonstrates that older storks rely strongly on previous experiences to perform their migrations efficiently.
Zusammenfassung
Der Einfluss des Alters auf räumliche und zeitliche Zugmuster von Schwarzstörchen aus Deutschland.
Wie sich das Zugverhalten von Zugvögel im Laufe ihres Lebens verändert, ist eine in der Ornithologie weitgehend unbeantwortete Forschungsfrage. In unserer Studie haben wir uns die GPS-Daten von 70 jungen Schwarzstörchen (Ciconia nigra) angesehen, um herauszufinden, wie Alter und Erfahrung ihre Flugrouten beeinflussen. Wir haben uns besonders darauf konzentriert, welche Wege sie wählen, wie sie sich in ihren Winterquartieren bewegen und wie sich ihre Abflugzeiten mit dem Alter ändern. Wir haben herausgefunden, dass die meisten deutschen Schwarzstörche (82%) eine westliche Zugroute wählen, während 16% den östlichen Weg bevorzugen. Nur zwei Vögel haben sich für die zentrale Route durch Italien oder Griechenland entschieden und das auch nicht mehr als einmal in ihrem Leben. Interessanterweise haben wir auch festgestellt, dass die Störche im Laufe der ersten drei Jahre ihres Lebens ihr Winterquartier um fast 60% verkleinern. Außerdem brechen sie, je älter sie werden, im Frühling früher zu ihren Brutplätzen auf. Aber im Herbst, wenn es Zeit ist von den Brutplätzen wegzuziehen, haben wir keine Veränderungen festgestellt. Unsere Untersuchung zeigt, dass das Alter und die Erfahrungen der Störche einen großen Einfluss darauf haben, wie sie ihre Reise planen. Ältere Störche verlassen sich stark auf ihre bisherigen Erfahrungen, um ihre Reisen effizient zu gestalten.
Similar content being viewed by others
Introduction
The German population of Black Storks (Ciconia nigra) was nearly driven to extinction due to persecution and intensified agricultural and forestry practices at the beginning of the last century, especially in the southern and western regions. However, from the 1930s onwards, there has been a gradual recovery, with the species re-establishing from Eastern Europe (Janssen et al. 2004). The German Black Stork population has now increased to between 800 and 900 pairs and the population trend remains positive (Gerlach et al. 2019; Bundesamt für Naturschutz 2020). To maintain this positive population trend and support the Black Stork population, it is essential to study not only local breeding behavior but also explore migratory patterns, like routes, destinations, and timing. Knowledge of these critical parameters in migration behavior will enable successful conservation decisions in the future (Flack et al. 2022).
Black Storks are long-distance migrants that, in the western Palaearctic, show a migratory divide (Bobek et al. 2008). Birds from the western flyway cross Europe and northern Africa to overwinter in the west and central Sahel region (Cano and Tellería, 2013; Chevallier et al. 2011; Jadoul et al. 2003). Over the past 20 years, there has been an increase in telemetry studies, providing new insights into individual migration behavior, especially regarding routes, wintering, and stopover sites (Chevallier et al. 2010a, 2011). However, these telemetry projects often had relatively small sample sizes (Bobek et al. 2008; Larue et al. 2016), or the storks were captured as adults (Chevallier et al. 2011; Jadoul et al. 2003) which did not allow the study of the development of the migratory behavior of juvenile Black Storks.
The ontogeny of migration behavior is still an area in ornithology that is largely characterized by knowledge gaps (Flack et al. 2022). While the migration behavior of short‐lived migrants may be rather fixed and stable, long‐lived migrants can progressively adjust their migration behavior to the needs of their ongoing life stage. We know that migration strategies and performances can vary enormously between individual Black Storks (Cano and Tellería 2013), but do these strategies also change throughout the life of an individual? It has been shown that selective mortality can shape lifelong migration patterns at the population level, selecting for individuals with efficient routes (Sergio et al. 2014). In addition, at the individual level, individuals can improve their migration behavior during their lifetime through learning (Aikens et al. 2024). However, due to a lack of long-term tracking data from early life onward, it is for most species unclear whether individuals change their migration patterns with age.
In this paper, we examine a dataset of 70 juvenile Black Storks that were monitored with GPS devices between 2017 and 2022. Despite high juvenile mortality and some technical failures of the transmitters, this project provides a large data, tracking juvenile Black Storks over a maximum of 3 consecutive years. Thus, we examine how the spatial and temporal patterns of the migration behavior of juvenile Black Storks change over the first 3 years of their life. To achieve this goal, we first examine the temporal features of the migration behavior and explore how they change with increasing age. Since Black Storks reach sexual maturity between the ages of 3 and 4 (Janssen et al. 2004), we expect that especially, in their third year of life, a clear temporal shift in departure date becomes visible. We predict that spring departure dates (i.e., departures from the wintering area) take place significantly earlier in older birds, because an earlier arrival at the breeding area may be essential to finding and defending a breeding territory. Second, we also examine the spatial features of the storks’ migration behavior. We assume that age influences the area used during the wintering period. Learning and experiences may allow birds to spend less time exploring and visiting areas they have encountered in previous years (Lewis et al. 2021). There is evidence that juvenile birds show lower fidelity, especially in the wintering area, as they explore the region that is new to them (Fayet 2020). Thus, we expect to see a decrease in the distance traveled during winter with increasing age of the birds.
Methods
Telemetry project and GPS devices
To examine Black Stork migration behavior in Germany, we equipped 70 Black Stork nestlings from 24 nests with a miniature data logger (Table 1). The nests of these birds were in Brandenburg (nnest = 1, nind = 3), Bavaria (nnest = 17, nind = 56), and Mecklenburg Vorpommern (nnest = 4, nind = 11). Nests were chosen based on good accessibility. The selected breeding sites are located in special reference areas of each federal state. We always tagged all juveniles from the nests, except one nest in Bavaria where only four out of five juveniles were tagged, and one nest in Brandenburg where only three of four juveniles were tagged. The solar-powered GPS devices are custom-made by the Max Planck Institute of Animal Behavior (size 25 × 38 × 15 mm, weight 21 g). Each data logger consists of a central 8-bit processor with a 16 MByte flash memory. Position data are generated via a GPS receiver, communication is by SMS via a GSM module. In addition to positional data (accuracy of approximately 2.5 m), timestamp, height above sea level, speed, time to first fix, and battery voltage are also stored. At full battery power, the devices take hourly GPS positions. Yet, when battery power is below a threshold, the devices stop recording GPS positions, until batteries are re-charged by the solar panels. Thus, sampling frequency of the dataset ranges from hourly positions to a position every couple of days. Data are sent via GSM, or data that were not received due to a missing GSM connection (e.g., Sahara) could be retrieved afterward with the help of a downlink command when the logger was again in a reception area. All data were stored in Movebank (Kays et al. 2022).
The devices were attached as a leg band above the intertarsal joint when the juveniles were approximately 7 weeks old. To do this, author CR climbed up to the nest to attach the device and the leg band (ELSA ring; Fiedler et al. 2002). Tagging happened in the years 2017 (4), 2018 (15), and 2019 (51; see Table 1). The devices tracked the position of the birds until the death of the individual or until the device stopped working. We were not able to distinguish between these two scenarios which is why we did not analyze survival and mortality.
Data cleaning
After a first visual examination of the GPS trajectories, we removed outliers, i.e., unrealistic locations (721 locations, i.e., 2.4% of all collected locations), by filtering locations based on the calculated flight speed between consecutive positions. We only included locations that did not exceed a flight speed of 60 km/h between two data points (Bobek et al. 2008; Chevallier et al. 2010a). After filtering, we reduced the number of GPS locations to a total of 29,551 locations.
Data analyses
All data analyses and visualizations were performed using R (Version 4.2.2). We used the packages dplyr, tidyverse, geosphere, move, ggplot2, sf, and lmerTest for analyses, visualizations, and statistical analyses.
We determined departure and arrival dates based on the crossing of certain latitudes. For the autumn migration, the first position south of 45° latitude, which marks the crossing of the Alps, was set as departure date. Arrival at the wintering site was defined as the first position south of 18° latitude, i.e., the southern border of the Sahara. The same reference lines were used for the spring migration.
The migration flyway was determined for each individual based on their route around the Mediterranean Sea (east vs. west). Individuals who crossed the Mediterranean Sea were classified as central migrants. For each migration year, we calculated the cumulative travel distance for both the wintering period and the summer period, as well as for the two migration journeys. We calculated the sum of all distances between successive GPS locations within the given period. The distinction between the different periods was made using the previously calculated departure and arrival dates.
For the wintering areas, we drew minimum convex polygons (MCPs) (Nilsen et al. 2008) around all recorded GPS points during the wintering period to calculate the home range. The departure and arrival dates were again used as a reference, but the time was reduced by 10 days after arrival and 10 days before departure to reduce the effect of the flight distance to and from the wintering area.
To test for an effect of age on migration patterns, we used linear mixed models (with a Gaussian distribution), since there were repeated measurements for the same individual over the years (Gałecki and Burzykowski 2013). Individual ID was used as a random factor. We explored the influence of age (as categorical variable) on departure dates in autumn and spring, on flight distances during the summer and winter periods, and on the area visited during summer and winter.
Results
Migratory flyways of juvenile Black Storks
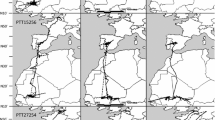
Of the 70 tagged Black Storks, 59 individuals departed on migration in their first year (see Table 1 for details on sample sizes and Fig. 1 for trajectories of the different years). Further, 49 juveniles reached their wintering grounds. This means that the migratory behavior of the monitored individuals was divided into birds moving along the western flyway via Spain and the Strait of Gibraltar to Western Sub-Saharan Africa (year 1: n = 40, year 2: n = 18) and the eastern flyway via the Balkans and the Bosporus to Eastern Africa (year 1: n = 8; year 2: n = 4). Moreover, two individuals were observed using the central migratory flyway by flying either through Italy or Greece (these birds did that in their first and second migration, respectively).
Of all 70 tracked individuals, 24 Black Storks completed at least 2 autumn migrations, allowing us to observe changes in subsequent migration patterns. Three individuals changed their migration behavior profoundly by switching between migratory flyways: two storks switched from the central to the eastern flyway, whereas one bird changed from the eastern to the western flyway. No bird flew repeatedly along the central flyway.
Of a total of 24 monitored nests, 6 nests had different migration flyways between the siblings, i.e., some individuals took the western migration route, while their siblings took the eastern migratory route.
Development of temporal migration patterns.
Next, we examined how departure dates during spring and autumn changed throughout an individual’s life. Median autumn departure date showed that the autumn departure dates of year 1 did not differ from those in year 2 (Fig. 2A, estimate = -1.364, SE = 6.381, t = -0.214, p = 0.831) and 3 (estimate = 5.6, SE = 7.583, t = 0.739, p = 0.462).
In spring, on the other hand, departures from the wintering sites in Sub-Saharan Africa changed significantly as individuals aged (Fig. 2B). More specifically, there is no difference between the departures of year 1 and 2 (estimate = -6.594, SE = 4.160, t = -1.585, p = 0.125), but departure dates are significantly later in year 1 compared to year 3 (estimate = -23.314, SE = 6.398, t = -3.644, p < 0.01). The median departure date decreased from April 16th (year 1) to April 5th (in year 2), and to March 13th (in year 3).
Development of spatial behavior during breeding and wintering
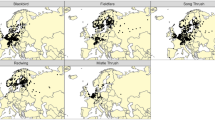
Next, we examined whether age influenced the movements during the breeding and wintering periods. During their first summer, the cumulative flight distance varied strongly between individuals, ranging from 828 to 6248 km (Fig. 3A). However, we found no significant difference in summer distance between years 1 and 2 (estimate = 210.18, SE = 261.80, t = 0.803, p = 0.435).
This was different when looking at the cumulative flight distance during the wintering periods (Fig. 3B). The cumulative flight distance during winter decreased stepwise as individuals became older. Distances flown in winter differed between first and second year (estimate = -1574.67, SE = 363.46, df = 24.85, t = -4.332, p < 0.001) and between first and third year (estimate = -1809.65, SE = 531.39, df = 26.21, t = -3.406, p = 0.002). Whereas in year 1, the maximum distance covered was 9754 km, it decreased to 8342 km in year 2 and 4245 km in year 3. This decrease in movements was also reflected in the overall size of the area in winter. We examined the sizes of the wintering areas of different years by calculating the smallest possible areas of the recorded locations within the wintering period. As seen in the two example birds (Fig. 4A, B), the wintering range decreased strongly as individuals grew older.
Finally, we compared the size of wintering areas of all individuals across all years. For this analysis, we removed one outlier that exhibited an extraordinarily large wintering area in year 2 (see Fig. 4C). Median home ranges of all birds decreased by 58% from 84,089 km2 in winter 1 to 35,335 km2 in the second winter and further shrunk to 23,463 km2 in winter 3. Thus, wintering area was significantly smaller in year 2 than in year 1 (estimate = -51,247.81, SE = 19,839.09, t = -2.583, p = 0.019), but there was no significant difference between year 1 and year 3 (estimate = – 38,423.70, SE = 29,594.48, t = – 1.298, p = 0.210).
Discussion
We found that as Black Storks become older, they shift their departure from their wintering sites to earlier in the season. In contrast to studies of other migratory bird species (Kokko 1999), we did not detect any change in the departure date from the breeding area. Yet, we also found that the cumulative flight distances during winter and the total area of their wintering sites decreased with age. This indicates that with the transition to the breeding stage, young Black Storks adjust their migration patterns to arrive earlier at their breeding sites and reduce their ranging behavior in winter.
These results are similar to what has been observed in some other bird species (Aikens et al. 2024; Campioni et al. 2020; Sergio et al. 2014). Black kites (Milvus migrans) also depart earlier from their wintering area after reaching sexual maturity (Sergio et al. 2014). Black Storks start to breed when approximately 3 years old (Janssen et al. 2004), thus older birds may experience an increased pressure to return early to the breeding grounds to find a good nesting site and a suitable mate. Immature individuals, in contrast, are not in a hurry and can spend more time exploring their wintering habitats to find the most suitable locations during winter. This ontogenetic shift is also confirmed by our finding that the size of the wintering area is largest in year 1 and decreases as individuals gain more experience, similar to what has also been found by Chevallier et al. (2010b). The decreasing size of the wintering ranges can represent a learning process by which animals integrate new information about the distribution of resources in the overwintering habitat (Lewis et al. 2021). With time and experience, explorative movements shift to exploiting movements that can be seen as repeated visits to familiar areas and do not require long-distance travel (de Grissac et al. 2016). Further, it has been stated that adult Black Storks exhibit territorial behavior, even at their wintering sites (Jadoul et al. 2003). Thus, juveniles, with their lower body mass, may have to expand their search to avoid competition and secure suitable wintering territories. Juveniles show, in general, lower foraging efficiency (Chevallier et al. 2010b) which, combined with higher energy consumption during flight (Rotics et al. 2016) and poorer territory quality due to intraspecific competition, can result in longer flights and larger wintering areas to meet their energy requirements.
Studies on black kites, a soaring migratory raptor, have shown that survival rate, longevity, and reproductive performance were directly linked to spring departure dates, making it the most important aspect of their migration decisions. In black kites, a delay at departure made the birds increase their daily flight distance and reduce their number of stopover days. It also led to a significantly reduced recruitment probability (Sergio et al. 2014). A late departure may, therefore, be compensated for by a more energy-consuming migration, which possibly results in less energy available for competing for a mate and a breeding territory.
Departure dates during fall did not vary between ages. This may be because, as previously demonstrated, older individuals significantly influence the departure and route choice of first-year migrants, highlighting that there is most likely an overlap between juvenile and adult departure times. However, in white storks, younger birds usually migrate earlier in the season, mainly because they have lower flight speeds with higher energetic costs, requiring longer resting periods (Bontekoe et al. 2023; Rotics et al. 2016). We tracked the Black Storks only until the age of 3 and it is conceivable that shifts in the autumn departure dates, or other adjustments in migration patterns occur later in life. It is also possible that departure dates did not shift temporally because Black Storks perform dispersal flights in fall. Young white storks check for new breeding sites usually in spring after returning from their migration, allowing first-time migrants to depart shortly after fledging. Young Black Storks exhibit sometimes extensive exploratory movements after fledging before departing on their first migration (some even exhibiting northward movements), preventing first-time migrants from departing much earlier than the breeding birds.
Conclusion
Here we found a significant influence of age on migratory decisions in Black Storks. The noticeable shift toward earlier departure dates in spring is most likely driven by a need to reach breeding sites on time to compete for suitable nesting sites. These are still limited in Germany due to a lack of mature forests with large old trees. In addition, a reduction in wintering movements indicates that birds can benefit from experiences and spatial memories formed in previous year.
These ontogenetic developments are also important to quantify because of phenological and habitat changes induced by anthropogenic impacts. Spring is anticipated to start earlier, thereby affecting reproductive outcomes. Further, smaller wintering areas in older years highlight the new need to protect these learned habitats because storks rely on their experiences to efficiently perform their migrations and, thereby, increase their breeding output. Thus, additional research may be necessary to understand the factors determining migration patterns to be able to predict whether Black Storks can adapt to our human-changed environment.
Data, materials, and software availability
Data is stored in Movebank study (LifeTrack Black Stork).
References
Aikens EO, Nourani E, Fiedler W, Wikelski M, Flack A (2024) Learning shapes the development of migratory behavior. Proc Natl Acad Sci 121:e2306389121. https://doi.org/10.1073/pnas.2306389121
Bobek M, Hampl R, Peške L, Pojer F, Šimek J, Bureš S (2008) African Odyssey project - satellite tracking of black storks Ciconia nigra breeding at a migratory divide. J Avian Biol 39:500–506. https://doi.org/10.1111/j.0908-8857.2008.04285.x
Bontekoe ID, Hilgartner R, Fiedler W, Flack A (2023) The price of being late: short- and long-term consequences of a delayed migration timing. Proc R Soc B Biol Sci 290:20231268. https://doi.org/10.1098/rspb.2023.1268
Bundesamt für Naturschutz (2020) Bericht zur Lage der Natur 2020 (No. Steckbrief Nr. 4)
Campioni L, Dias MP, Granadeiro JP, Catry P (2020) An ontogenetic perspective on migratory strategy of a long-lived pelagic seabird: Timings and destinations change progressively during maturation. J Anim Ecol 89:29–43. https://doi.org/10.1111/1365-2656.13044
Cano LS, Tellería JL (2013) Migration and winter distribution of Iberian and central European black storks Ciconia nigra moving to Africa across the Strait of Gibraltar: a comparative study. J Avian Biol 44:189–197. https://doi.org/10.1111/j.1600-048X.2012.05824.x
Chevallier HY, Georges J-Y, Baillon F, Brossault P, Aurouet A, Le Maho Y, Massemin S (2010a) Influence of weather conditions on the flight of migrating black storks. Proc R Soc B Biol Sci 277:2755–2764. https://doi.org/10.1098/rspb.2010.0422
Chevallier HY, Le Maho Y, Baillon F, Duponnois R, Dieulin C, Brossault P, De Franclieu P, Lorge P, Aurouet A, Massemin S (2010b) Human activity and the drying up of rivers determine abundance and spatial distribution of Black Storks Ciconia nigra on their wintering grounds. Bird Study 57:369–380. https://doi.org/10.1080/00063651003678467
Chevallier HY, Le Maho Y, Brossault P, Baillon F, Massemin S (2011) The use of stopover sites by Black Storks (Ciconia nigra) migrating between West Europe and West Africa as revealed by satellite telemetry. J Ornithol 152:1–13. https://doi.org/10.1007/s10336-010-0536-6
de Grissac S, Börger L, Guitteaud A, Weimerskirch H (2016) Contrasting movement strategies among juvenile albatrosses and petrels. Sci Rep 6:26103. https://doi.org/10.1038/srep26103
Fayet AL (2020) Exploration and refinement of migratory routes in long-lived birds. J Anim Ecol 89:16–19. https://doi.org/10.1111/1365-2656.13162
Fiedler W, Feld W, Baumann F (2002) Der ELSA-Ring: ein neuartiger Markierungsring für Grossvögel. J Ornithol 143:247
Flack A, Aikens EO, Kölzsch A, Nourani E, Snell KRS, Fiedler W, Linek N, Bauer H-G, Thorup K, Partecke J, Wikelski M, Williams HJ (2022) New frontiers in bird migration research. Curr Biol 32:R1187–R1199. https://doi.org/10.1016/j.cub.2022.08.028
Gałecki A, Burzykowski T (2013) Linear Mixed-Effects Models Using R: A Step-by-Step Approach, Springer Texts in Statistics. Springer New York, New York, NY. https://doi.org/10.1007/978-1-4614-3900-4
Gerlach B, Dröschmeister R, Langgemach T, Borkenhagen K, Busch M, Hauswirth M, Heinicke T, Kamp J, Karthäuser J, König C, Markones N, Prior N, Trautmann S, Wahl J, Sudfeldt C (2019) Übersichten zur Bestandssituation. Vögel in Deutschland. Dachverband Deutscher Avifaunisten e.V, Münster
Jadoul G, Hourlay F, Toussaint A-C (2003) Suivi de la migration automnale de la Cigogne noire (Ciconia nigra) par télémétrie satellitaire. Aves 40:155–164
Janssen G, Hormann M, Rohde C (2004) Der Schwarzstorch: Ciconia nigra, 1. Aufl, Die Neue Brehm-Bücherei. Westarp Wissenschaften, Hohenwarsleben
Kays R, Davidson SC, Berger M, Bohrer G, Fiedler W, Flack A, Hirt J, Hahn C, Gauggel D, Russell B, Kölzsch A, Lohr A, Partecke J, Quetting M, Safi K, Scharf A, Schneider G, Lang I, Schaeuffelhut F, Landwehr M, Storhas M, Van Schalkwyk L, Vinciguerra C, Weinzierl R, Wikelski M (2022) The Movebank system for studying global animal movement and demography. Methods Ecol Evol 13:419–431. https://doi.org/10.1111/2041-210X.13767
Kokko H (1999) Competition for early arrival in migratory birds. J Anim Ecol 68:940–950. https://doi.org/10.1046/j.1365-2656.1999.00343.x
Larue M, Boos M, Brossard C, Baillon F, Boutteaux J-J, Bernard J, Brossault P, Petit O, Chevallier D (2016) Migration of juvenile Black Storks: stay with the family or go alone? Ringing Migr 31:74–76. https://doi.org/10.1080/03078698.2016.1190616
Lewis MA, Fagan WF, Auger-Méthé M, Frair J, Fryxell JM, Gros C, Gurarie E, Healy SD, Merkle JA (2021) Learning and Animal Movement. Front Ecol Evol 9:681704. https://doi.org/10.3389/fevo.2021.681704
Nilsen EB, Pedersen S, Linnell JDC (2008) Can minimum convex polygon home ranges be used to draw biologically meaningful conclusions? Ecol Res 23:635–639. https://doi.org/10.1007/s11284-007-0421-9
Rotics S, Kaatz M, Resheff YS, Turjeman SF, Zurell D, Sapir N, Eggers U, Flack A, Fiedler W, Jeltsch F, Wikelski M, Nathan R (2016) The challenges of the first migration: movement and behaviour of juvenile vs. adult white storks with insights regarding juvenile mortality. J Anim Ecol 85:938–947. https://doi.org/10.1111/1365-2656.12525
Sergio F, Tanferna A, De Stephanis R, Jiménez LL, Blas J, Tavecchia G, Preatoni D, Hiraldo F (2014) Individual improvements and selective mortality shape lifelong migratory performance. Nature 515:410–413. https://doi.org/10.1038/nature13696
Acknowledgements
We thank all the people and organizations who helped during fieldwork. This work was supported by the Max Planck Society. AF was supported by the German Research Foundation (DFG, Emmy Noether Fellowship 463925853), the Hans und Helga Maus-Stiftung, and the James Heineman research award of the Minerva Stiftung.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
FF and AF conceived the research question, analyzed the data and wrote the paper, MW and GH developed the biologgers, MW funded the research, GH and CR collected the data. All other authors provided critical feedback that improved earlier versions of the manuscript.
Corresponding author
Ethics declarations
Competing Interest
The authors declare no competing interests.
Additional information
Communicated by F. Bairlein.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fisel, F., Heine, G., Rohde, C. et al. Influence of age on spatial and temporal migratory patterns of Black Storks from Germany. J Ornithol (2024). https://doi.org/10.1007/s10336-024-02170-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10336-024-02170-3