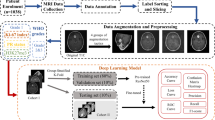
Abstract
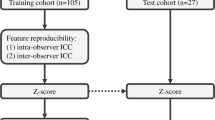
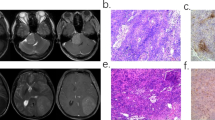
The objective of this study was to predict Ki-67 proliferation index of meningioma by using a nomogram based on clinical, radiomics, and deep transfer learning (DTL) features. A total of 318 cases were enrolled in the study. The clinical, radiomics, and DTL features were selected to construct models. The calculation of radiomics and DTL score was completed by using selected features and correlation coefficient. The deep transfer learning radiomics (DTLR) nomogram was constructed by selected clinical features, radiomics score, and DTL score. The area under the receiver operator characteristic curve (AUC) was calculated. The models were compared by Delong test of AUCs and decision curve analysis (DCA). The features of sex, size, and peritumoral edema were selected to construct clinical model. Seven radiomics features and 15 DTL features were selected. The AUCs of clinical, radiomics, DTL model, and DTLR nomogram were 0.746, 0.75, 0.717, and 0.779 respectively. DTLR nomogram had the highest AUC of 0.779 (95% CI 0.6643–0.8943) with an accuracy rate of 0.734, a sensitivity value of 0.719, and a specificity value of 0.75 in test set. There was no significant difference in AUCs among four models in Delong test. The DTLR nomogram had a larger net benefit than other models across all the threshold probability. The DTLR nomogram had a satisfactory performance in Ki-67 prediction and could be a new evaluation method of meningioma which would be useful in the clinical decision-making.





Similar content being viewed by others
Abbreviations
- ADC:
-
Apparent diffusion coefficient
- AUC:
-
Area under the receiver operating characteristic curve
- CI:
-
Confidence interval
- CNN:
-
Convolutional neural networks
- DCA:
-
Decision curve analysis
- DL:
-
Deep learning
- DTL:
-
Deep transfer learning
- DTLR:
-
Deep transfer learning radiomics
- ICC:
-
Intraclass correlation efficient
- LASSO:
-
Least absolute shrinkage and selection operator
- LR:
-
Logistic regression
- MRI:
-
Magnetic resonance imaging
- ROC:
-
Receiver operating characteristic
- ROI:
-
Region of interest
- WHO:
-
World Health Organization
References
Louis DN, Perry A, Wesseling P, et al. The 2021 WHO Classification of Tumors of the Central Nervous System: a summary. Neuro Oncol. 2021;23(8):1231-1251. https://doi.org/10.1093/neuonc/noab106
Ostrom QT, Gittleman H, Truitt G, Boscia A, Kruchko C, Barnholtz-Sloan JS. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2011–2015 [published correction appears in Neuro Oncol. 2018 Nov 17;:null]. Neuro Oncol. 2018;20(suppl_4):iv1-iv86. https://doi.org/10.1093/neuonc/noy131
Goldbrunner R, Stavrinou P, Jenkinson MD, et al. EANO guideline on the diagnosis and management of meningiomas. Neuro Oncol. 2021;23(11):1821-1834. https://doi.org/10.1093/neuonc/noab150
Rogers L, Barani I, Chamberlain M, et al. Meningiomas: knowledge base, treatment outcomes, and uncertainties. A RANO review. J Neurosurg. 2015;122(1):4-23. https://doi.org/10.3171/2014.7.JNS131644
Li D, Jiang P, Xu S, et al. Survival impacts of extent of resection and adjuvant radiotherapy for the modern management of high-grade meningiomas. J Neurooncol. 2019;145(1):125-134. https://doi.org/10.1007/s11060-019-03278-w
Black PM, Villavicencio AT, Rhouddou C, Loeffler JS. Aggressive surgery and focal radiation in the management of meningiomas of the skull base: preservation of function with maintenance of local control. Acta Neurochir (Wien). 2001;143(6):555-562. https://doi.org/10.1007/s007010170060
Martin B, Paesmans M, Mascaux C, et al. Ki-67 expression and patients survival in lung cancer: systematic review of the literature with meta-analysis. Br J Cancer. 2004;91(12):2018-2025.
Berlin A, Castro-Mesta JF, Rodriguez-Romo L, et al. Prognostic role of Ki-67 score in localized prostate cancer: a systematic review and meta-analysis. Urol Oncol.2017; 35(8):499-506.
Kim MS, Kim KH, Lee EH, et al. Results of immunohistochemical staining for cell cycle regulators predict the recurrence of atypical meningiomas. J Neurosurg. 2014; 121(5):1189-1200.
Oya S, Kawai K, Nakatomi H, Saito N. Significance of Simpson grading system in modern meningioma surgery: integration of the grade with MIB-1 labeling index as a key to predict the recurrence of WHO grade I meningiomas. J Neurosurg. 2012; 117(1):121-128.
Liu N, Song SY, Jiang JB, Wang TJ, Yan CX. The prognostic role of Ki-67/MIB-1 in meningioma: a systematic review with meta-analysis. Medicine.2020; 99(9):e18644.
Mirian C, Skyrman S, Bartek J Jr, et al. The Ki-67 Proliferation Index as a Marker of Time to Recurrence in Intracranial Meningioma. Neurosurgery. 2020;87(6):1289-1298. https://doi.org/10.1093/neuros/nyaa226
Lu Y, Liu L, Luan S, Xiong J, Geng D, Yin B. The diagnostic value of texture analysis in predicting WHO grades of meningiomas based on ADC maps: an attempt using decision tree and decision forest. Eur Radiol 2019; 29: 1318–28. https://doi.org/10.1007/s00330-018-5632-7
Park YW, Oh J, You SC, et al. Radiomics and machine learning may accurately predict the grade and histological subtype in meningiomas using conventional and diffusion tensor imaging. Eur Radiol. 2019;29(8):4068-4076. https://doi.org/10.1007/s00330-018-5830-3
Ke C, Chen H, Lv X, et al. Differentiation Between Benign and Nonbenign Meningiomas by Using Texture Analysis From Multiparametric MRI. J Magn Reson Imaging. 2020;51(6):1810-1820. https://doi.org/10.1002/jmri.26976
Yan PF, Yan L, Hu TT, et al. The Potential Value of Preoperative MRI Texture and Shape Analysis in Grading Meningiomas: A Preliminary Investigation. Transl Oncol. 2017;10(4):570-577. https://doi.org/10.1016/j.tranon.2017.04.006
Duan C, Zhou X, Wang J, et al. A radiomics nomogram for predicting the meningioma grade based on enhanced T1WI images. Br J Radiol. 2022;95(1137):20220141. https://doi.org/10.1259/bjr.20220141
Duan CF, Li N, Li Y, et al. Comparison of different radiomic models based on enhanced T1-weighted images to predict the meningioma grade. Clin Radiol. 2022;77(4):e302-e307. https://doi.org/10.1016/j.crad.2022.01.039
Gillies RJ, Kinahan PE, Hricak H. Radiomics: images are more than pictures, they are data. Radiology 2016; 278: 563–77. https://doi.org/10.1148/radiol.2015151169
Kumar V, Gu Y, Basu S, et al. Radiomics: the process and the challenges. Magn Reson Imaging. 2012;30(9):1234-1248. https://doi.org/10.1016/j.mri.2012.06.010
Lambin P, Rios-Velazquez E, Leijenaar R, et al. Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer. 2012;48(4):441-446. https://doi.org/10.1016/j.ejca.2011.11.036
Khanna O, Fathi Kazerooni A, Farrell CJ, et al. Machine Learning Using Multiparametric Magnetic Resonance Imaging Radiomic Feature Analysis to Predict Ki-67 in World Health Organization Grade I Meningiomas. Neurosurgery. 2021;89(5):928-936. https://doi.org/10.1093/neuros/nyab307
Zhao Y, Xu J, Chen B, Cao L, Chen C. Efficient Prediction of Ki-67 Proliferation Index in Meningiomas on MRI: From Traditional Radiological Findings to a Machine Learning Approach. Cancers (Basel). 2022;14(15):3637. Published 2022 Jul 26. https://doi.org/10.3390/cancers14153637
Tustison NJ, Avants BB, Cook PA et al (2010) N4ITK: improved N3 bias correction. IEEE Trans Med Imaging 29:1310–1320
Depeursinge A, Foncubierta-Rodriguez A, Van De Ville D, Muller H (2014) Three-dimensional solid texture analysis in biomedical imaging: review and opportunities. Med Image Anal 18:176–196
Bozdağ M, Er A, Ekmekçi S. Association of apparent diffusion coefficient with Ki-67 proliferation index, progesterone-receptor status and various histopathological parameters, and its utility in predicting the high grade in meningiomas. Acta Radiol. 2021;62(3):401-413. https://doi.org/10.1177/0284185120922142
Tang Y, Dundamadappa SK, Thangasamy S, et al. Correlation of apparent diffusion coefficient with Ki-67 proliferation index in grading meningioma. AJR Am J Roentgenol. 2014;202(6):1303-1308. https://doi.org/10.2214/AJR.13.11637
Baskan O, Silav G, Bolukbasi FH, Canoz O, Geyik S, Elmaci I. Relation of apparent diffusion coefficient with Ki-67 proliferation index in meningiomas. Br J Radiol. 2016;89(1057):20140842. https://doi.org/10.1259/bjr.20140842
Lambin P, Leijenaar RTH, Deist TM, et al. Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol. 2017;14(12):749-762. https://doi.org/10.1038/nrclinonc.2017.141
Aerts HJ, Velazquez ER, Leijenaar RT, et al. Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach [published correction appears in Nat Commun. 2014;5:4644. Cavalho, Sara [corrected to Carvalho, Sara]]. Nat Commun. 2014;5:4006. Published 2014 Jun 3. https://doi.org/10.1038/ncomms5006
Litjens G, Kooi T, Bejnordi BE, et al. A survey on deep learning in medical image analysis. Med Image Anal. 2017;42:60-88. https://doi.org/10.1016/j.media.2017.07.005
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Chongfeng Duan, Nan Li, and Xuejun Liu. The first draft of the manuscript was written by Chongfeng Duan, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
This is an observational study. The institutional review board of the Affiliated Hospital of Qingdao University has confirmed that no ethical approval is required.
Consent to Participate
Informed consent was not necessary to obtain because the study used MRI images which did not include identifying information.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key Points
• The DTLR nomogram had an AUC of 0.779 (95% CI 0.6643–0.8943) with an accuracy rate of 0.734, a sensitivity value of 0.719, and a specificity value of 0.75.
• The DTLR nomogram had a satisfactory performance in Ki-67 prediction and could be a new evaluation method of meningioma which would be useful in the clinical decision-making.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Duan, C., Hao, D., Cui, J. et al. An MRI-Based Deep Transfer Learning Radiomics Nomogram to Predict Ki-67 Proliferation Index of Meningioma. J Digit Imaging. Inform. med. 37, 510–519 (2024). https://doi.org/10.1007/s10278-023-00937-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10278-023-00937-3