Abstract
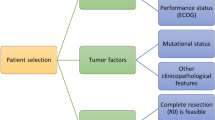
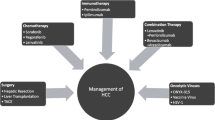
Hepatocellular carcinoma (HCC) is the most frequent primary neoplasm of the liver and it is the most relevant cause of death among patients with chronic liver disease. Prognosis assessment is a critical step in the management of HCC patients. Any attempt to assess the prognosis should consider not only information regarding tumor stage but also data about the degree of liver function impairment and the presence of cancer-related symptoms. In addition, any system aimed to be clinically successful should optimally attempt to link prognostic prediction and treatment indication. The Barcelona Clinic Liver Cancer staging system is the most accepted staging system. It has been externally validated, takes into account the most relevant prognostic parameters, and suggests the treatment approach based on the best scientific evidence. This article summarizes the most updated treatment strategy of HCC and future perspectives.
Résumé
Le carcinome hépatocellulaire (CHC) est la néoplasie hépatique primitive la plus fréquente et constitue la principale cause de décès chez les patients souffrant d’une maladie chronique du foie. L’évaluation du pronostic est une étape essentielle à la prise en charge des patients souffrant de CHC. Toute tentative d’évaluation du pronostic doit prendre en compte non seulement le stade de la tumeur, mais également les données quant au degré d’atteinte de la fonction hépatique et à la présence de symptômes associés au cancer. De plus, tout système visant un succès clinique doit essayer, de manière optimale, d’associer pronostic et indication de traitement. La classification de la clinique pour le cancer du foie de Barcelone (Barcelona Clinic Liver Cancer [BCLC]) est le système de classification le plus largement accepté. Elle a fait l’objet d’une validation externe, prend en compte les paramètres pronostiques les plus pertinents et propose l’approche thérapeutique fondée sur les meilleures données scientifiques.
Similar content being viewed by others
Références
Ferlay J, Soerjomataram I, Dikshit R, et al (2015) Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136:E359–E86
Forner A, Díaz-González A, Liccioni A, et al (2014) Prognosis prediction and staging. Best Pract Res Clin Gastroenterol 28:855–65
Bruix J, Reig M, Sherman M (2016) Evidence-based diagnosis, staging, and treatment of patients with hepatocellular carcinoma. Gastroenterology 150:835–53
Llovet JM, Bru C, Bruix J (1999) Prognosis of hepatocellular carcinoma: the BCLC staging classification. Semin Liver Dis 19:329–38
Forner A, Llovet JM, Bruix J (2012) Hepatocellular carcinoma. Lancet 379:1245–55
Cho YK, Kim JK, Kim WT, et al (2010) Hepatic resection versus radiofrequency ablation for very early stage hepatocellular carcinoma: a Markov model analysis. Hepatology 51:1284–90
Berzigotti A, Reig M, Abraldes JG, et al (2015) Portal hypertension and the outcome of surgery for hepatocellular carcinoma in compensated cirrhosis: a systematic review and meta-analysis. Hepatology 61:526–36
Ishizawa T, Hasegawa K, Aoki T, et al (2008) Neither multiple tumors nor portal hypertension are surgical contraindications for hepatocellular carcinoma. Gastroenterology 134:1908–16
Mazzaferro V (2016) Squaring the circle of selection and allocation in liver transplantation for HCC: an adaptive approach. Hepatology 63:1707–17
Bruix J, Han KH, Gores G, et al (2015) Liver cancer: approaching a personalized care. J Hepatol 62:S144–S56
Bruix J, Takayama T, Mazzaferro V, et al (2015) Adjuvant sorafenib for hepatocellular carcinoma after resection or ablation (STORM): a phase 3, randomised, double-blind, placebocontrolled trial. Lancet Oncol 16:1344–54
Llovet JM, Real MI, Montana X, et al (2002) Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet 359:1734–9
Lo CM, Ngan H, Tso WK, et al (2002) Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology 35:1164–71
Llovet JM, Bruix J (2003) Systematic review of randomized trials for unresectable hepatocellular carcinoma: chemoembolization improves survival. Hepatology 37:429–42
EASL-EORTC Clinical Practice Guidelines (2012) Management of hepatocellular carcinoma. J Hepatol 56:908–43
Forner A, Gilabert M, Bruix J, et al (2014) Treatment of intermediate-stage hepatocellular carcinoma. Nat Rev Clin Oncol 11:525–35
Lammer J, Malagari K, Vogl T, et al (2010) Prospective randomized study of doxorubicin-eluting-bead embolization in the treatment of hepatocellular carcinoma: results of the PRECISION V study. Cardiovasc Interv Radiol 33:41–52
Golfieri R, Giampalma E, Renzulli M, et al (2014) Randomised controlled trial of doxorubicin-eluting beads vs conventional chemoembolisation for hepatocellular carcinoma. Br J Cancer 111:255–64
Burrel M, Reig M, Forner A, et al (2012) Survival of patients with hepatocellular carcinoma treated by transarterial chemoembolisation (TACE) using drug eluting beads. Implications for clinical practice and trial design. J Hepatol 56:1330–5
Malagari K, Pomoni M, Moschouris H, et al (2012) Chemoembolization with doxorubicin-eluting beads for unresectable hepatocellular carcinoma: five-year survival analysis. Cardiovasc Interv Radiol 35:1119–28
Takayasu K, Arii S, Kudo M, et al (2012) Superselective transarterial chemoembolization for hepatocellular carcinoma. Validation of treatment algorithm proposed by Japanese guidelines. J Hepatol 56:886–92
Bruix J, Reig M, Rimola J, et al (2011) Clinical decision making and research in hepatocellular carcinoma: pivotal role of imaging techniques. Hepatology 54: 2238–44
Lencioni R, Llovet JM, Han G, et al (2016) Sorafenib or placebo plus TACE with doxorubicin-eluting beads for intermediate stage HCC: the SPACE trial. J Hepatol 64:1090–8
Kudo M, Han G, Finn RS, et al (2014) Brivanib as adjuvant therapy to transarterial chemoembolization in patients with hepatocellular carcinoma: a randomized phase III trial. Hepatology 60:1697–707
Llovet JM, Ricci S, Mazzaferro V, et al (2008) Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 359:378–90
Cheng AL, Kang YK, Chen Z, et al (2009) Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol 10:25–34
Reig M, Torres F, Rodriguez-Lope C, et al (2014) Early dermatologic adverse events predict better outcome in HCC patients treated with sorafenib. J Hepatol 61: 318–24
Bruix J, Qin S, Merle P, et al (2017) Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebocontrolled, phase 3 trial. Lancet 389:56–66
Sangro B (2014) Chemoembolization and radioembolization. Best Pract Res Clin Gastroenterol 28:909–19
Ribeiro de Souza A, Reig M, Bruix J (2016) Systemic treatment for advanced hepatocellular carcinoma: the search of new agents to join sorafenib in the effective therapeutic armamentarium. Expert Opin Pharmacother 17:1923–36
El-Khoueiry AB, Sangro B, Yau T, et al (2017) Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet-pii: S0140-6736(17)31046-2. doi: 10.1016/S0140-6736(17)31046-2
Santoro A, Rimassa L, Borbath I, et al (2013) Tivantinib for second-line treatment of advanced hepatocellular carcinoma: a randomised, placebo-controlled phase 2 study. Lancet Oncol 14:55–63
Zhu AX, Park JO, Ryoo BY, et al (2015) Ramucirumab versus placebo as second-line treatment in patients with advanced hepatocellular carcinoma following first-line therapy with sorafenib (REACH): a randomised, double-blind, multicentre, phase 3 trial. Lancet Oncol 16:859–70
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Díaz-González, A., Forner, A. Hepatocellular carcinoma: Therapeutic strategy in 2017. Oncologie 19, 177–183 (2017). https://doi.org/10.1007/s10269-017-2713-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10269-017-2713-1