Abstract
In the realm of acute respiratory infections, coronavirus disease-19 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), poses a global public health challenge. The application of corticosteroids (CSs) in COVID-19 remains a contentious topic among researchers. Accordingly, our team performed a comprehensive meta-analysis of randomized controlled trials (RCTs) to meticulously evaluate the safety and efficacy of CSs in hospitalized COVID-19 patients. To explore efficacy of CSs in the treatment of COVID-19 patients, we meticulously screened RCTs across key databases, including PubMed, Web of Science, Embase, Cochrane Library, ClinicalTrials.gov, as well as China's CNKI and Wanfang Data. We focused on assessing the 28 days mortality rates. We evaluated the data heterogeneity using the Chi-square test and I2 values, setting significance at 0.1 and 50%. Data from 21 RCTs involving 5721 participants were analyzed. The analysis did not demonstrate a significant association between CSs intervention and the 28 days mortality risk in hospitalized COVID-19 patients (relative risk [RR] = 0.93; 95% confidence interval [95% CI]: 0.84–1.03; P = 0.15). However, subgroup analysis revealed a significant reduction in 28 days mortality among patients with moderate-to-severe COVID-19 (RR at 0.85; 95% CI: 0.76–0.95; P = 0.004). Specifically, short-term CS administration (≤ 3 days) was associated with a substantial improvement in clinical outcomes (RR = 0.24; 95% CI: 0.09–0.63; P = 0.004), as was longer-term use (≥ 8 days) (RR = 0.88; 95% CI: 0.77–0.99; P = 0.04). Additionally, in patients with moderate-to-severe COVID-19, the administration of dexamethasone increased the number of 28 days ventilator-free days (Mean Difference = 1.92; 95% CI: 0.44–3.40; P = 0.01). Methylprednisolone also demonstrated significant benefits in improving clinical outcomes (RR = 0.24; 95% CI: 0.09–0.63; P = 0.004). Our meta-analysis demonstrated that although there is no significant difference in 28 days mortality rates among hospitalized COVID-19 patients, the use of CSs may be beneficial in improving clinical outcomes in moderate or severe COVID-19 patients. There was no significant increase in the occurrence of adverse events associated with the use of CSs. Our meta-analysis provides evidence that while CSs may not be suitable for all COVID-19 patients, they could be effective and safe in severely ill COVID-19 patients. Consequently, it is recommended to administer CSs for personalized treatments in COVID-19 cases to improve the clinical outcomes while minimizing adverse events.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The COVID-19, caused by SARS-CoV-2, has become the most severe global public health emergency and greatly impacted the world since its outbreak at the end of 2019 [1]. The COVID-19 infection is characterized by increased mobility and high mortality rate, especially among the elderly patients with underlying diseases [2]. By December 6, 2023, this pandemic has resulted in over 772 million cases and 6.98 million fatalities worldwide, presenting unprecedented challenges to human health and healthcare systems [3].
For individuals with confirmed COVID-19, novel targeted therapeutics for COVID-19 treatment still remain limited. To date, the supportive care remains the primary medical interventions for COVID-19 cases, and medications often prove ineffective and may carry the risk of toxicity, particularly when used in combination [4]. It is noteworthy that the World Health Organization recommended the application of CSs in severe and critical COVID-19 patients [5]. Initial recommendations from the Infectious Diseases Society of America in September 2022 were against the routine use of CSs for COVID-19, unless there were critically ill COVID-19 hospitalized patients or other reasons, such as asthma, or refractory shock [6]. However, the efficacy and safety of CSs in COVID-19 patients have been a debate within the medical community [7]. On the one hand, several studies underscore the potential therapeutic benefits of CSs administration; on the other hand, there is a growing body of literature highlighting adverse outcomes associated with CSs. For example, Li and colleagues found the use of CSs in COVID-19 patients is associated with prolonged viral RNA shedding, raising concerns about potential adverse effects [8, 9]. In addition, there were several studies that have shown either no benefit or an increased mortality in different subgroup of cases [10,11,12,13]. This ongoing controversy highlights the critical need for continued and comprehensive systematic review and meta-analysis to provide more evidence for the efficacy of CSs therapy in COVID-19 cases.
Considering all factors mentioned above, we conducted a systematic review and meta-analysis to assess the safety and efficacy of CSs therapy in hospitalized COVID-19 patients, juxtaposed with non-steroid treated groups and cohorts receiving different steroid dosages. Moreover, our analysis further analyzed different CSs subgroups in terms of disease severity, dosage, the specific type of CSs employed, and the duration of treatment. Our aim is to offer more therapeutic options to physicians and the medical community.
Methods
Databases and search strategy
In accordance with the rigorous standards set by the PRISMA declaration, our study was duly registered on the PROSPERO platform (CRD42023486275). Our methodological approach entailed an extensive and strategic literature search across a suite of databases including PubMed, Web of Science, Embase, Cochrane Library, ClinicalTrials.gov, CNKI, and Wanfang Data. We designed a comprehensive search algorithm, blending both MeSH terms and free-text terms in a Boolean framework. The MeSH terms are listed as follows: COVID-19, glucocorticoid, dexamethasone, prednisolone, and prednisone. The free-text terms are SARS-CoV-2, 2019 Novel Coronavirus, methylfluorprednisolone, hexadecadrol, and so on (see Table 1 of Supplemental Material file online for more details regarding the search strategies). The search string was crafted to capture a wide range of literature related to COVID-19 and CSs therapy, leading to a comprehensive retrieval of 5757 articles, including one identified through manual search, up to the cutoff date of November 21, 2023.
Eligibility criteria
This investigation entailed a systematic appraisal of RCTs, following stringent criteria encompassing participant characteristics, intervention strategies, comparison groups, outcome measures, and study designs. Inclusion was limited to patients aged 18 and above, diagnosed with COVID-19 according to established clinical guidelines [14]. The interventions under review were various CSs therapies, set against control groups receiving either alternate dosages of CSs, standard supportive care, or placebo [14].
Study selection was undertaken independently by two researchers (Xiangrong Ye and Ye Li), who diligently reviewed titles and abstracts for relevance. Disparities in selection were resolved through joint discussions or by consulting a third arbitrator (Feng Luo). For articles lacking complete data, efforts were made to obtain additional information from the study's corresponding authors. The Cochrane Risk of Bias Tool 2.0 was utilized to assess the methodological robustness and potential bias of the studies included [15].
Data collection process
The primary outcomes of this investigation were the 28 days mortality rate. The secondary outcomes evaluated included the duration of hospitalization, the number of days free from mechanical ventilation within the first 28 days, rates of ICU transfer, and the 60 days mortality rate. The study also analyzed adverse events, specifically focusing on secondary infections such as bacteremia and fungal infections, as well as significant adverse events categorized as Grade 3–4. Patients requiring prolonged CSs or immunosuppressive treatment for chronic ailments were systematically excluded. Data collation and extraction were executed independently by two distinct groups, ensuring the integrity and precision of the data collection process.
Assessment of risk of bias and quality of evidence
In conducting this study, evaluators Ye and Li independently applied the Cochrane Risk of Bias Tool for a detailed and systematic assessment of the randomized controlled trials under review. This comprehensive evaluation targeted seven critical aspects: (1) the methodology of random sequence generation, (2) the integrity of allocation concealment, (3) the effectiveness of blinding for participants and personnel, (4) the impartiality of outcome assessment blinding, (5) the completeness and reliability of outcome data, (6) the presence of selective reporting biases, and (7) the potential influence of other bias sources. Each domain was meticulously appraised, classified into categories of 'low,' 'unclear,' or 'high' risk of bias, with the overarching bias risk for each trial being determined by the highest risk level noted across these domains.
Subgroup analysis
Subgroup analyses within the study focused on several pivotal variables: CSs type (dexamethasone, methylprednisolone, and placebo), patient condition severity (mild, moderate to severe), duration of treatment (≤ 3 days, 4–7 days, ≥ 8 days), and dosage intensity (high at > 2 mg/kg/day, low at ≤ 2 mg/kg/day).
Data analysis
For the statistical analysis, the Review Manager (RevMan) version 5.3 was used, a tool by the Cochrane Collaboration. Heterogeneity among study results was assessed with Chi-square and I2 tests, applying thresholds of 0.1 and 50%, respectively, under a random-effects model framework. Preference was given to the I2 test results in cases of inconsistency. The Mantel–Haenszel method was utilized for binary data synthesis, deriving pooled RR along with their 95% CIs. Continuous outcomes were analyzed using the inverse variance approach, with mean differences (MDs) and corresponding 95% CIs as the metrics. In situations where only medians and interquartile ranges were reported, the mean–variance estimation method was employed for mean and standard deviation estimations [16]. Publication bias was probed using the funnel plots, Egger’s test, and Begg’s test [17].
Results
Study selection and study characteristics
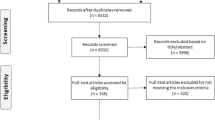
Utilizing the defined PICOS criteria, this systematic review initially retrieved a total of 5757 publications. After removing 1657 duplicates and subsequent excluding 3946 records based on title and abstract assessments, 154 articles were subjected to full-text evaluation. This process culminated in the inclusion of 21 studies for detailed data extraction and analysis, as depicted in Fig. 1, Tables 1, and 2. The flow diagram was created in accordance with PRISMA guidelines [18]. Published between 2020 and 2023, these studies collectively involved 5721 participants and were all RCTs. Among them, 15 studies [19,20,21,22,23,24,25,26,27,28,29,30,31,32,33] compared CSs therapy against standard treatment, three [34,35,36] used a placebo as the comparator, and three [37,38,39] utilized low-dose CSs. Methylprednisolone was the intervention in nine studies [19, 21, 22, 25, 29, 30, 34,35,36], while dexamethasone was used in twelve [20, 23, 24, 26,27,28, 31,32,33, 37,38,39], with treatment durations ranging from 3 to 10 days.
Risk of bias and quality of evidence
Within the analyzed cohort of studies, fourteen were adjudged to have a low risk of bias. Six studies were categorized as having unclear risk, primarily due to ambiguities in the implementation of blinding, measurement of outcomes, and descriptions of allocation concealment. One study was identified to present a high risk of bias, specifically attributed to inadequacies in allocation concealment (Figs. 2 and 3).
Primary outcomes: 28 days mortality
Across 20 studies [19, 23, 28,29,30,31,32, 34, 35, 37, 39], involving 4898 subjects, the 28 days mortality rates were analyzed. Within the CSs-treated cohort, the mortality rate stood at 19.4%, whereas the control group exhibited a rate of 20.2%. The computed RR was 0.93 (95% CI: 0.84–1.03; P = 0.15), suggesting there is no statistically significant correlation between CSs intervention and the control group with regard to the 28 days mortality rate among hospitalized COVID-19 patients (Fig. 4). Thus, CSs treatment did not confer a reduced risk of 28 days mortality. The heterogeneity assessment, conducted using Chi-square and I2 tests, indicated the absence of significant heterogeneity (30.19 and 37%, respectively).
Secondary outcomes
Analyzing data from 13 studies [21, 24,25,26,27,28,29, 32,33,34,35,36,37] focused on the impact of CSs treatment on hospitalization durations in COVID-19 patients, we found a mean difference (MD) of 0.83 days (95% CI: 0.32 to 1.33; P = 0.001). This indicates the use of CSs is associated with longer hospital stays (Fig. 5). Regarding 60 days mortality rates, the results (RR = 1.05; 95% CI: 0.84 to 1.32; P = 0.66) (Fig. 6) indicated no significant association with CSs use. Contrastingly, a significant reduction in 28 days ventilator-free days was observed in hospitalized COVID-19 patients (RR = 0.83; 95% CI: −3.69 to 2.95; P = 0.83) (Fig. 7). Moreover, the investigation revealed marked heterogeneity in the reports on hospital stay duration (Chi-square = 16.92, I2 = 29%) and 28 days Ventilator-Free Days (Chi-square = 26.10, I2 = 81%), but not in the 60 days mortality data (Chi-square = 0.08, I2 = 0%).
Adverse events
Our analysis revealed no significant differences in the incidence of serious adverse events among hospitalized COVID-19 patients [19, 20, 22, 23, 25, 36,37,38] (RR = 1.10; 95% CI: 0.97 to 1.26; P = 0.15), hospital-acquired secondary infections [19,20,21, 24, 25, 27,28,29, 31, 33, 38] (RR = 1.01; 95% CI: 0.85 to 1.20; P = 0.90), bacteremia [20, 25, 27, 28, 33] (RR = 0.90; 95% CI: 0.63 to 1.31; P = 0.59), and secondary fungal infections [23, 24, 31, 33] (RR = 0.58; 95% CI: 0.36 to 0.93; P = 0.02). Statistical analyses demonstrated no meaningful heterogeneity in studies examining serious adverse events (Chi-square = 10.05, I2 = 30%), nosocomial infections (Chi-square = 15.20, I2 = 34%), and bacteremia (Chi-square = 6.24, I2 = 36%). In COVID-19 patients, high-dose CSs therapy significantly reduced the incidence of fungal infections, and these findings showed homogeneity in statistical analysis (Chi-square = 0.97, I2 = 0%) (Fig. 8).
Subgroup analysis
The subgroup analyses, based on disease severity, demonstrated that CSs application in moderate or severe COVID-19 patients had lower rates of the 28 days mortality [20, 22, 24, 25, 28, 29, 35, 36, 39] and longer 28 days ventilator-free days [20, 23] compared with those without CSs application (Table 3, online supplemental Figs. 1 and 2). Subgroup analyses according to CSs type showed there is no statistically significant difference in 28 days mortality rates among patients treated with dexamethasone [23, 24, 28, 32, 33, 37, 39], methylprednisolone [19, 29, 30, 35, 36], or a combination therapy of these two CSs [21, 22, 25] (Table 3, online supplemental Fig. 3). Notably, despite significant heterogeneity (Chi-square = 10.63, I2 = 81%), the treatment of methylprednisolone alone [19, 30, 36] (RR = 0.24; 95% CI: 0.09 to 0.63; P = 0.004) significantly reduced the risk of 28 days mortality (RR = 0.24; 95% CI: 0.09 to 0.63; P = 0.004) when specifically analyzing moderate or severe COVID-19 patients (Table 3, online supplemental Fig. 4). Importantly, patients receiving short-term CSs application (≤ 3 days) [19, 30, 36] or treatments extending ≥ 8 days [20, 22,23,24,25, 28] tended to exhibit a lower rate of 28 days mortality when compared with those with an intermediate duration of CSs (4–7 days) [29, 35, 37] (Table 3, online supplemental Fig. 5). Analysis of the studies involving methylprednisolone alone and CSs usage of ≤ 3 days showed high heterogeneity, whereas most other analyses had low heterogeneity (Table 3).
Discussion
In contrast with previously published meta-analyses, our study presents several distinct advantages. It incorporates an expanded cohort of 21 RCTs, including cases of moderate or severe COVID-19. In addition, our research features a larger sample size, which enhances the quality of this study. In our findings, the benefits of CSs application were less evident in the general hospitalized patient population. Nevertheless, our findings show that CSs significantly reduce the 28 days mortality rate and increase the VFD over a 20 days period in COVID-19 patients with moderate or severe ARDS, which is possibly due to variations in disease severity and the diversity of CSs dosages administered to COVID-19 patients.
Focusing on COVID-19, the pharmacodynamics of hydrocortisone and dexamethasone and their impact on COVID-19 patients have been highlighted in recent WHO-sponsored trials and meta-analyses [40]. Our findings are in line with the RECOVERY trial, which demonstrated the reduction in 28 days mortality for patients with hypoxia or mechanical ventilation when treated with dexamethasone [41].
Based on results of our analysis, the use of CSs did not increase the risk of serious adverse events, secondary infections, bacteremia, and fungal infections in COVID-19 patients with ARDS. It is notable that, in severe COVID-19 cases, CSs application was associated with a reduced risk of fungal infections. This may be attributed to the potent anti-inflammatory effects of CSs, which can improve clinical outcomes [42]. Critically ill COVID-19 patients may benefit even more from higher dosage of CSs [43]. Furthermore, some studies suggest potential benefits of inhaled CSs in preventing disease deterioration and secondary organ damage in patients with COVID-19, which could be a new therapeutic option [44, 45].
In the present study, CSs treatment was not found to improve the disease prognosis in certain subgroups, such as the intermediate treatment duration (4–7 days) of CSs and the combination therapy of dexamethasone and methylprednisolone and intermediate duration (4–7 days) of CSs treatment, underscoring the necessity for further targeted clinical research [2]. As such, the current body of evidence does not support a universal application of CSs therapy among all hospitalized COVID-19 patients, and further stratified clinical research is required to demonstrate its efficacy [5]. This underscores the critical necessity for careful consideration in the clinical decision-making regarding the use of CSs as the clinical application of CSs is fraught with complexity. Inappropriate administration of CSs is associated with heightened risks of infection, osteoporosis, hyperglycemia, and bleeding, potentially worsening the patient's condition [46, 47].
Accordingly, identifying the optimal timing for CSs therapy is crucial. Our subgroup analysis demonstrates that durations of CSs treatment (≤ 3 days or ≥ 8 days) can significantly reduce mortality in severe COVID-19 patients, which may be related to the timing of CSs application during the inflammatory storm. An observational cohort study provides evidence that early short course of CSs combined with furosemide reduced the 28 days mortality in non-critically ill COVID-19 patients [48]. Findings from L. Mahajan and collaborators highlight that 10 days of dexamethasone treatment reduced the number of ICU admissions and mortality among COVID-19 patients [49]. Due to this result from subgroup analysis, we could emphasize there may be biased. Further clinical trials are required to confirm the conclusion from subgroup analysis.
Variations in recommended treatment durations for CSs might be attributed to the distinct metabolic pathways activated by different CSs, such as methylprednisolone and dexamethasone. They exert potent anti-inflammatory effects by binding with glucocorticoid response elements, modulating transcription factors, and influencing secondary messenger pathways [50]. This underscores the significance of selecting the appropriate CSs type for COVID-19 treatment [51], as the choice of CSs for treating COVID-19 remains an area of ongoing exploration. New clinical studies are required to explore the specific populations that may benefit from CSs, as well as to refine the timing and duration of CSs application, with the aim of optimizing the benefits of CSs while minimizing their potential harm.
Limitations
We acknowledge several inherent limitations. Firstly, considering the methodology of systematic reviews and meta-analyses, the issue of heterogeneity is inevitable, exacerbated by the diversity in patient demographics, therapeutic approaches, and the types of CSs administered. The second limitation arises from the inadequate reporting of adverse events in certain studies we analyzed, which constrains the conclusiveness of subgroup analysis in this context. Moreover, the short follow-up periods in the included RCTs limit the understanding of long-term adverse effects of CSs administered at varying doses and durations. This necessitates further research through observational studies or additional randomized trials to thoroughly understand the prolonged implications of CSs therapy in such cases.
Conclusion
CSs have been identified as an effective therapeutic intervention for reducing mortality risk in patients with severe COVID-19 pneumonia. Dose, duration, and underlying comorbidities of CSs treatment are key factors for clinical outcome of severe COVID-19. In our research, although there was no significant difference in 28 days mortality rates among hospitalized COVID-19 patients, our results show that CSs may be beneficial in improving clinical outcomes by choosing appropriate duration and type of application specially for COVID-19 patients with moderate or severe ARDS. Additionally, there was no significant increase in the incidence of adverse events associated with the use of CSs. It should be noted that our research has inherent limitations and bias is inevitable. Consequently, it is recommended to administer CSs for an personalized duration in moderate or severe COVID-19 cases to improve the clinical outcomes while minimizing adverse events. Our meta-analysis provides evidence that CSs are not suitable for all COVID-19 patients, but they could be effective and safe in severely ill COVID-19 patients.
Data availability
The datasets analyzed in the present study are available from the published papers that have been cited in the present manuscript.
References
Lamers MM, Haagmans BL. SARS-CoV-2 pathogenesis. Nat Rev Microbiol. 2022;20(5):270–84. https://doi.org/10.1038/s41579-022-00713-0.
Prescott HC, Rice TW. Corticosteroids in COVID-19 ARDS: evidence and hope during the pandemic. JAMA. 2020;324(13):1292–5. https://doi.org/10.1001/jama.2020.16747.
Sanders JM, Monogue ML, Jodlowski TZ, Cutrell JB. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): a review. JAMA. 2020;323(18):1824–36.
Waterer GW, Rello J, Wunderink RG. COVID-19: first do no harm. Am J Respir Crit Care Med. 2020;201(11):1324–5. https://doi.org/10.1164/rccm.202004-1153ED.
Agarwal A, Hunt B, Stegemann M, Rochwerg B, Lamontagne F, Siemieniuk RA, et al. A living WHO guideline on drugs for covid-19. BMJ. 2020;370:m3379. https://doi.org/10.1136/bmj.m3379.
Bhimraj A, Morgan RL, Shumaker AH, Baden L, Cheng VCC, Edwards KM, et al. Infectious diseases society of america guidelines on the treatment and management of patients with COVID-19. Clin Infect Dis. 2022. https://doi.org/10.1093/cid/ciac724.
Shang L, Zhao J, Hu Y, Du R, Cao B. On the use of corticosteroids for 2019-nCoV pneumonia. Lancet. 2020;395(10225):683–4. https://doi.org/10.1016/s0140-6736(20)30361-5.
Majmundar M, Kansara T, Lenik JM, Park H, Ghosh K, Doshi R, et al. Efficacy of corticosteroids in non-intensive care unit patients with COVID-19 pneumonia from the New York Metropolitan region. PLoS ONE. 2020;15(9):e0238827.
Li Y, Meng Q, Rao X, Wang B, Zhang X, Dong F, et al. Corticosteroid therapy in critically ill patients with COVID-19: a multicenter, retrospective study. Crit Care. 2020;24(1):698. https://doi.org/10.1186/s13054-020-03429-w.
Lu X, Chen T, Wang Y, Wang J, Yan F. Adjuvant corticosteroid therapy for critically ill patients with COVID-19. Crit Care. 2020;24(1):241. https://doi.org/10.1186/s13054-020-02964-w.
Russell CD, Millar JE, Baillie JK. Clinical evidence does not support corticosteroid treatment for 2019-nCoV lung injury. Lancet. 2020;395(10223):473–5. https://doi.org/10.1016/s0140-6736(20)30317-2.
Jung C, Wernly B, Fjølner J, Bruno RR, Dudzinski D, Artigas A, et al. Steroid use in elderly critically ill COVID-19 patients. Eur Respir J. 2021;58(4):2100979. https://doi.org/10.1183/13993003.00979-2021.
Liang MY, Chen P, He M, Tang J, Li H, He XL, et al. Corticosteroids treatment of patients with coronavirus disease 2019: a propensity score matching study. Curr Med Sci. 2021;41(1):24–30. https://doi.org/10.1007/s11596-021-2313-6.
Hu B, Guo H, Zhou P, Shi Z-L. Characteristics of SARS-CoV-2 and COVID-19. Nat Rev Microbiol. 2021;19(3):141–54.
Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, et al. The cochrane collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:5928.
Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5(1):1–10.
Egger M, Smith GD, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–34.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. PLoS Med. 2021;18(3):e1003583. https://doi.org/10.1371/journal.pmed.1003583.
Edalatifard M, Akhtari M, Salehi M, Naderi Z, Jamshidi A, Mostafaei S, et al. Intravenous methylprednisolone pulse as a treatment for hospitalised severe COVID-19 patients: results from a randomised controlled clinical trial. Eur Respir J. 2020;56(6):2002808. https://doi.org/10.1183/13993003.02808-2020.
Tomazini BM, Maia IS, Cavalcanti AB, Berwanger O, Rosa RG, Veiga VC, et al. Effect of dexamethasone on days alive and ventilator-free in patients with moderate or severe acute respiratory distress syndrome and COVID-19: the CoDEX randomized clinical trial. JAMA. 2020;324(13):1307–16. https://doi.org/10.1001/jama.2020.17021.
Taher A, Lashkari M, Keramat F, Hashemi SH, Sedighi L, Poorolajal J, et al. Comparison of the efficacy of equivalent doses of dexamethasone, methylprednisolone, and hydrocortisone for treatment of COVID-19-related acute respiratory distress syndrome: a prospective three-arm randomized clinical trial. Wien Med Wochenschr. 2023;173(5–6):140–51. https://doi.org/10.1007/s10354-022-00993-4.
Salton F, Confalonieri P, Centanni S, Mondoni M, Petrosillo N, Bonfanti P, et al. Prolonged higher dose methylprednisolone versus conventional dexamethasone in COVID-19 pneumonia: a randomised controlled trial (MEDEAS). Eur Respir J. 2023;61(4):2201514. https://doi.org/10.1183/13993003.01514-2022.
Munch MW, Myatra SN, Vijayaraghavan BKT, Saseedharan S, Benfield T, Wahlin RR, et al. Effect of 12 mg vs 6 mg of dexamethasone on the number of days alive without life support in adults with COVID-19 and severe Hypoxemia: the COVID STEROID 2 randomized trial. JAMA. 2021;326(18):1807–17. https://doi.org/10.1001/jama.2021.18295.
Rabascall CX, Lou BX, Dhar S, Hasan Z, Fryman C, Izard S, et al. Randomized open investigation determining steroid dose in severe COVID-19: the ROIDS-dose clinical trial. Cureus. 2022;14(11):e31086. https://doi.org/10.7759/cureus.31086.
Corral-Gudino L, Cusacovich I, Martín-González JI, Muela-Molinero A, Abadía-Otero J, González-Fuentes R, et al. Effect of intravenous pulses of methylprednisolone 250 mg versus dexamethasone 6 mg in hospitalised adults with severe COVID-19 pneumonia: an open-label randomised trial. Eur J Clin Invest. 2023;53(1):e13881. https://doi.org/10.1111/eci.13881.
Jamaati H, Hashemian SM, Farzanegan B, Malekmohammad M, Tabarsi P, Marjani M, et al. No clinical benefit of high dose corticosteroid administration in patients with COVID-19: a preliminary report of a randomized clinical trial. Eur J Pharmacol. 2021;897:173947. https://doi.org/10.1016/j.ejphar.2021.173947.
Bouadma L, Mekontso-Dessap A, Burdet C, Merdji H, Poissy J, Dupuis C, et al. High-dose dexamethasone and oxygen Support strategies in intensive care unit patients with severe COVID-19 acute hypoxemic respiratory failure: the COVIDICUS randomized clinical trial. JAMA Intern Med. 2022;182(9):906–16. https://doi.org/10.1001/jamainternmed.2022.2168.
Taboada M, Rodríguez N, Varela PM, Rodríguez MT, Abelleira R, González A, et al. Effect of high versus low dose of dexamethasone on clinical worsening in patients hospitalised with moderate or severe COVID-19 pneumonia: an open-label, randomised clinical trial. Eur Respir J. 2022;60(2):2102518. https://doi.org/10.1183/13993003.02518-2021.
Corral-Gudino L, Bahamonde A, Arnaiz-Revillas F, Gómez-Barquero J, Abadía-Otero J, García-Ibarbia C, et al. Methylprednisolone in adults hospitalized with COVID-19 pneumonia : an open-label randomized trial (GLUCOCOVID). Wien Klin Wochenschr. 2021;133(7–8):303–11. https://doi.org/10.1007/s00508-020-01805-8.
Fadel R, Morrison AR, Vahia A, Smith ZR, Chaudhry Z, Bhargava P, et al. Early short-course corticosteroids in hospitalized patients with COVID-19. Clin Infect Dis. 2020;71(16):2114–20. https://doi.org/10.1093/cid/ciaa601.
Naik NB, Puri GD, Kajal K, Mahajan V, Bhalla A, Kataria S, et al. High-dose dexamethasone versus tocilizumab in moderate to severe COVID-19 Pneumonia: a randomized controlled trial. Cureus. 2021;13(12):e20353. https://doi.org/10.7759/cureus.20353.
Maskin LP, Bonelli I, Olarte GL, Palizas F Jr, Velo AE, Lurbet MF, et al. High-versus low-dose dexamethasone for the treatment of COVID-19-related acute respiratory distress syndrome: a multicenter, randomized open-label clinical trial. J Intensive Care Med. 2022;37(4):491–9. https://doi.org/10.1177/08850666211066799.
Wu H, Daouk S, Kebbe J, Chaudry F, Harper J, Brown B. Low-dose versus high-dose dexamethasone for hospitalized patients with COVID-19 pneumonia: a randomized clinical trial. PLoS ONE. 2022;17(10):e0275217. https://doi.org/10.1371/journal.pone.0275217.
Tang X, Feng YM, Ni JX, Zhang JY, Liu LM, Hu K, et al. Early use of corticosteroid may prolong SARS-CoV-2 shedding in non-intensive care unit patients with COVID-19 Pneumonia: a multicenter, single-blind, randomized control trial. Respir Int Rev Thorac Dis. 2021;100(2):116–26. https://doi.org/10.1159/000512063.
Jeronimo CMP, Farias MEL, Val FFA, Sampaio VS, Alexandre MAA, Melo GC, et al. Methylprednisolone as adjunctive therapy for patients hospitalized with coronavirus disease 2019 (COVID-19; Metcovid): a randomized, double-blind, phase IIb. Placebo-controlled trial. Clin Infect Dis. 2021;72(9):e373–81. https://doi.org/10.1093/cid/ciaa1177.
Salvarani C, Massari M, Costantini M, Merlo DF, Mariani GL, Viale P, et al. Intravenous methylprednisolone pulses in hospitalised patients with severe COVID-19 pneumonia: a double-blind, randomised, placebo-controlled trial. Eur Respir J. 2022;60(4):2200025. https://doi.org/10.1183/13993003.00025-2022.
Sadeghi S, Arezoomandi N, Ardestani MM, Ardestani ME, Ghiasi F, Farajzadegan Z. Efficacy and safety comparison of two different doses of dexamethasone in hospitalized patients with COVID-19: a randomized clinical trial. J Res Pharm Pract. 2022;11(4):136–43. https://doi.org/10.4103/jrpp.jrpp_42_22.
Wolfe CR, Tomashek KM, Patterson TF, Gomez CA, Marconi VC, Jain MK, et al. Baricitinib versus dexamethasone for adults hospitalised with COVID-19 (ACTT-4): a randomised, double-blind, double placebo-controlled trial. Lancet Respir Med. 2022;10(9):888–99. https://doi.org/10.1016/S2213-2600(22)00088-1.
Montalvan E, Carcamo B, Palacion D, Rivera S, Estevez R, Norwood D, et al. High-dose vs low-dose dexamethasone in patients with COVID-19 in a tertiary hospital in western honduras. Chest. 2021;160(4):A1117–8. https://doi.org/10.1016/j.chest.2021.07.1027.
Pulakurthi YS, Pederson JM, Saravu K, Gupta N, Balasubramanian P, Kamrowski S, et al. Corticosteroid therapy for COVID-19: a systematic review and meta-analysis of randomized controlled trials. Medicine. 2021;100(20):e25719.
van Paassen J, Vos JS, Hoekstra EM, Neumann KMI, Boot PC, Arbous SM. Corticosteroid use in COVID-19 patients: a systematic review and meta-analysis on clinical outcomes. Crit Care. 2020;24(1):696. https://doi.org/10.1186/s13054-020-03400-9.
Rhen T, Cidlowski JA. Antiinflammatory action of glucocorticoids–new mechanisms for old drugs. N Engl J Med. 2005;353(16):1711–23. https://doi.org/10.1056/NEJMra050541.
Gogali A, Kyriakopoulos C, Kostikas K. Corticosteroids in COVID-19: one size does not fit all. Eur Respir J. 2021;57(4):2100224. https://doi.org/10.1183/13993003.00224-2021.
Bobot M, Tonon D, Peres N, Guervilly C, Lefèvre F, Max H, et al. Impact of dexamethasone and inhaled nitric oxide on severe acute kidney injury in critically Ill patients with COVID-19. J Clin Med. 2022;11(20):6130. https://doi.org/10.3390/jcm11206130.
Agusti A, Torres F, Faner R. Early treatment with inhaled budesonide to prevent clinical deterioration in patients with COVID-19. Lancet Respir Med. 2021;9(7):682–3. https://doi.org/10.1016/s2213-2600(21)00171-5.
Fang F, Zhang Y, Tang J, Lunsford LD, Li T, Tang R, et al. Association of corticosteroid treatment with outcomes in adult patients with sepsis: a systematic review and meta-analysis. JAMA Intern Med. 2019;179(2):213–23. https://doi.org/10.1001/jamainternmed.2018.5849.
Rabe KF, Martinez FJ, Ferguson GT, Wang C, Singh D, Wedzicha JA, et al. Triple inhaled therapy at two glucocorticoid doses in moderate-to-very-severe COPD. N Engl J Med. 2020;383(1):35–48. https://doi.org/10.1056/NEJMoa1916046.
Kevorkian JP, Riveline JP, Vandiedonck C, Girard D, Galland J, Féron F, et al. Early short-course corticosteroids and furosemide combination to treat non-critically ill COVID-19 patients: an observational cohort study. J Infect. 2021;82(1):e22–4. https://doi.org/10.1016/j.jinf.2020.08.045.
Mahajan L, Singh AP, Singla A, Singh G. Role of Dexamethasone on oxygen requirement, mortality, and survival incidence among COVID-19 patients: quasi-experimental study. Anesth Essays Res. 2021;15(1):62–6. https://doi.org/10.4103/aer.aer_70_21.
Chaudhuri D, Sasaki K, Karkar A, Sharif S, Lewis K, Mammen MJ, et al. Corticosteroids in COVID-19 and non-COVID-19 ARDS: a systematic review and meta-analysis. Intensive Care Med. 2021;47(5):521–37. https://doi.org/10.1007/s00134-021-06394-2.
Meduri GU, Annane D, Confalonieri M, Chrousos GP, Rochwerg B, Busby A, et al. Pharmacological principles guiding prolonged glucocorticoid treatment in ARDS. Intensive Care Med. 2020;46(12):2284–96. https://doi.org/10.1007/s00134-020-06289-8.
Acknowledgements
Thanks to all the colleagues who have contributed to this article for their guidance and help, especially Teacher Luo.
Funding
This study was supported by the Guangzhou Clinical Characteristic Technology Project [grant number C3230125].
Author information
Authors and Affiliations
Contributions
All authors contributed extensively to this work. YXR involved in conceptualization, resources, data curation, software, formal analysis, writing—original draft, project administration, writing—review & editing. LY took part in conceptualization, resources, data curation, software, formal analysis, writing—original draft, project administration, writing—review & editing. LF took part in conceptualization, resources, data curation, software, formal analysis, writing—original draft, project administration, writing—review & editing. XZB took part in software, formal analysis, supervision, methodology, project administration. KK involved in data curation, supervision, methodology, project administration. WJ involved in supervision, project administration. XPH involved in software, supervision, methodology. TCJ took part in conceptualization, supervision, methodology, project administration. YH involved in conceptualization, supervision, methodology, project administration. LYF involved in conceptualization, resources, data curation, formal analysis, supervision, validation, investigation, visualization, methodology, project administration, writing—review & editing.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical approval
All analyses were based on previous published studies. Thus, no ethical approval and patient consent are required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ye, X., Li, Y., Luo, F. et al. Efficacy and safety of glucocorticoids in the treatment of COVID-19: a systematic review and meta-analysis of RCTs. Clin Exp Med 24, 157 (2024). https://doi.org/10.1007/s10238-024-01405-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10238-024-01405-0