Abstract
Individual acoustic monitoring (IAM), based on the analysis of vocal cues, is particularly suitable for the identification and tracking of birds with temporally stable song or call characteristics. Unlike mark-recapture methods, IAM does not require the physical manipulation of individuals, which can have long-lasting behavioural effects. So far, IAM has usually focused on males, as singing females tend to be overlooked in temperate zones. Here, we evaluated the suitability of IAM for both sexes in an isolated population of the Tawny Pipit (Anthus campestris, Motacillidae), a migratory Palearctic species critically endangered in Central Europe, for which female singing has been occasionally documented. We confirmed that songs of all 101 studied individuals, both males and females, were individually distinct. Most individuals used only a single song type in their repertoires, with only three males using two. Of 45 ringed males (that could be unambiguously recognized visually), only two changed their song structure to some extent, either within or between seasons. Multiple individuals often sang structurally similar song types, which nevertheless consistently differed in minor characteristics; such differences were detectable by visual inspection and also affected quantitative analyses of song similarity. Songs sung by females did not have any apparent sex-specific characteristics. Unlike previously suggested, females did not adapt their vocalization to their breeding partner, and we presume their song is also temporally stable. Our findings support IAM as a reliable approach for studying the behaviour and ecology of this passerine species with a small repertoire and simple songs.
Zusammenfassung
Wer singt denn da? Analyse des individuellen akustischen Monitorings von Männchen und Weibchen des Brachpiepers, eines Zugvogels mit einfachem Gesang.
Individuelles akustisches Monitoring (IAM) auf der Basis von Lautäußerungen ist besonders geeignet für die Identifizierung und Verfolgung von Vögeln mit über längere Zeit hinweg stabilen Gesangs- oder Rufmerkmalen. Im Gegensatz zu Wiederfangmethoden erfordert das IAM keine physische Handhabung der Einzeltiere mit möglicherweise daraus folgenden, länger anhaltenden Auswirkungen auf ihr Verhalten. Bisher hat sich das IAM in der Regel auf Männchen konzentriert, da singende Weibchen in den gemäßigten Zonen eher übersehen werden. In dieser Untersuchung bewerteten wir die Eignung von IAM für beide Geschlechter in einer isolierten Population des Brachpiepers (Anthus campestris, Motacillidae), einer in Mitteleuropa stark bedrohten paläarktischen Zugvogelart, für die gelegentlich der Gesang auch von Weibchen dokumentiert worden ist. Wir stellten fest, dass die Gesänge aller 101 untersuchten Einzeltiere, sowohl die der Männchen als auch die der Weibchen, individuell unterschiedlich waren. Die meisten Tiere hatten nur einen einzigen Liedtyp in ihrem Repertoire, nur drei Männchen hatten zwei Liedtypen. Von 45 beringten Männchen (die visuell eindeutig erkannt werden konnten) änderten nur zwei ihre Gesangsstruktur in gewissem Umfang, entweder innerhalb oder zwischen den Jahreszeiten. Mehrere Individuen sangen oft Liedtypen, die sich in ihrer Struktur ähnelten, aber durchweg in kleineren Merkmalen unterschieden; solche Unterschiede waren durch visuelle Kontrolle nachweisbar und wirkten sich auch auf die quantitativen Analysen der Gesangsähnlichkeit aus. Die Gesänge der Weibchen wiesen keine offensichtlichen geschlechtsspezifischen Merkmale auf. Anders als bislang angenommen, passten die Weibchen ihren Gesang nicht an den Brutpartner an, und wir vermuten, dass ihr Gesang auch über längere Zeit hinweg stabil ist. Unsere Ergebnisse unterstützen das IAM als einen zuverlässigen Ansatz für die Untersuchung des Verhaltens und der Ökologie dieser Sperlingsart mit einem kleinen Repertoire und einfachen Gesängen.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bird songs have multiple important roles, especially for intraspecific interactions such as mate attraction and territory defence. In addition, birds often rely on vocalizations to recognize conspecifics from others (Matyjasiak 2005; Hick et al. 2016; Hodgson et al. 2018; Darolová et al. 2020). Recognition might also take place at the individual level if birds show consistent individually distinctive patterns in their acoustic signals (e.g., Terry et al. 2005; Osiejuk et al. 2007; Petrusková et al. 2016). In this way, birds can discriminate between familiar and unfamiliar conspecifics in different social contexts (e.g., neighbour vs. stranger discrimination, parent–offspring recognition, kin recognition; Lambrechts and Dhondt 1995). Not only do vocal signatures play an important role in social interactions, but under some circumstances they can also be effectively used to monitor bird individuals (e.g., Blumstein et al. 2011; Petrusková et al. 2016, 2021; Sebastián-González et al. 2018; Pérez-Granados et al. 2019).
Individual acoustic monitoring (IAM) is a non-invasive monitoring approach based on the analysis of vocal cues of individuals, which is particularly suitable for bird taxa that are highly vocally active (reviewed in Terry et al. 2005). Acoustic monitoring does not require the manipulation of trapped individuals, unlike more traditional mark-recapture methods (e.g., use of mist nets or clap traps for capturing and subsequent ringing). Though these catch-and-ring techniques do not necessarily have a negative effect on bird survival and fitness (e.g., Calvo and Furness 1992; Clewley et al. 2018; Petrusková et al. 2021), playback luring plus capturing and handling may have cumulative effects on the subsequent behaviour of passerines (Linhart et al. 2012; Budka et al. 2019; Oñate-Casado et al. 2021). In contrast, due to its non-invasive approach IAM can be used on wild species with fewer concerns about impacts on the behaviour of target individuals (reviewed in Terry et al. 2005), and might even provide more accurate information about behaviour or population dynamics (e.g., Laiolo et al. 2007; Petrusková et al. 2016). Besides being a good alternative for species that are sensitive to capturing and handling, IAM is also suitable for studying birds that are difficult to spot because they inhabit areas with poor visibility, such as dense vegetation (e.g., Hobson et al. 2008; Celis-Murillo et al. 2009; Petrusková et al. 2016).
Despite the advantages that IAM offers in identifying and tracking birds, it has not been used very frequently. Most of the existing studies have focused on non-passerine species with simple vocalizations, such as Great Bitterns (Gilbert et al. 2002), ducks (Volodin et al. 2005), and owls (Galeotti and Sacchi 2001; Tripp and Otter 2006; Grava et al. 2008; Choi et al. 2019). Studies on songbirds, whose songs develop under the influence of vocal learning and are culturally transmitted, have also mainly concerned species with simple song structures (e.g., Laiolo et al. 2007; Vögeli et al. 2008; Adi et al. 2010). This is because the implementation of IAM for passerines with higher song complexity is time-intensive, especially at the beginning, when the temporal stability and individual uniqueness of target species’ song must be evaluated (Petrusková et al. 2016). When vocal signatures are temporally stable, IAM can be effectively used to follow songbirds over time and provide information both within a single season (e.g., population density) and over several years (e.g., survival rates in sedentary species, return rates in migratory species, changes in territorial dynamics and replacements). This has been reported for species with simple vocalizations such as the calls of Dupont’s Lark (Chersophilus duponti; Laiolo et al. 2007; Vögeli et al. 2008), songs of Mexican Ant-thrushes (Formicarius moniliger; Kirschel et al. 2011) and Ortolan Buntings (Emberiza ortulana; Adi et al. 2010), as well as in a species with a substantially more complex song structure, the Tree Pipit (Anthus trivialis; Petrusková et al. 2016).
Most, if not all existing IAM studies on passerines have only targeted male individuals. In songbirds, songs are traditionally considered to be primarily produced by males (Catchpole and Slater 2008). However, recent reviews have shown that female song is widespread among songbird species (Passeri). Odom and Benedict (2018) reported (based on data from Webb et al. 2016) that sex-specific song information is available for only 27% of all songbird species; of those, however, almost two thirds have females that sing at least occasionally. A review by Garamszegi et al. (2007) indicated that approximately 40% of European songbird species have females that may sing, while among North American passerines, females seem to sing in 43% of duetting and 34% of non-duetting species (Benedict 2007). Nonetheless, there is still a lack of documentation on female songs for most songbirds, especially in temperate areas where studies on female songs are much less prevalent than in the tropics (Langmore 1998; Odom et al. 2014; Hall and Langmore 2017).
Part of this above-described discrepancy may be due to the fact that in temperate regions, songbird females can be relatively quiet and visually cryptic (Price 2019). That leads to the increased detection of songbird males, which usually are more vocally active (Bennett et al. 2019). Additionally, birds in these regions only sing intensively for a short period during the breeding season, and this time window might be even shorter for females (e.g., McDonald and Greenberg 1991). Many species are also monomorphic, which makes it difficult to distinguish between the sexes (Langmore 1998; Hahn et al. 2013; Odom and Benedict 2018), and singing females, even when in fact observed, may be assumed to be males (Sierro et al. 2022). Despite this, however, in some temperate songbird species it has been shown not only that females sing but also that songs can be used to discriminate between males and females (Yamaguchi 1998). In such cases, acoustic monitoring might also provide reliable information about the sex of the vocalizing individuals.
The Tawny Pipit (Anthus campestris) is one of the temperate songbird species for which field studies have reported that both sexes sing (Neuschulz 1986; Alström and Mild 2003; Calero-Riestra and García 2019). Although the Tawny Pipit song is simple, males of this species apparently possess individually unique song types (Neuschulz 1986; Osiejuk et al. 2007), and Neuschulz (1986) mentioned that they seem stable between two consecutive seasons. The individuality and stability of their song would make this pipit species suitable for IAM. In the same study, however, Neuschulz (1986) also claimed that females copy the song of the males they are paired with. If true, this would suggest that female Tawny Pipits are able to adapt their vocalization over time. However, there are no publications following the preliminary report by Neuschulz (1986), and all his conclusions were only illustrated by a few spectrograms of male and female songs.
In our study, we performed a detailed analysis of inter-individual and temporal variation of song from an isolated Central European Tawny Pipit population. This way, we assessed the suitability of IAM for tracking both males and females of this territorial migratory songbird. Further, we compared the song structure of males and females, and for the available data on male–female breeding pairs, we also evaluated the claim of Neuschulz (1986) that females copy the song of their partner.
Methods
Study species and fieldwork
The Tawny Pipit (Anthus campestris) is one of over 40 globally distributed species in the genus Anthus. These sub-Saharan migrants are widespread in the Palearctic, and on a whole-European scale their conservation status is categorized as “Least Concern” (IUCN 2022). However, they are undergoing severe population declines, especially in central and western Europe, due to the loss of suitable habitats (Briedis et al. 2016; Tyler and Christie 2016). Because of this, the species is categorized differently in the regional red lists of many European countries (e.g., Switzerland: Endangered, Czechia: Critically Endangered, Netherlands: Regionally Extinct; www.nationalredlist.org).
During the breeding season, Tawny Pipits inhabit dry, sandy steppe-like habitats, and require bare ground with sparse vegetation cover for nesting (Beran et al. 2018; Calero-Riestra and García 2019). They are socially monogamous and cryptically coloured with no sexual dimorphism (Alström and Mild 2003). The song should thus be critical for male success in territorial defence and mate attraction. According to a few published reports (Neuschulz 1986; Alström and Mild 2003; Calero-Riestra and García 2019), females of this species also occasionally sing, mainly in flight while approaching and leaving the nest with nestlings (see the “Results” for more details).
Our study was conducted on the only remaining population of Tawny Pipits in Czechia (Central Europe), located in an active brown coal mine area in the Ústí nad Labem region (50.48–50.56 °N, 13.48–13.58 °E; Briedis et al. 2016). Suitable habitats for the species are mostly found at the edges of open-cast mines, which host up to 200 breeding Tawny Pipit pairs (Briedis et al. 2016; Beran et al. 2018). The population has been intensively monitored for several years (most intensively between 2015 and 2017), and the birds captured during the survey were equipped with a ring with a unique alphanumeric code allowing their visual identification from a distance. Individuals in our study were recorded from two coal mines: ČSA (50.54 °N, 13.53 °E) and Vršany (50.49 °N, 13.54 °E).
Singing individuals were repeatedly recorded during four different breeding seasons (from May to mid-July 2015, 2016, 2017 and 2021) using a Sennheiser ME-67 shotgun directional microphone connected to a Marantz PMD 661 recorder. We noted the date, time, geographic coordinates and bird behaviour for each recording and observation. For those individuals that were ringed at the time of recording, we additionally took note of their ring code, if visible. For both sexes, most of the recordings were obtained in the seasons 2016 and 2017 (Table 1).
We attempted to record at least 3 min of singing bouts from each male; however, substantially shorter recordings of good quality were also considered fully adequate because male Tawny Pipits generally use a single song type (Osiejuk et al. 2007). Since female pipits sing rarely, the number of songs for females was much more limited, and we analysed all songs of sufficient quality that were available.
Individual identification and song measurements
Individual identification of Tawny Pipit males and females was based on ring observations and/or song recording analyses, following the method of Petrusková et al. (2016). We identified non-ringed birds as males or females based on their behaviour and pairing status with ringed individuals, if known. In total, we recorded 81 males and 20 females (Table 1), 45 (56%) and 8 (40%) of them ringed, respectively. From all the recordings, we obtained the songs of nine breeding pairs formed by nine female and eight male individuals (one ringed male was paired with two different ringed females in different breeding seasons).
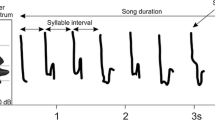
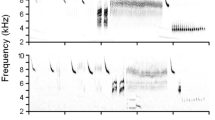
To characterize individual repertoires, we first visually inspected all recordings in the program Avisoft-SASLab Lite 5 (Specht 2007) with the following set of parameters: FFT length 256; Frame 100%; and Hamming window. We created standardized spectrograms that were visually inspected and catalogued as distinct song types according to differences in their element structure. Following Lachlan (2011), we defined an “element” as a discrete unit of continuous sound separated from other elements by silence, and a “song” as the sum of all the elements it contains. Minor but highly consistent differences in element shapes between song types (i.e., those consistently retained across different songs and recordings), observable in the spectrograms by an experienced user, were considered sufficient to classify songs as distinct (see Figs. 1, S2, S3).
Spectrograms illustrating the variation of song types of six Tawny Pipit individuals from the studied population (A, D–F: males, B–C: females). A illustrates the temporal stability of songs sung by the same bird on different dates across two years. A, B and E are distinct but structurally similar song types sung by different individuals (two males and one female), while B, C and D demonstrate structurally different song types sung by different individuals. Two distinct song types sung by the same male are shown in F. The position of all song types shown here is indicated in the dendrogram of song similarity in Fig. 3A. More spectrograms are provided in Supplementary Figures S1 (different song types sung by the same males), S2 (song types sung by individuals paired together) and S3 (spectrograms representing the overall variation in the dataset, highlighting “song families”, i.e., groups of structurally similar song types)
Additionally, we used the bioacoustics software Luscinia 2.20.03.11.01 (http://github.com/rflachlan/luscinia) to measure and quantitatively compare acoustic characteristics for representative recordings of each song type observed in the population. When needed, we removed the noise and improved the signal by altering the dereverberation (100%), dereverberation range (50 ms), dynamic range (40 dB) and high pass threshold (1000 Hz) settings. We then characterized the parameters for every single element within a song, only considering recordings with songs of sufficient quality (i.e., those in which song elements could be clearly distinguished from the background noise).
Song analysis and validation of the method
We compared in a pairwise manner the structure of all song types using the “dynamic time-warping algorithm” (DTW) in Luscinia. DTW aligns two acoustic (time-dependent) sequences, which may vary in speed, and calculate dissimilarity scores based on the Euclidean distance between their acoustic features. This is achieved by warping the time and/or frequency axis until an optimal match between those two sequences is found. Weights and parameters of the DTW analyses (for their explanation, see Luscinia documentation) were set as follows: time (5), fundamental frequency (1), fundamental frequency change (1), FF norm (1), compression factor (0.25), minimum element length (2), time SD weighting (1), ArcTan transform weight for frequency slope (0.02), cost for alignment error (0.2), stitch threshold (100 ms), and maximum warp (100%). Fundamental frequency was log-transformed.
Through the DTW employed by Luscinia, we obtained a pairwise matrix of song dissimilarities, which was used to visualize and interpret the patterns of acoustic similarity between analysed songs as well as in formal tests of specific hypotheses about song similarity. To visualize the patterns in a dendrogram, we carried out a hierarchical clustering analysis using a unweighted pair group method with arithmetic mean (UPGMA).
Assuming that male Tawny Pipits possess individual acoustic signatures (Osiejuk et al. 2007), different songs from the same bird should be acoustically more similar to each other than songs from different ones, at least for males. Hence, we inspected results from Luscinia to evaluate whether different song types clustered according to the male and female individuals they belonged to. Firstly, we successfully tested that the quantitative approach was able to cluster songs from known birds on a subset representing ringed individuals only, in which songs were unambiguously assigned to a particular male. This subset was formed by three songs from each bird, recorded in the same time period (within one day or two consecutive days).
Then, we evaluated the temporal song stability in known (ringed) males by comparing the acoustic similarity between songs from the same bird at different moments within the same breeding season, as well as between seasons. We included up to three periods for a given year, at least a week apart, and spread as much as possible in time from each other. If available, we tried to cover the beginning (until mid-May), middle (mid-May to mid-June) and end (mid-June to late July) of the season, but we occasionally also included recordings from the same period if taken more than one week apart. Finally, for returning birds, we also analysed songs representing different years (an additional song for each extra season in which the returning bird was recorded).
After confirming the assumptions of song individuality and stability on a subset of recordings from ringed individuals, we also considered recordings from non-ringed males and females for further analyses. As with ringed individuals, we tried to process at least three songs per non-ringed bird, and cover within- and between-season temporal variation. However, in the case of three females and one male, we only included two songs due to the scarcity of available recordings of sufficient quality.
Based on this comprehensive dataset, we generated a single circular UPGMA dendrogram depicting the acoustic similarity of representative songs for all individuals (Figs. 2, 3, S3), which was visualized and annotated in iTOL 6 (Letunic and Bork 2021). On this dendrogram, we highlighted features of interest (e.g., ringed birds, males vs. females, individuals paired together), as well as indicated groups of multiple song types (representing at least four birds) clustering together below a certain arbitrary threshold (“song families”; Figs. 3A, S3). These groups were also highlighted in a multidimensional scaling plot (Fig. 4) that was based on the same pairwise dissimilarity matrix as the dendrogram.
Statistical analyses
In order to corroborate the results obtained through visual inspection and clustering analyses, we applied Mantel tests (with 10,000 random replicates) to test specific hypotheses about patterns of song (dis)similarity at the individual level (within and between individuals) and sex level (within and between sexes, within and between male–female pairs). We created pairwise binary dissimilarity matrices to represent individuals, sex, and couple membership, using 0 and 1 to denote songs from the same and different individual/sex/couple, respectively. These matrices thus characterized alternative hypotheses of a priori models that were tested by randomization of the dataset.
The selection of songs to be included in the tests depended on the tested hypothesis. When evaluating how consistent the individually unique songs are, each song type was represented by three renditions (except for five above-mentioned individuals with two songs only). For all other comparisons, each song type was represented just once. This way, we performed Monte Carlo tests to investigate possible relationships between group membership and the acoustic dissimilarity values obtained from the DTW algorithm in Luscinia. Significant results of the test can be interpreted as the acoustic similarity being higher between members of the same group than between members of different groups. The Mantel tests were run in R 4.1.3 (R Core Team 2021) using the package vegan (Dixon 2003).
Results
Male and female singing patterns
Based on our field observations, Tawny Pipit males frequently sing throughout most of the breeding season, usually from higher perches in their territory or during flight, often with many songs in a sequence. Females sing much more rarely than males, and most females were not recorded or heard singing at all. Of those that did sing, we noted their singing activity only occasionally, and these were usually no more than 3–5 flight songs while approaching or leaving the nest. If singing occurred while flying to the nest, females tended to land away from the nest (ca. 10–30 m) and approached it on the ground. We mainly observed females singing when feeding older nestlings (from the age of ca. 5 days), but occasionally also during the incubation period. Accordingly, the period in which the recordings of females were obtained was short (from June to July).
Both male and female Tawny Pipits sing relatively short songs (on average about 0.5 s) composed of several elements forming a simple song type (Fig. 1). Most of the studied individuals sang only one song type, but three males (i.e., 3.3%) produced two different song types (Figs. 2B, S1A) that were both consistently captured in recordings of those individuals, either singing multiple repetitions of the same song type before switching to the other one or alternating between short sequences of 1–3 songs of each type.
Individuality and the temporal stability of songs
The song types were individually distinct. When analysing songs of ringed birds, we never encountered identical song types sung by different individuals. However, multiple birds often used structurally similar song types, only differing from each other in minor structures (Figs. 1, S2). These resemblances were reflected in lower dissimilarity scores of such song types, which thus formed distinct clusters of “song families” in the UPGMA dendrogram (Figs. 3A, S3). Songs belonging to clusters also formed distinct aggregates when their variation was visualized in the multidimensional scaling plot (Fig. 4).
UPGMA dendrogram of song similarity based on the output of the dynamic time warp algorithm implemented in Luscinia software. Ringed birds are denoted by blue and non-ringed by brown branches; males by solid and females by dashed branches. The year of recording (2015–17, 2021) is abbreviated by its last digit, numbers in different colours mean different parts of the respective season. Segments in the outer ring indicate songs sung by the same individuals (whose codes are provided in Fig. S3). Specific symbols in the ring mark those individuals for which not all songs formed a single cluster in the dendrogram: males singing two song types (denoted in colour in Fig. 3C) are marked by a plus sign, the male 7H.16 (dark grey in Fig. 3C) by a triangle, interspersed songs of males NA.16 (black in Fig. 3C) and SX.17 by dots. Stars indicate adjacent songs of the same individual that do not cluster together due to variation in recording quality
UPGMA dendrograms (same topology as in Fig. 2) with highlighted features of interest. “Song families”, i.e., structurally similar songs of at least four individuals that cluster together under an arbitrarily chosen threshold (marked by a dashed ring), are marked by colour in A; letters along the dendrogram edge indicate the position of song types shown in Fig. 1. Representative spectrograms across these “song families” are shown in Fig. S3. Male songs (black) and female songs (red) are highlighted in B, males with different or variable song types in C, and paired individuals (males by solid and females by dashed lines in corresponding colour) in D. Spectrograms of song types highlighted in C and D are provided in Figs. S1 and S2, respectively
A multidimensional scaling plot as an alternative visualization of the variation in song similarity among studied Tawny Pipit individuals, based on the dissimilarity matrix provided by the dynamic time warp algorithm implemented in Luscinia software. Each dot indicates one song; colours match those of the “song families” in Figs. 3A, S3. Grey open dots are songs not assigned to any family, which are scattered across the dendrogram (Fig. 3A)
In most cases, song renditions of the same singing individuals (whether male or female) were alike, regardless of whether these originated from the same recording, from recordings obtained in different parts of the same season, or from different years (Figs. 1, S2). In two out of 45 ringed males (NA.16 and 7H.16), however, we observed within-individual temporal variation (Fig. S1B). Specifically, the terminal part of songs of the male NA.16 varied in the duration and number of repeated elements, both between and within recordings. In late June 2016, this male had a tendency to sing longer variants than in late May, 28 days earlier. Songs of the male 7H.16 differed between two subsequent breeding seasons by an extra terminal element that was consistently absent in 2017 but almost always present in 2016 (except for one single song in one of the 2016 recordings, which also lacked this element).
Accordingly, the clustering in the UPGMA dendrogram (Figs. 2, 3, S3) was mostly arranged by individuals. For 70 out of 81 males and all 20 females, all songs of the same bird clustered together (Figs. 2, S3). The exceptions included the three males singing two distinct song types, as well as the two above-mentioned males that showed temporal variation in song structures (one of the songs of NA.16 was clustered with structurally similar songs of male SX.17, and songs of 7H.16 from consecutive years were separated in two clusters representing “song families” differing in the terminal elements; Figs. 3C, S3). All remaining cases where one of the songs of a particular individual was not clustering with the others in the dendrogram (star symbols in Fig. 2) were apparently caused by variation of the overall recording quality, not structural differences among the songs themselves. The prevailing within-individual consistency of songs was reflected in the highly significant results of the test that evaluated the differences between songs sung by the same individual and the others (nsongs = 242, nbirds = 81, p < 0.001).
For all three males singing two different song types, both types were individually distinct but not similar to each other (Figs. 3C, S1A). Correspondingly, the dissimilarity scores of song types sung by the same male were comparable to those picked at random from all males (nsongs = 251, nbirds = 81, p = 0.99).
Female songs
As was found for the males, females also had individually unique song types. Some of these song types were highly distinct, others closely similar to either that of one of the males or of another female (Figs. 2, 3B). Only one female (T3.16F) was recorded in two different seasons (during which it was paired with the same male, 3L.15), and its song did not vary (Fig. S2). There was no song feature that could be considered sex-specific; male and female song types were interspersed in the dendrogram. However, the pattern was not entirely random due to the occasional clustering of two females directly next to each other or within the same “song family” (Figs. 2, 3B, S3). This was reflected in the results of the test evaluating whether the acoustic similarity of songs of the same sex is higher than between sexes (nmale birds = 81, nfemale birds = 20, p = 0.06).
When nine confirmed breeding pairs were considered, songs of the male and the respective female were usually not located close to each other in the dendrogram (Figs. 3D, S2, S3). Only for one of the pairs (female 02.16F and male 74.15) were the partners’ songs more similar to each other than to songs of other birds (Figs. 3D, S2). However, this was not the case for the remaining pairs; other birds with no social bond often sang songs similar to those of a given male or female (Fig. S2). Correspondingly, the Mantel test indicated that the acoustic similarity of a female’s songs was not higher to songs of her partner than to songs of a randomly chosen male in our recorded study population (ncouples = 9, nsongs = 90, nfemale birds = 9, nmale birds = 81, p = 0.59). Interestingly, the songs of two ringed females (NK.16F and YT.17F) that were paired with the same male (HT.16) in different years (2015 and 2016–17, respectively) were very similar, but they clearly differed from this male’s song (Figs. 2D, S2).
Discussion
Our findings confirm that the songs of male and female Tawny Pipits are individually distinct, although consistent differences between similar songs are often minor, and require careful assessment of the respective spectrograms. Identification based on song recordings was consistent with the observation of ring codes, indicating that when analysing the spectrograms with care, we could distinguish individuals of both sexes with a considerably high level of certainty. In the case of males, these results support the claims of Neuschulz (1986) and Osiejuk et al. (2007). As the male songs were confirmed to be temporally stable both within and between seasons, we may conclude that the Tawny Pipit, as a migratory passerine species with a small repertoire and low song complexity, seems suitable for IAM. This methodological approach may be thus used for studying the behaviour and ecology of the species (see Oñate-Casado et al. 2021).
Traditional capture-mark methods involve physical manipulation of the individuals, which might have subsequent negative effects on the health (Marco et al. 2006; Mulcahy et al. 2011; Spotswood et al. 2012) or the behaviour of the studied animals (Linhart et al. 2012; Budka et al. 2019; Oñate-Casado et al. 2021). In a previous study on the same population (Oñate-Casado et al. 2021), we found that male Tawny Pipits associate playback stimulation with capturing events and remember such an experience over a period comparable to their life span in the wild. This implies that long-term negative associations might affect the results of studies that use similar methods for re-capturing individuals (e.g., for retrieving data loggers, estimating survival and return rates, etc.). IAM offers the great advantage of being non-intrusive, and thus can be used on Tawny Pipits with fewer concerns about impacts on their behaviour. As reported in a congeneric species, the Tree Pipit (Petrusková et al. 2016), IAM seems more reliable than ring observations for determining the density of singing individuals as well as estimating their return rates and within-season spatial territorial dynamics. It may also reveal complementary or more detailed information about species behaviour than classical methods (Laiolo et al. 2007; Kirschel et al. 2011; Petrusková et al. 2016).
Our approach was very time-efficient in the field. We initially intended to spend a maximum of 5 min to obtain a recording of sufficient quality for a spontaneous singing male. However, we usually just needed one minute once the male started singing to record enough songs for reliable identification. This took longer (ca. 10–30 min) for singing females, since they sing much less frequently than males, have only short song sequences, and apparently only sing in a specific context (see also Neuschulz 1986; Alström and Mild 2003; Calero-Riestra and García 2019). However, even this amount of time was substantially lower than usually required to unambiguously read ring codes, in particular for females whose behaviour is very secretive. Our method was also not overly time-consuming from a technical perspective: a dataset of three recorded songs per individual was sufficient to characterize an individual’s acoustic signature, and to assess with certainty whether an individual was already known or not. Once the individual’s song structure had been characterized, even a single well-recorded song was sufficient for its identification.
Our visual observations of differences in song mostly corresponded to the dissimilarity scores obtained from the DTW algorithm implemented in Luscinia. As a result, songs clustered correctly according to individuals for most Tawny Pipits, both males and females. However, for about 10% of individuals, the analysed songs were not arranged in a single cluster (usually, one song was slightly separated from the others). After visually examining the respective spectrograms, we concluded that such songs did not differ from the others sung by the same bird in structure but rather in recording quality, which affected the precision of designating individual song elements in the software and thus the corresponding dissimilarity scores. This highlights that in the case of very similar songs, the DTW algorithm as applied by us seems sensitive to even small differences in fine element structure. While the clustering based on dissimilarity scores alone was surprisingly efficient and may be applicable on large-scale datasets, repertoire-based IAM still requires the assessment and/or correction of results by experienced users. This is particularly true for highly similar song types that only differ in tiny but consistent features. In our study population, we recorded several such “song families” that required careful inspection to reveal individually specific characteristics (Fig. S3).
The songs of two males (7H.16 and NA.16) did not all cluster together in the dendrogram due to clear structural differences rather than recording quality. These differences might be related to the song learning process. Tawny Pipits seem to be age-limited learners (also known as closed-ended learners; Brainard and Doupe 2002). In such bird species, the song is learned for a limited period during the early stages of life, presumably during their first and sometimes the second year of life. During that time, birds memorize a conspecific song model from one or more tutors, and they then start producing a generic, variable and quiet vocalization called a subsong (Brenowitz et al. 1997). They then go through a period in which they produce, repeat, reorder, add and drop vocal elements until they eventually produce a stereotyped or “crystallized” song that resembles the one memorized from tutors (Immelmann 1969; Marler 1991; Williams 2004).
All but one song of male 7H.16 recorded in 2016 contained an extra element that completely disappeared in all songs recorded in 2017. Similarly, the prevailing song structure of male “NA” differed between late May and late June 2016. Since birds tend to produce more sounds during the song learning phase, with several plastic changes occurring over many weeks and vocal performances before reaching a crystallized song (Mooney et al. 2008), it is possible that both males were young (presumably one year old) in 2016 and thus still undergoing song stabilization.
As in previous studies studying the song of male Tawny Pipits (Neuschulz 1986; Osiejuk et al. 2007), we found that most males in our study population sang a single song type. However, we recorded a second song type in the vocalization of three males, which, to our knowledge, has not yet been reported for this species. Both song types sung by the same male were unique but their structures were not acoustically similar to each other. Osiejuk et al. (2007) suggested that there is no sexual selection towards an increased repertoire size for this species since its repertoire is formed by a single song. The proportion of two-song males in our study (3/81; 3.7%) seems too low to have resulted from selection, so this could simply be a learning issue with no adaptive value. Two-song repertoires are not exclusive to male Tawny Pipits from our study site since we have also observed this phenomenon in other populations in Spain and Italy, always in a proportion comparable to the Czech population (J. Oñate-Casado, A. Petrusek, T. Petrusková, pers. obs.). A more intensive study of such individuals would be needed to determine whether there are any consistent patterns in the use of the different song types, or other behavioural differences from males that only sing one song type.
Females in our study site always sang under the same conditions (i.e., 3–5 flight songs when arriving to the nest, and rarely when leaving it), supporting what was previously described by Neuschulz (1986), Alström and Mild (2003), and Calero-Riestra and García (2019). Notably, however, our study provides the first evidence of female Tawny Pipits singing individually unique song types. We additionally observed that songs of males and females are neither distinguishable by ear in the field nor structurally different between sexes. Apparently, sexing Tawny Pipits based on the song acoustic structures alone seems impossible, although we cannot dismiss that pipits themselves may recognize the sex of conspecifics based on song. However, females seemingly sing under the same specific circumstances across several European populations, and we never observed ringed males singing while feeding (V. Beran and M. Porteš, pers. obs.). Hence, we believe that it is possible to deduce the sex of singing non-ringed Tawny Pipits by considering the behavioural and seasonal context in which their song is produced.
Female vocalizations when leaving and/or arriving at the nest, usually calls, have only rarely been reported for other temperate passerine species (Yasukawa 1989; McDonald and Greenberg 1991; Grunst et al. 2014), and such female calls have been suggested to prevent mistaken attacks by their mates in habitats with low visibility. The same function for the female song in Tawny Pipits was proposed by Neuschulz (1986); however, they breed in open habitats so males can easily use visual cues to identify their partners. We observed that our female pipits tend to land a certain distance (a few to tens of meters) from the nest, and then approach it by walking, just like Swamp Sparrows and Meadowlarks (McDonald and Greenberg 1991). Considering this, females may also use songs to alert their mate about an unprotected nest, to which they return by sneaking silently through the sparse vegetation in order to counteract the increased risk of nest predation associated with their singing activity.
Although always observed in the same context, and thus likely functional, female singing in Tawny Pipits remains rare. It is possible that the female propensity to sing is influenced by some intrinsic factors, such as age or hormonal shifts. For example, increased testosterone levels promote singing in female European Robins (Kriner and Schwabl 1991; Schwabl 1992). The level of this hormone in females often fluctuates temporally (Ketterson et al. 2005), and may differ substantially among individual females in various monogamous songbird species (e.g., Moreno et al. 2014; Těšický et al. 2022). Unfortunately, we lack any specific information about the status, age or physiological state of the females recorded in our study.
Although Neuschulz (1986) reported that Tawny Pipit females copy the song of the male they are paired with, from our observations it appears that most females have songs with a structure that clearly differs from those of their mates. Indeed, the male and female had very similar songs in only one of our recorded pairs. In contrast, we also found one male paired consecutively with two females, each in different years, and the songs of those two females were very similar to each other but not to that of their male partner. Furthermore, numerous males and females sang structurally similar songs irrespectively of their social relationship. We assume these “song families” result from the learning and introduction of fine individual-specific variation, and thus pairing of mates with similar songs arises by chance. Unfortunately, the results and spectrograms presented by Neuschulz (1986) lack sufficient detail, so it is not possible to check whether there were minor song differences between males and females of the pairs he recorded, or if other individuals in that population also sung similar songs. Although our results indicate that female Tawny Pipits do not adapt their song structure to that of their actual mate, we cannot rule out that they copy the song of their first nesting partner. However, it seems at least as plausible that females learn from other males in the population.
To determine whether female vocalization is influenced by their partners’ songs or not, it is necessary to record the songs of paired birds—ideally also including females that switch partners—in consecutive years. By doing so, it could be also assessed with certainty whether female Tawny Pipit songs are temporally stable between seasons. Previous studies on White-crowned Sparrows and Northern Cardinals evaluated sex differences in the timings of the sensitive phase of song learning (Nelson et al. 1997; Yamaguchi 2001). In both studies, females and males were both confirmed to be closed-ended learners, but song acquisition was shorter in females. Assuming the same pattern, we may expect that female Tawny Pipits are also age-limited song learners, as expected for males, and thus also have a temporally stable song. In our dataset, one female was recorded in two different years and its song indeed did not change. However, there is no systematic study addressing whether female and male songbirds differ in the timing of song learning (see Riebel 2016).
To conclude, our study clearly demonstrates that songs of Tawny Pipit males and females are individually unique and suitable for IAM, although numerous individuals within a population may share highly similar song types. Clustering based on song comparison using the DTW approach was surprisingly successful in grouping songs of the same individual, but could not fully replace visual inspection by an experienced observer when songs differed only in fine structures and the recording quality was suboptimal. Neither our observations nor DTW-based clustering distinguished between the sexes. However, we cannot exclude that birds themselves do differentiate between songs of males and females; this hypothesis may be testable by playback experiments. Studies focusing on recording and evaluating songs from a greater number of female Tawny Pipits across years can bring more evidence for their presumed temporal song stability.
Availability of data and materials
The dataset generated and analysed during this study, including raw recordings and cut-outs of analysed songs in the WAV format, is available online (Zenodo data repository: https://doi.org/10.5281/zenodo.7670228). In addition, the recordings of songs used to create spectrograms in Figs. 1, S1 and S2 have been deposited in the Animal Sound Archive at the Museum for Natural History in Berlin and are available under the CC-BY-SA Licence at the following link: https://doi.org/10.7479/0k86-kd69.
References
Adi K, Johnson MT, Osiejuk TS (2010) Acoustic censusing using automatic vocalization classification and identity recognition. J Acoust Soc Am 127:874–883. https://doi.org/10.1121/1.3273887
Alström P, Mild K (2003) Pipits and Wagtails of Europe, Asia and North America: identification and systematics. Christopher Helm, London
Benedict L (2007) Occurrence and life history correlates of vocal duetting in North American passerines. J Avian Biol 39:57–65. https://doi.org/10.1111/j.2008.0908-8857.04103.x
Bennett RE, Rodewald AD, Rosenberg KV (2019) Overlooked sexual segregation of habitats exposes female migratory landbirds to threats. Biol Conserv 240:108266. https://doi.org/10.1016/j.biocon.2019.108266
Beran V, Poledníková K, Poledník L et al (2018) Certified methodology for planning management measures and creating suitable habitats for European Pipit in active mining area. ALKA Wildlife o.p.s. (In Czech with English summary)
Blumstein DT, Mennill DJ, Clemins P et al (2011) Acoustic monitoring in terrestrial environments using microphone arrays: applications, technological considerations and prospectus. J Appl Ecol 48:758–767. https://doi.org/10.1111/j.1365-2664.2011.01993.x
Brainard MS, Doupe AJ (2002) What songbirds teach us about learning. Nature 417:351–358. https://doi.org/10.1038/417351a
Brenowitz EA, Margoliash D, Nordeen KW (1997) An introduction to birdsong and the avian song system. J Neurobiol 33:495–500. https://doi.org/10.1002/(sici)1097-4695(19971105)33:5%3c495::aid-neu1%3e3.0.co;2-%23
Briedis M, Beran V, Hahn S et al (2016) Annual cycle and migration strategies of a habitat specialist, the Tawny Pipit Anthus campestris, revealed by geolocators. J Ornithol 157:619–626. https://doi.org/10.1007/s10336-015-1313-3
Budka M, Matyjasiak P, Typiak J et al (2019) Experienced males modify their behaviour during playback: the case of the Chaffinch. J Ornithol 160:673–684. https://doi.org/10.1007/s10336-019-01647-w
Calero-Riestra M, García JT (2019) Bisbita campestre – Anthus campestris (Linnaeus, 1758). Enciclopedia Virtual los Vertebrados Españoles. http://www.vertebradosibericos.org. Accessed 12 Apr 2022 (in Spanish)
Calvo B, Furness RW (1992) A review of the use and the effects of marks and devices on birds. Ringing Migr 13:129–158. https://doi.org/10.1080/03078698.1992.9674036
Catchpole CK, Slater PJB (2008) Bird song: biological themes and variations, 2nd edn. Cambridge University Press, Cambridge
Celis-Murillo A, Deppe JL, Allen MF (2009) Using soundscape recordings to estimate bird species abundance, richness, and composition. J Field Ornithol 80:64–78. https://doi.org/10.1111/j.1557-9263.2009.00206.x
Choi W, Lee JH, Sung HC (2019) A case study of male tawny owl (Strix aluco) vocalizations in South Korea: call feature, individuality, and the potential use for census. Anim Cells Syst (Seoul) 23:90–96. https://doi.org/10.1080/19768354.2019.1592022
Clewley GD, Robinson RA, Clark JA (2018) Estimating mortality rates among passerines caught for ringing with mist nets using data from previously ringed birds. Ecol Evol 8:5164–5172. https://doi.org/10.1002/ece3.4032
Darolová A, Krištofík J, Knauer F et al (2020) Behavioural response of Eurasian Blackcaps to acoustically simulated conspecific and heterospecific male intruders. J Ornithol 161:447–458. https://doi.org/10.1007/s10336-019-01743-x
Dixon P (2003) VEGAN, a package of R functions for community ecology. J Veg Sci 14:927–930. https://doi.org/10.1111/j.1654-1103.2003.tb02228.x
Galeotti P, Sacchi R (2001) Turnover of territorial Scops Owls Otus scops as estimated by spectrographic analyses of male hoots. J Avian Biol 32:256–262. https://doi.org/10.1111/j.0908-8857.2001.320308.x
Garamszegi LZ, Pavlova DZ, Eens M et al (2007) The evolution of song in female birds in Europe. Behav Ecol 18:86–96. https://doi.org/10.1093/beheco/arl047
Gilbert G, Tyler G, Smith K (2002) Local annual survival of booming male Great Bittern Botaurus stellaris in Britain, in the period 1990–1999. Ibis 144:51–61. https://doi.org/10.1046/j.0019-1019.2001.00012.x
Grava T, Mathevon N, Place E et al (2008) Individual acoustic monitoring of the European Eagle Owl Bubo bubo. Ibis 150:279–287. https://doi.org/10.1111/j.1474-919X.2007.00776.x
Grunst ML, Grunst AS, Rotenberry JT (2014) Phenotypic plasticity in nest departure calls: weighing costs and benefits. Anim Behav 90:31–39. https://doi.org/10.1016/j.anbehav.2014.01.010
Hahn AH, Krysler A, Sturdy CB (2013) Female song in black-capped chickadees (Poecile atricapillus): acoustic song features that contain individual identity information and sex differences. Behav Process 98:98–105. https://doi.org/10.1016/j.beproc.2013.05.006
Hall ML, Langmore NE (2017) Fitness costs and benefits of female song. Frontiers Media, Lausanne
Hick KG, Doucet SM, Mennill DJ (2016) Tropical wrens rely more on acoustic signals than visual signals for inter- and intra-specific discrimination. Anim Behav 118:153–163. https://doi.org/10.1016/j.anbehav.2016.05.024
Hobson KA, Rempel RS, Greenwood H et al (2008) Acoustic surveys of birds using electronic recordings: new potential from an omnidirectional microphone system. Wildl Soc Bull 30:709–720
Hodgson L, Waas JR, Foote JR (2018) Early singers attend to conspecific but not heterospecific behavioural cues at dawn. J Avian Biol 49:e01749. https://doi.org/10.1111/jav.01749
Immelmann K (1969) Song development in the zebra finch and other estrildid finches. In: Hinde RA (ed) Bird vocalizations. Cambridge University Press, Cambridge, pp 61–77
IUCN (2022) The IUCN red list of threatened species. Version 2022–1. https://www.iucnredlist.org. Accessed 15 Apr 2022
Ketterson ED, Nolan V Jr, Sandell M (2005) Testosterone in females: mediator of adaptive traits, constraints on sexual dimorphism, or both? Am Nat 164:S85–S88. https://doi.org/10.1086/444602
Kirschel ANG, Cody ML, Harlow ZT et al (2011) Territorial dynamics of Mexican Ant-thrushes Formicarius moniliger revealed by individual recognition of their songs. Ibis 153:255–268. https://doi.org/10.1111/j.1474-919X.2011.01102.x
Kriner E, Schwabl H (1991) Control of winter song and territorial aggression of female robins (Erithacus rubecula) by testosterone. Ethology 87:37–44. https://doi.org/10.1111/j.1439-0310.1991.tb01186.x
Lachlan RF (2011) Luscinia: a bioacoustics analysis computer program. http://rflachlan.github.io/Luscinia/. Accessed 2 Aug 2021
Laiolo P, Vögeli M, Serrano D et al (2007) Testing acoustic versus physical marking: two complementary methods for individual-based monitoring of elusive species. J Avian Biol 38:672–681. https://doi.org/10.1111/j.2007.0908-8857.04006.x
Lambrechts MM, Dhondt AA (1995) Individual voice discrimination in birds. In: Power DM (ed) Current ornithology. Springer, Boston, pp 115–139
Langmore NE (1998) Functions of duet and solo songs of female birds. Trends Ecol Evol 13:136–140. https://doi.org/10.1016/S0169-5347(97)01241-X
Letunic I, Bork P (2021) Interactive tree of life (iTOL) v5: an online tool for phylogenetic tree display and annotation. Nucleic Acids Res 49:W293–W296. https://doi.org/10.1093/nar/gkab301
Linhart P, Fuchs R, Poláková S et al (2012) Once bitten twice shy: Long-term behavioural changes caused by trapping experience in willow warblers Phylloscopus trochilus. J Avian Biol 43:186–192. https://doi.org/10.1111/j.1600-048X.2012.05580.x
Marco I, Mentaberre G, Ponjoan A et al (2006) Capture myopathy in little bustards after trapping and marking. J Wildl Dis 42:889–891. https://doi.org/10.7589/0090-3558-42.4.889
Marler P (1991) Song-learning behavior: the interface with neuroethology. Trends Neurosci 14:199–206. https://doi.org/10.1016/0166-2236(91)90106-5
Matyjasiak P (2005) Birds associate species-specific acoustic and visual cues: recognition of heterospecific rivals by male blackcaps. Behav Ecol 16:467–471. https://doi.org/10.1093/beheco/ari012
McDonald MV, Greenberg R (1991) Nest departure calls in female songbirds. Condor 93:365–373. https://doi.org/10.2307/1368952
Mooney R, Prather J, Roberts T (2008) Neurophysiology of birdsong learning. Elsevier, Oxford
Moreno J, Gil D, Cantarero A et al (2014) Extent of a white plumage patch covaries with testosterone levels in female Pied Flycatchers Ficedula hypoleuca. J Ornithol 155:639–648. https://doi.org/10.1007/s10336-014-1046-8
Mulcahy DM, Gartrell B, Gill RE et al (2011) Coelomic implantation of satellite transmitters in the bar-tailed godwit (Limosa lapponica) and the bristle-thighed curlew (Numenius tahitiensis) using propofol, bupivacaine, and lidocaine. J Zoo Wildl Med 42:54–64. https://doi.org/10.1638/2010-0040.1
Nelson DA, Marler P, Soha JA et al (1997) The timing of song memorization differs in males and females: a new assay for avian vocal hearing. Anim Behav 54:587–597. https://doi.org/10.1006/anbe.1996.0456
Neuschulz F (1986) Zum Gesang des männlichen und weiblichen Brachpiepers Anthus campestris. [On the song of male and female Tawny Pipit Anthus campestris]. J Ornithol 127:514–515. https://doi.org/10.1007/BF01640266 (In German with English summary)
Odom KJ, Benedict L (2018) A call to document female bird songs: applications for diverse fields. Auk 135:314–325. https://doi.org/10.1642/AUK-17-183.1
Odom KJ, Hall ML, Riebel K et al (2014) Female song is widespread and ancestral in songbirds. Nat Commun 5:3379. https://doi.org/10.1038/ncomms4379
Oñate-Casado J, Porteš M, Beran V et al (2021) An experience to remember: lifelong effects of playback-based trapping on behaviour of a migratory passerine bird. Anim Behav 182:19–29. https://doi.org/10.1016/j.anbehav.2021.09.010
Osiejuk TS, Grzybek J, Tryjanowski P (2007) Song structure and repertoire sharing in the Tawny Pipit Anthus campestris in Poland. Acta Ornithol 42:157–165. https://doi.org/10.3161/068.042.0210
Pérez-Granados C, Bota G, Giralt D et al (2019) Vocal activity rate index: a useful method to infer terrestrial bird abundance with acoustic monitoring. Ibis 161:901–907. https://doi.org/10.1111/ibi.12728
Petrusková T, Pišvejcová I, Kinštová A et al (2016) Repertoire-based individual acoustic monitoring of a migratory passerine bird with complex song as an efficient tool for tracking territorial dynamics and annual return rates. Methods Ecol Evol 7:274–284. https://doi.org/10.1111/2041-210X.12496
Petrusková T, Kahounová H, Pišvejcová I et al (2021) Evaluating the potential effects of capturing and handling on subsequent observations of a migratory passerine through individual acoustic monitoring. J Avian Biol 52:e02739. https://doi.org/10.1111/jav.02739
Price JJ (2019) Sex differences in song and plumage color do not evolve through sexual selection alone: new insights from recent research. J Ornithol 160:1213–1219. https://doi.org/10.1007/s10336-019-01681-8
Riebel K (2016) Understanding sex differences in form and function of bird song: the importance of studying song learning processes. Front Ecol Evol 4:62. https://doi.org/10.3389/fevo.2016.00062
Schwabl H (1992) Winter and breeding territorial behaviour and levels of reproductive hormones of migratory European robins. Ornis Scand 23:271–276. https://doi.org/10.2307/3676649
Sebastián-González E, Camp RJ, Tanimoto AM et al (2018) Density estimation of sound-producing terrestrial animals using single automatic acoustic recorders and distance sampling. Avian Conserv Ecol 13:7. https://doi.org/10.5751/ACE-01224-130207
Sierro J, de Kort SR, Riebel K et al (2022) Female blue tits sing frequently: a sex comparison of occurrence, context, and structure of song. Behav Ecol 33:912–925. https://doi.org/10.1093/beheco/arac044
Specht R (2007) Avisoft-SASLab Pro ver. 4.5, 5. Sound analysis and synthesis software. Avisoft Bioacoustics, Berlin
Spotswood EN, Goodman KR, Carlisle J et al (2012) How safe is mist netting? Evaluating the risk of injury and mortality to birds. Methods Ecol Evol 3:29–38. https://doi.org/10.1111/j.2041-210X.2011.00123.x
Terry AMR, Peake TM, McGregor PK (2005) The role of vocal individuality in conservation. Front Zool 2:10. https://doi.org/10.1186/1742-9994-2-10
Těšický M, Krajzingrová T, Eliáš J et al (2022) Inter-annual repeatability and age-dependent changes in plasma testosterone levels in a longitudinally monitored free-living passerine bird. Oecologia 198:53–66. https://doi.org/10.1007/s00442-021-05077-5
Tripp TM, Otter KA (2006) Vocal individuality as a potential long-term monitoring tool for Western Screech-owls, Megascops kennicottii. Can J Zool 84:744–753. https://doi.org/10.1139/Z06-055
Tyler S, Christie DA (2016) Tawny Pipit (Anthus campestris). In: del Hoyo J, Elliot A, Sargatal J, Christie DA, de Juana E (eds) Handbook of the Birds of the World Alive. Lynx Editions, Barcelona
Vögeli M, Laiolo P, Serrano D et al (2008) Who are we sampling? Apparent survival differs between methods in a secretive species. Oikos 117:1816–1823. https://doi.org/10.1111/j.1600-0706.2008.17225.x
Volodin IA, Volodina EV, Klenova AV et al (2005) Individual and sexual differences in the calls of the monomorphic White-faced Whistling Duck Dendrocygna viduata. Acta Ornithol 40:43–52. https://doi.org/10.3161/068.040.0110
Webb WH, Brunton DH, Aguirre JD et al (2016) Female song occurs in songbirds with more elaborate female coloration and reduced sexual dichromatism. Front Ecol Evol 4:22. https://doi.org/10.3389/fevo.2016.00022
Williams H (2004) Birdsong and singing behavior. Ann N Y Acad Sci 1016:1–30. https://doi.org/10.1196/annals.1298.029
Yamaguchi A (1998) A sexually dimorphic learned birdsong in the Northern Cardinal. The Condor 100:504–511. https://doi.org/10.2307/1369716
Yamaguchi A (2001) Sex differences in vocal learning in birds. Nature 411:257–258. https://doi.org/10.1038/35077143
Yasukawa K (1989) The costs and benefits of a vocal signal: the nest associated ‘chit’ of the female red-winged blackbird, Agelaius phoeniceus. Anim Behav 38:866–874. https://doi.org/10.1016/S0003-3472(89)80118-6
Acknowledgements
We thank Vršanská uhelná a.s., a Sev.en Energy company, for providing access to the active coal mine areas. Many thanks to Robert Lachlan and Elisabeth Zandberg for advice on the use of Luscinia software. We also thank Sailee Sakhalkar for providing useful comments that improved the quality of the manuscript as well as for assistance with statistics. The study was compliant with all national legislation of the Czech Republic on animal welfare and the protection of wildlife. The capturing and ringing of birds were performed by authorized ringers (VB and MP).
Funding
Open access publishing supported by the National Technical Library in Prague. Financial support was provided by the Technology Agency of the Czech Republic (project no. TA04021269), the Czech Science Foundation (project no. 21-04023 K), and various resources of Charles University (Charles University Grant Agency project no. 400422, and SVV projects no. 260569 and 260436).
Author information
Authors and Affiliations
Contributions
TP, MP and JO-C conceived the study. MP and VB recorded most of the songs, performed long-term monitoring of the study population, ringed and identified the ringed birds. JO-C performed bioacoustic analyses under the supervision of TP and analysed the data with the support of AP and TP; JO-C and AP prepared figures. JO-C, TP and AP wrote the first draft. All authors approved the interpretation of the results and the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by T. S. Osiejuk.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Table S1
. Summary information about recorded birds, their sex, ringing status, and the number of days in each of the studied seasons their songs were recorded. (XLSX 13 KB)
Table S2.
Information about analysed recordings (recording date, individual assignment, period of the season, and a file name under which the recording is available in the file repository). (XLSX 26 KB)
Supplementary Figures S1 to S3.
Spectrograms of songs of Tawny Pipit males with variable songs (S1), of paired individuals (S2), and detailed dendrogram of song similarity with examples of song families (S3). (PDF 653 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Oñate-Casado, J., Porteš, M., Beran, V. et al. Guess who? Evaluating individual acoustic monitoring for males and females of the Tawny Pipit, a migratory passerine bird with a simple song. J Ornithol 164, 845–858 (2023). https://doi.org/10.1007/s10336-023-02058-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-023-02058-8