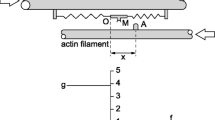
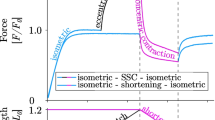
Abstract
Muscles exhibit highly complex, multi-scale architecture with thousands of muscle fibers, each with different properties, interacting with each other and surrounding connective structures. Consequently, the results of single-fiber experiments are scarcely linked to the macroscopic or whole muscle behavior. This is especially true for human muscles where it would be important to understand of how skeletal muscles disorders affect patients’ life. In this work, we developed a mathematical model to study how fast and slow muscle fibers, well characterized in single-fiber experiments, work and generate together force and displacement in muscle bundles. We characterized the parameters of a Hill-type model, using experimental data on fast and slow single human muscle fibers, and comparing experimental data with numerical simulations obtained from finite element (FE) models of single fibers. Then, we developed a FE model of a bundle of 19 fibers, based on an immunohistochemically stained cross section of human diaphragm and including the corresponding properties of each slow or fast fiber. Simulations of isotonic contractions of the bundle model allowed the generation of its apparent force–velocity relationship. Although close to the average of the force–velocity curves of fast and slow fibers, the bundle curve deviates substantially toward the fast fibers at low loads. We believe that the present model and the characterization of the force–velocity curve of a fiber bundle represents the starting point to link the single-fiber properties to those of whole muscle with FE application in phenomenological models of human muscles.





Similar content being viewed by others
References
Biewener AA, Wakeling JM, Lee SS, Arnold AS (2014) Validation of hill-type muscle models in relation to neuromuscular recruitment and force–velocity properties: predicting patterns of in vivo muscle force. Integr Comp Biol 54:1072–1083. doi:10.1093/icb/icu070
Bottinelli R, Canepari M, Pellegrino MA, Reggiani C (1996) Force–velocity properties of human skeletal muscle fibres: myosin heavy chain isoform and temperature dependence. J Physiol 495:573
Bottinelli R, Reggiani C (2000) Human skeletal muscle fibres: molecular and functional diversity. Prog Biophys Mol Biol 73:195–262. doi:10.1016/S0079-6107(00)00006-7
Capitanio M, Canepari M, Maffei M et al (2012) Ultrafast force-clamp spectroscopy of single molecules reveals load dependence of myosin working stroke. Nat Methods 9:1013–1019. doi:10.1038/nmeth.2152
Cleworth DR, Edman KaP (1972) Changes in sarcomere length during isometric tension development in frog skeletal muscle. J Physiol 227:1–17. doi:10.1113/jphysiol.1972.sp010016
Edman KA (1979) The velocity of unloaded shortening and its relation to sarcomere length and isometric force in vertebrate muscle fibres. J Physiol 291:143–159
Ehret AE, Böl M, Itskov M (2011) A continuum constitutive model for the active behaviour of skeletal muscle. J Mech Phys Solids 59:625–636. doi:10.1016/j.jmps.2010.12.008
Ervasti JM (2003) Costameres: the Achilles’ Heel of Herculean muscle. J Biol Chem 278:13591–13594. doi:10.1074/jbc.R200021200
Hamouda A, Kenney L, Howard D (2016) Dealing with time-varying recruitment and length in Hill-type muscle models. J Biomech. doi:10.1016/j.jbiomech.2016.08.030
Hauraix H, Nordez A, Guilhem G et al (2015) In vivo maximal fascicle-shortening velocity during plantar flexion in human. J Appl Physiol. doi:10.1152/japplphysiol.00542.2015
He ZH, Bottinelli R, Pellegrino MA et al (2000) ATP consumption and efficiency of human single muscle fibers with different myosin isoform composition. Biophys J 79:945. doi:10.1016/S0006-3495(00)76349-1
Hill AV (1938) The heat of shortening and the dynamic constants of muscle. Proc R Soc Lond B Biol Sci 126:136–195. doi:10.1098/rspb.1938.0050
Holt NC, Wakeling JM, Biewener AA (2014) The effect of fast and slow motor unit activation on whole-muscle mechanical performance: the size principle may not pose a mechanical paradox. Proc R Soc B Biol Sci. doi:10.1098/rspb.2014.0002
Hooijman PE, Beishuizen A, Witt CC et al (2015) Diaphragm muscle fiber weakness and ubiquitin–proteasome activation in critically Ill patients. Am J Respir Crit Care Med 191:1126–1138. doi:10.1164/rccm.201412-2214OC
Johansson T, Meier P, Blickhan R (2000) A finite-element model for the mechanical analysis of skeletal muscles. J Theor Biol 206:131–149. doi:10.1006/jtbi.2000.2109
Josephson RK, Edman KAP (1988) The consequences of fibre heterogeneity on the force-velocity relation of skeletal muscle. Acta Physiol Scand 132:341–352. doi:10.1111/j.1748-1716.1988.tb08338.x
Kitamura K, Tokunaga M, Iwane AH, Yanagida T (1999) A single myosin head moves along an actin filament with regular steps of 5.3 nanometres. Nature 397:129–134. doi:10.1038/16403
Kojic M, Mijailovic S, Zdravkovic N (1998) Modelling of muscle behaviour by the finite element method using Hill’s three-element model. Int J Numer Methods Eng 43:941–953. doi:10.1002/(SICI)1097-0207(19981115)43:5<d941::AID-NME435>3.0.CO;2-3
Lee SSM, Arnold AS, Miara M de B et al (2013) Accuracy of gastrocnemius muscles forces in walking and running goats predicted by one-element and two-element Hill-type models. J Biomech 46:2288–2295. doi:10.1016/j.jbiomech.2013.06.001
Levine S, Bashir MH, Clanton TL et al (2013) COPD elicits remodeling of the diaphragm and vastus lateralis muscles in humans. J Appl Physiol 114:1235–1245. doi:10.1152/japplphysiol.01121.2012
Lieber RL, Ward SR (2011) Skeletal muscle design to meet functional demands. Philos Trans R Soc B Biol Sci 366:1466. doi:10.1098/rstb.2010.0316
Marsh RL, Bennett AF (1986) Thermal dependence of contractile properties of skeletal muscle from the lizard Sceloporus occidentalis with comments on methods for fitting and comparing force–velocity curves. J Exp Biol 126:63
Sanchez GN, Sinha S, Liske H et al (2015) In vivo imaging of human sarcomere twitch dynamics in individual motor units. Neuron 88:1109–1120. doi:10.1016/j.neuron.2015.11.022
Sharafi B, Blemker SS (2010) A micromechanical model of skeletal muscle to explore the effects of fiber and fascicle geometry. J Biomech 43:3207–3213. doi:10.1016/j.jbiomech.2010.07.020
Spudich JA (1994) How molecular motors work. Nature 372:515–518. doi:10.1038/372515a0
Tang CY, Zhang G, Tsui CP (2009) A 3D skeletal muscle model coupled with active contraction of muscle fibres and hyperelastic behaviour. J Biomech 42:865–872. doi:10.1016/j.jbiomech.2009.01.021
Virgilio KM, Martin KS, Peirce SM, Blemker SS (2015) Multiscale models of skeletal muscle reveal the complex effects of muscular dystrophy on tissue mechanics and damage susceptibility. Interface Focus 5:20140080. doi:10.1098/rsfs.2014.0080
Wakeling JM, Lee SSM, Arnold AS et al (2012) A muscle’s force depends on the recruitment patterns of its fibers. Ann Biomed Eng 40:1708–1720. doi:10.1007/s10439-012-0531-6
Acknowledgements
LM work was supported by European Commission, Seventh Framework Programme (FP7/2007-2013) under Grant Agreement No. 600376.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Marcucci, L., Reggiani, C., Natali, A.N. et al. From single muscle fiber to whole muscle mechanics: a finite element model of a muscle bundle with fast and slow fibers. Biomech Model Mechanobiol 16, 1833–1843 (2017). https://doi.org/10.1007/s10237-017-0922-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-017-0922-6