Abstract
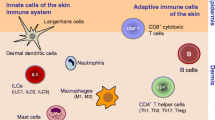
Mast cells (MCs) are immune cells derived from myeloid lineage present in all classes of vertebrates and have emerged preceding much time the development of adaptive immunity. MCs are involved in inflammatory processes, allergic reactions, and host responses to parasites and bacteria infectious diseases. MCs are located at the host-environment interface, at many sites of initial antigen entry, including skin, lung and gastrointestinal tract, and have part of a protective mechanism. Skin has an important role in protecting the host from invasion both as physical barriers and by employing an intricate network of resident immune and non-immune cells include macrophages, T and B lymphocytes, MCs, neutrophils, eosinophils, and Langerhans cells. In this review we discussed the role of MCs in human skin cancers.




Similar content being viewed by others
References
Bos JD, Kapsenberg ML. The skin immune system Its cellular constituents and their interactions. Immunol Today. 1986;7:235–40.
Nestle FO, Di Meglio P, Qin J-Z, Nickoloff BJ. Skin immune sentinels in health and disease. Nat Rev Immunol. 2009;9:679–91.
Crivellato E, Ribatti D. The mast cell: an evolutionary perspective. Biol Rev Camb Philos Soc. 2010;85:347–60.
Bischoff SC. Physiological and pathophysiological functions of intestinal mast cells. Semin Immunopathol. 2009;31:185–205.
Weber A, Knop J, Maurer M. Pattern analysis of human cutaneous mast cell populations by total body surface mapping. Br J Dermatol. 2003;148:224–8.
Pilkington SM, Barron MJ, Watson REB, Griffiths CEM, Bulfone-Paus S. Aged human skin accumulates mast cells with altered functionality that localize to macrophages and vasoactive intestinal peptide-positive nerve fibres. Br J Dermatol. 2019;180:849–58.
Ito N, Sugawara K, Bodó E, Takigawa M, van Beek N, Ito T, et al. Corticotropin-Releasing Hormone Stimulates the In Situ Generation of Mast Cells from Precursors in the Human Hair Follicle Mesenchyme. J Invest Dermatol. 2010;130:995–1004.
Kallman S. In discussion on Meyers J, Urticaria pigmentosa (case report). Arch Dermatol Chicago. 1960;81:161.
Hashimoto K, Tarnowski WM. Lever WF. Maturation and degranulation of mast cells in the human skin. Electron microscopic studies. Hautarzt. 1967;18:318–24.
Douaiher J, Succar J, Lancerotto L, Gurish MF, Orgill DP, Hamilton MJ, et al. Development of mast cells and importance of their tryptase and chymase serine proteases in inflammation and wound healing. Adv Immunol. 2014;122:211–52.
Weidner N, Austen KF. Heterogeneity of Mast cells at multiple body sites. Pathol Res Pract. 1993;189:156–62.
Flynn EA, Schwartz JL, Shklar G. Sequential mast cell infiltration and degranulation during experimental carcinogenesis. J Cancer Res Clin Oncol. 1991;117:115–22.
Coussens LM, Raymond WW, Bergers G, Laig-Webster M, Behrendtsen O, Werb Z, et al. Inflammatory mast cells up-regulate angiogenesis during squamous epithelial carcinogenesis. Genes Dev. 1999;13:1382–97.
Ribatti D, Crivellato E. Mast cells, angiogenesis, and tumour growth. Biochim Biophys Acta. 2012;1822:2–8.
Hart PH, Townley SL, Grimbaldeston MA, Khalil Z, Finlay-Jones JJ. Mast cells, neuropeptides, histamine, and prostaglandins in UV-induced systemic immunosuppression. Methods. 2002;28:79–89.
Asboe-Hansen G. The origin in synovial mucin; Ehrlich’s mast cell, a secretory element of the connective tissue. Ann Rheum Dis. 1950;9:149–58.
Grebennikov VA, Kuligin AG, Grebennikova NV. Relationship between the state of mast cells and the content of serotonin in the skin of patients with disseminated neurodermatitis. Vestn Dermatol Venerol. 1976:21–6.
Vadivel JK, Govindarajan M, Somasundaram E, Muthukrishnan A. Mast cell expression in oral lichen planus: A systematic review. J Investig Clin Dent. 2019;10:e12457.
Marone G, Casolaro V, Cirillo R, Stellato C, Genovese A. Pathophysiology of human basophils and mast cells in allergic disorders. Clin Immunol Immunopathol. 1989;50:S24-40.
Soter NA. Morphology of atopic eczema. Allergy. 1989;44(Suppl 9):16–9.
Macedo AC, Isaac L. Systemic lupus erythematosus and deficiencies of early components of the complement classical pathway. Front Immunol. 2016;7:55.
Brown M, O’Reilly S. The immunopathogenesis of fibrosis in systemic sclerosis. Clin Exp Immunol. 2019;195:310–21.
Aroni K, Voudouris S, Ioannidis E, Grapsa A, Kavantzas N, Patsouris E. Increased angiogenesis and mast cells in the centre compared to the periphery of vitiligo lesions. Arch Dermatol Res. 2010;302:601–7.
Ghaly NR, Roshdy OA, Nassar SA, Hamad SM, El-Shafei AM. Role of mast cells and T-lymphocytes in pemphigus vulgaris: significance of CD44 and the c-kit gene product (CD117). East Mediterr Health J. 2005;11:1009–17.
Ribatti D, Vacca A, Schiraldi G, Sorino S, Caprio F, Mazzotta F, et al. Pyogenic granuloma stimulates angiogenesis in the chick embryo chorioallantoic membrane. Int J Microcirc Clin Exp. 1996;16:82–8.
Antonopoulos D, Tsilioni I, Balatsos NAA, Gourgoulianis KI, Theoharides TC. The mast cell–neurofibromatosis connection. J Biol Regul Homeost Agents. 2019;33:657–9.
Diaconu NC, Kaminska R, Naukkarinen A, Harvima RJ, Harvima IT. The increase in tryptase- and chymase-positive mast cells is associated with partial inactivation of chymase and increase in protease inhibitors in basal cell carcinoma. J Eur Acad Dermatol Venereol. 2007;21:908–15.
Harvima IT, Naukkarinen A, Paukkonen K, Harvima RJ, Aalto ML, Schwartz LB, et al. Mast cell tryptase and chymase in developing and mature psoriatic lesions. Arch Dermatol Res. 1993;285:184–92.
Huttunen M, Harvima IT. Mast cell tryptase and chymase in chronic leg ulcers: chymase is potentially destructive to epithelium and is controlled by proteinase inhibitors. Br J Dermatol. 2005;152:1149–60.
Huttunen M, Naukkarinen A, Horsmanheimo M, Harvima IT. Transient production of stem cell factor in dermal cells but increasing expression of Kit receptor in mast cells during normal wound healing. Arch Dermatol Res. 2002;294:324–30.
Yamamoto T, Katayama I, Nishioka K. Expression of stem cell factor in basal cell carcinoma. Br J Dermatol. 1997;137:709–13.
Netscher DT, Spira M. Basal cell carcinoma: an overview of tumor biology and treatment. Plast Reconstr Surg. 2004;113:74e–94e.
Rudolph R, Zelac DE. Squamous cell carcinoma of the skin. Plast Reconstr Surg. 2004:82e–94e.
Wagner JD, Gordon MS, Chuang T-Y, Coleman JJ. Current therapy of cutaneous melanoma. Plast Reconstr Surg. 2000;105:1774–99.
Cawley EP. Association of tissue mast cells and skin tumors. Arch Dermatol. 1961;83:92.
Humphreys TR, Monteiro MR, Murphy GF. Mast cells and dendritic cells in basal cell carcinoma stroma. Dermatol Surg. 2000;26:200–4.
Janowski P, Strzelecki M, Brzezinska-Blaszczyk E, Zalewska A. Computer analysis of normal and basal cell carcinoma mast cells. Med Sci Monit. 2001;7:260–5.
Aoki M, Pawankar R, Niimi Y, Kawana S. Mast cells in basal cell carcinoma express VEGF, IL-8 and RANTES. Int Arch Allergy Immunol. 2003;130:216–23.
Claudatus JC Jr, d’Ovidio R, Lospalluti M, Meneghini CL. Skin tumors and reactive cellular infiltrate: further studies. Acta Derm Venereol. 1986;66:29–34.
Iamaroon A, Pongsiriwet S, Jittidecharaks S, Pattanaporn K, Prapayasatok S, Wanachantararak S. Increase of mast cells and tumor angiogenesis in oral squamous cell carcinoma. J Oral Pathol Med. 2003;32:195–9.
Gomes APN, Johann JE, Lovato GG, Ferreira AM. Comparative analysis of the mast cell density in normal oral mucosa, actinic cheilitis and lip squamous cell carcinoma. Braz Dent J. 2008;19:186–9.
Cheema VS, Ramesh V, Balamurali PD. The relevance of mast cells in oral squamous cell carcinoma. J Clin Diag Res JCDRR. 2012;6:1803–7.
Kathuriya PT, Bartake AR, Palaskar SJ, Narang BR, Patil SS, Pawar RB. Cd34 and mast cell analysis in normal oral mucosa and different grades of oral squamous cell carcinoma: a comparative study. J Clin Diag Res JCDR 2015;9:ZC61–4.
Dvorak AM, Mihm MC, Osage JE, Dvorak HF. Melanoma. An ultrastructural study of the host inflammatory and vascular responses. J Invest Dermatol. 1980;75:388–93.
Reed JA, McNutt NS, Bogdany JK, Albino AP. Expression of the mast cell growth factor interleukin-3 in melanocytic lesions correlates with an increased number of mast cells in the perilesional stroma: implications for melanoma progression. J Cutan Pathol. 2006;23:495–505.
Ribatti D, Ennas MG, Vacca A, Ferreli F, Nico B, Orru S, et al. Tumor vascularity and tryptase-positive mast cells correlate with a poor prognosis in melanoma. Eur J Clin Invest. 2003;33:420–5.
Ribatti D, Vacca A, Ria R, Marzullo A, Nico B, Filotico R, et al. Neovascularisation, expression of fibroblast growth factor-2, and mast cells with tryptase activity increase simultaneously with pathological progression in human malignant melanoma. Eur J Cancer. 2003;39:666–74.
Grimbaldeston MA, Pearce AL, Robertson BO, Coventry BJ, Marshman G, Finlay-Jones JJ, et al. Association between melanoma and dermal mast cell prevalence in sun-unexposed skin. Br J Dermatol. 2004;150:895–903.
Grimbaldeston MA, Skov L, Finlay-Jones JJ, Hart PH. Increased dermal mast cell prevalence and susceptibility to development of basal cell carcinoma in humans. Methods. 2002;28:90–6.
Demis DJ. The mastocytosis syndrome: clinical and biological studies. Ann Intern Med. 1963;59:194.
Castells M, Metcalfe DD, Escribano L. Diagnosis and treatment of cutaneous mastocytosis in children: practical recommendations. Am J Clin Dermatol. 2011;12:259–70.
Bodemer C, Hermine O, Palmerini F, Yang Y, Grandpeix-Guyodo C, Leventhal PS, et al. Pediatric mastocytosis is a clonal disease associated with D816V and other activating c-KIT mutations. J Invest Dermatol. 2010;130:804–15.
Valent P, Horny H-P, Escribano L, Longley BJ, Li CY, Schwartz LB, et al. Diagnostic criteria and classification of mastocytosis: a consensus proposal. Leuk Res. 2001;25:603–25.
Ribatti D, Nico B, Finato N, Crivellato E, Beltrami CA. Co-localization of tryptase and cathepsin-G in mast cells in cutaneous mastocytosis. Cancer Lett. 2009;279:209–12.
Ranieri G, Passantino L, Patruno R, Passantino G, Jirillo F, Catino A, et al. The dog mast cell tumour as a model to study the relationship between angiogenesis, mast cell density and tumour malignancy. Oncol Rep. 2003;10:1189–93.
Patruno R, Arpaia N, Gadaleta CD, Passantino L, Zizzo N, Misino A, et al. VEGF concentration from plasma-activated platelets rich correlates with microvascular density and grading in canine mast cell tumour spontaneous model. J Cell Mol Med. 2009;13:555–61.
Ranieri G, Marech I, Pantaleo M, Piccinno M, Roncetti M, Mutinati M, et al. In vivo model for mastocytosis: a comparative review. Crit Rev Oncol Hematol. 2015;93:159–69.
Wang Z, Mascarenhas N, Eckmann L, Miyamoto Y, Sun X, Kawakami T, et al. Skin microbiome promotes mast cell maturation by triggering stem cell factor production in keratinocytes. J Allergy Clin Immunol. 2017;139(1205–16):e6.
Ribatti D. Mast cells as therapeutic target in cancer. Eur J Pharmacol. 2016;778:152–7.
Somasundaram R, Connelly T, Choi R, Choi H, Samarkina A, Li L, et al. Tumor-infiltrating mast cells are associated with resistance to anti-PD-1 therapy. Nat Commun. 2021;12:346.
Funding
There are not funding to support this work.
Author information
Authors and Affiliations
Contributions
DR involved in conceptualization, conceived the study and wrote the ms; RT, TA and EC involved in revise and editing the ms.
Corresponding author
Ethics declarations
Conflict of interest
There are not conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ribatti, D., Tamma, R., Annese, T. et al. The role of mast cells in human skin cancers. Clin Exp Med 21, 355–360 (2021). https://doi.org/10.1007/s10238-021-00688-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-021-00688-x