Abstract
The continuous use of single-use petrochemical-based plastics has created a global crisis with a significant buildup of plastic pollution. The use of biomass resources as a replacement source of plastic constituents, namely cellulose and starch, may contribute to alleviating the crisis. In this study, cellulose nanofiber-reinforced starch films were produced and studied for their pH response in terms of their swelling behavior and wet tensile strength in both freshwater and seawater conditions. Mechanically fibrillated cellulose nanofibers (MCNFs) were blended with dialdehyde starch (Di-aldS) and made into MCNF/Di-aldS films. The films were found to swell more and had a lower wet tensile strength at pH of 9. The film’s strength reduced to 1.60 MPa in basic conditions, 80% less than in acidic environment. This is related to hemiacetal crosslinking, resulted from the interactions between the modified moieties of the starches and cellulose nanofibers. Such bioplastics enhance the reusability of cellulose nanofibers and have the potential to replace conventional petrochemical plastics to create a carbon–neutral circular society.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Plastic packaging has played an important role in preservation, protection, handling, and consumer convenience in several industries. Most of the commercial plastic packaging materials are made from petrochemical polymers, namely polyolefin, polyester, polyvinyl chloride, and polystyrene [1,2,3,4]. These plastics commonly serve their single-use functions and are thrown out after use [5]. However, because of their inert nature and very slow degradation, there is a buildup of these plastics globally, along with the amount of microplastics worldwide from the plastics’ fragmentation [6,7,8]. Extensive studies into the potential replacement of these conventional polymers have been explored. Biopolymers extracted from renewable biomass resources have attracted much interest for their use as single-use packaging materials, mainly because of their biodegradability and renewability [9, 10].
Cellulose and starch-based films are considered suitable for single-use packaging owing to enzymatic hydrolysis by cellulase and amylase [11, 12]. On top of cellulose and starch being the most naturally abundant biomass resources available, their ability to degrade by bacteria, E. coli, and fungus pushes their suitability as components for the production of bioplastics [13,14,15,16]. However, their application is limited by their opacity, poor mechanical properties, and poor freshwater durability.
In a previous study, a water-stable transparent film was prepared by introducing aldehyde moieties to polysaccharides which prepared environmentally friendly cross linkers, that have been found to improve the films’ overall properties [17]. Starch films cross-linked by oxidized sucrose (di-aldehyde sucrose) [18], di-aldehyde starch [19], and di-aldehyde cellulose (~ 100% oxidized, amorphous) [20] exhibited water stability from hemiacetal crosslinking between the polysaccharides. Studies that have used modified starch to blend with (2,2,6,6-tetramethylpiperidin-1-yl)oxyl (TEMPO)-oxidized cellulose nanofibers (TCNF) produced films with high optical transparency (~ 90%) and a wet strength of ~ 300 kPa owing to the hemiacetal binding between the TCNF and starch [21,22,23]. A study by Soni et al. [24], showed that through the introduction of aldehyde moieties on the cellulose nanofiber surface, the films’ wet strength increased from ~ 2 MPa to ~ 35 MPa after blending with starch. The di-aldehyde moieties on cellulose reduced marine-microbial degradability compared to native cellulose but through the incorporation of starch, the microbial attachment and degradability was improved. Anionic TCNF was blended with cationic starch to prepare poly-ion complex (PIC) based films, which exhibited rapid disintegration in the marine environment owing to the presence of counterions [25]. These kinds of starch blended TCNF films have higher optical transparency, mechanical strength, water durability, and degradability. However, the large-scale production of cellulose-nanofiber is economically expensive, hazardous to the environment, and dangerous for forestry.
Recycling of cellulose has been reported to be possible by dissolving in different chloride salts, such as LiCl [26, 27], ZnCl2 [28], CaCl2 [29], and FeCl3 [30]. Moreover, NaOH/Urea solution [31,32,33] and ionic liquid [34,35,36] have also been widely reported to be used for recycling cellulose. However, such a recycling process breaks down the hydrogen bonding between the cellulose chains for dissolution, thus diminishes cellulose’s crystallinity during the process [37]. Therefore, recycling of cellulose nanofibers while maintaining its properties as aspect ratio, strength, and crystallinity, is proven to be a challenge with previously reported procedures. According to earlier studies that have stated the presence of hemiacetal crosslinking within the fiber network [38, 39], this chemical crosslinking has been found to alter its strengths with the regulation of different pH conditions [40,41,42]. So, through the introduction of inter- and intra-molecular hemiacetal crosslinking, a potential method of recycling the cellulose nanofibers while maintaining its properties can be explored.
In this study, a cellulose nanofiber-reinforced starch film that exhibited adequate freshwater durability and pH-responsive weakening and disintegration was prepared. Mechanically fibrillated cellulose nanofibers (MCNF) were blended with di-aldehyde starch (Di-aldS) to develop a MCNF/Di-aldS film. The film was kept under different pH conditions (4, 7, and 9) in both freshwater and seawater to analyze the freshwater durability and pH-responsive weakening. Moreover, rheology and Fourier transform infrared (FTIR) spectroscopy were performed to confirm the pH-responsive formation and disintegration of hemiacetal bonding between cellulose and starch. Thus, this study presents a novel and low-cost approach to developing pH-responsive disintegratable films that will increase the reusability of cellulose nanofibers.
Materials and methods
Materials
Commercially available cellulose powder was supplied by Nacalai Tesque, Japan. Tapioca starch modified with dialdehyde moieties (Di-aldS, DS: 0.04) was supplied by Nihon Shokuhin Kako Co. Ltd., Japan, and was used as received (Table 1). All reagents were of laboratory grade and were used as received. Artificial seawater solution was made using Daigo’s Artificial Seawater SP for Marine Microalgae Medium from Fujifilm Wako Chemicals, Japan. Milli-Q water was used throughout the experiment.
Preparation of mechanically fibrillated cellulose nanofiber (MCNF)
Cellulose powder (4.0 g) was added to 500 mL of Mili-Q water and dispersed through magnetic stirring for 1 h at temperatures between 25 to 27 °C. The obtained dispersion was processed via wet pulverization, which mechanically disintegrated the cellulose into cellulose nanofibers. During this process, the dispersion was first pressurized to approximately 245 MPa and is fed through a 100-μm aperture nozzle into the device chamber [43]. The dispersion was then pulverized with a ceramic ball using a Star Burst Mini wet pulverizing and dispersing device (Sugino Machine Co., Ltd., Japan) equipped with a ceramic ball collision chamber. This disintegration was repeated approximately 30 times to obtain a homogenously fibrillated suspension of cellulose nanofibers. The resultant suspension is referred to as mechanically fibrillated cellulose nanofibers (MCNF). Before the experiment, the suspension was thoroughly stirred to completely homogenize the nanofibrils.
Preparation of Di-aldS suspension
Starch powder (3.0 g) was added to 100 mL of Mili-Q water. The suspension was mixed using magnetic stirring at 90 °C for 1 h to obtain a homogeneous solution. The prepared solution was stored at room temperature for further experiments.
Preparation of MCNF/Di-aldS composite film
Prepared MCNF dispersion (1.0%, W/V) and Di-aldS solution (3%, W/V) were blended together in a 1: 0.6 weight ratio and stirred until a homogeneous dispersion was obtained. The dispersion was also centrifuged to remove any air bubbles before casting. The MCNF/Di-aldS mixer was cast in a Teflon PFA Petri dish and baked at 40 °C for 6 h [44]. Dried films were cut accordingly for the respective experiments.
Characterization
A stress-controlled rheometer (Haake Rheo Stress 6000, Thermo Scientific, USA) was used to examine the rheological behavior of the obtained MCNF/Di-aldS suspensions under different pH conditions. All measurements were conducted with an aluminum cone-plate geometry (diameter, 40 mm; angle, 2°). The strain amplitude value that was selected was 0.01, which was in the linear viscoelastic region for all samples. Frequency sweeps were performed from 0.1 to 10 rad/s. A temperature controller (Haake UTM controller, Thermo Scientific, USA) was used to maintain the temperature (25 °C) during the measurements. The MCNF/Di-aldS suspensions under different pH conditions were lyophilized and analyzed using FTIR (Nicolet iS5, Thermo Scientific, USA) to confirm the aldehyde moieties and were analyzed using OMNIC software.
A swelling test was performed in freshwater and seawater under different pH (4, 7, and 9) conditions. Dried film specimens (2 cm × 2 cm) were immersed in the water. After a fixed time period, swollen samples were gently taken out using tweezers, and excess water was removed with tissue paper before the samples were weighed. The swelling ratio (SR) was calculated as follows:
where Wds and Wss are the weights of the dried and swollen samples, respectively [45].
The mechanical properties of the swollen films were evaluated by the universal testing machine (UTM) (EZ Graph, Shimadzu, Japan) with a 10-N load cell and a strain rate of 5.0 mm/min. Five specimens (20 mm × 5 mm) were cut from each film and were kept in freshwater and seawater under different pH (4, 7, and 9) conditions for 2 h. A micrometer was used to measure the thickness of the swollen films before the tensile test. Measurements were taken at four different positions for each specimen, and the average value was used for the calculation.
Results and discussion
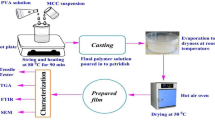
The film was prepared by mixing MCNF with dialdehyde starch (Di-aldS) at a weight ratio of 1.5:1. A general experimental scheme is illustrated in Fig. 1. These films were then immersed in both freshwater and seawater at pH values of 4, 7, and 9 and were then evaluated for their swelling behavior and wet strength, as shown in Fig. 2 and tabulated in Table 2.
To confirm the strengthening and weakening of intramolecular hemiacetal bonding in different pH conditions, the swelling ratio and tensile strengths of each film were measured. The MCNF/Di-aldS films reached saturated swelling in freshwater with a pH of 7 after a 2-h immersion with a swelling ratio of 243%. The swollen films were also subjected to the UTM with a 10-N load cell at a strain rate of 5 mm/min. The stress–strain curves were used to determine their wet tensile strength. The resulting specimens for the films with a pH of 7 had a value of 4.31 MPa. However, once placed in an acidic solution with a pH of 4, their swelling ratios reduced to 204%. With this reduction in swelling, the films displayed a higher wet strength of 7.03 MPa. While in an alkaline solution with a pH of 9, the films exhibited more swelling with a value of 302%. This resulted in a wet strength of 1.60 MPa for the film.
The film was also kept in artificial seawater under different pH conditions (pH 4, 8, and 9) to investigate the weakening of the films owing to the pH in marine environments. The swelling behavior and wet strength are shown in Fig. 2b, d and are summarized in Table 2. The films reached saturation in artificial seawater (pH 8) after being immersed for 5 h with a swelling ratio of 312%. Increasing the pH to 9 also increased the film’s swelling to 364%, while decreasing the pH resulted in a reduction in swelling. The changes in pH resulted in similar trends being observed compared with the films’ immersion in freshwater. With respect to the wet strength of the films, for a pH of 9, the value decreased to 2.66 MPa, then to 6.98 MPa for a pH of 4.
This phenomenon can be explained through the intramolecular hemiacetal crosslinking that occurs between the modified moieties of the starches and the hydroxyl groups of the fibers within the film’s network [21]. The incorporation of di-aldehyde starch, which acts as a flexible binder, improves the films’ water durability through hemiacetal crosslinking [21], therefore exhibiting less swelling in general. As mentioned before, the formation of hemiacetal bonding depends on pH conditions. The additional H+ ions available in acidic conditions allows the hemiacetal crosslinking to be more readily made as H+ ions function as catalysts for the interacting groups to convert to hemiacetal groups; this results in the films’ heightened integrity in water, thus taking up less water. The films became more rigid and had a higher wet tensile strength as a result. This was also the case for basic conditions, whereby with the relatively low number of H+ ions present, the aldehyde moieties of the network were not able to convert to hemiacetal crosslinks, thus resulted in a higher water uptake. The softer, swollen films would then lack rigidity and integrity, therefore, led to a weaker wet tensile strength. A rheology analysis was also performed to investigate the pH responsive disintegration of the hemiacetal crosslinking in MCNF/Di-aldS suspension. The results of this test as storage moduli (G′) and loss moduli (G″) are displayed in Fig. 3 and tabulated in Table 2. With the increase in pH, both the storage and loss moduli of the suspension decreased, confirming the effect of pH on the formation and disintegration of hemiacetal crosslinking, and an illustrative representation is shown in Fig. 4. Moreover, FTIR analysis of the lyophilized suspension was performed to analyze reversal of hemiacetal bonding in alkaline conditions. With the increase in the C=O peak (1732 cm−1) intensity observed for a pH of 9 and the decrease in intensity of the same peak at a pH of 4, this result confirms that there is a reversal of hemiacetal bonding corresponding to higher pH conditions.
Previous reports have shown use of metal chlorides, ionic liquid, and strong basic conditions for weakening of cellulose nanofiber reinforced films. However, these processes are expensive, hazardous and diminishes cellulose’s properties. MCNF/Di-aldS film with introduction of hemiacetal crosslinking has shown weakening of the films at higher pH conditions. Also, the film lost its strength, with a reduction to 1.60 MPa after 2 h immersion in alkaline water. Such weakened films can completely be disintegrated via mechanical stirring for reusability. In addition, the film rapidly loses strength in seawater compared to other packaging materials [46]. These polysaccharide-based, freshwater-durable, and pH-responsive disintegrable films have the potential to be next-generation packaging materials that can help to realize the reusability of cellulose nanofibers and reduce deforestation.
Conclusion
This study investigated the effects of pH on films made of mechanically fibrillated cellulose nanofibers (MCNF) blended with dialdehyde starch (Di-aldS). MCNF/Di-aldS film strength reduced to 1.60 MPa at pH 9 and disintegration under alkaline conditions. Moreover, rheology and FTIR confirmed the reversal of hemiacetal bonding in basic environments. This phenomenon will enhance the recyclability of cellulose nanofiber compared to existing methods. Further studies of MCNF/Di-aldS films will provide insight for further research on pH-responsive disintegrating, reusable and greener packaging films.
References
Marsh K, Bugusu B (2007) Food packaging—roles, materials, and environmental issues. J Food Sci 72(3):R39–R55. https://doi.org/10.1111/j.1750-3841.2007.00301.x
Cole M, Lindeque P, Halsband C, Galloway TS (2011) Microplastics as contaminants in the marine environment: a review. Mar Pollut Bull 62(12):2588–2597. https://doi.org/10.1016/j.marpolbul.2011.09.025
Browne MA, Dissanayake A, Galloway TS, Lowe DM, Thompson RC (2008) Ingested microscopic plastic translocates to the circulatory system of the mussel, Mytilus edulis (L.). Environ Sci Technol 42(13):5026–5031. https://doi.org/10.1021/es800249a
Browne MA et al (2011) Accumulation of microplastic on shorelines woldwide: sources and sinks. Environ Sci Technol 45(21):9175–9179. https://doi.org/10.1021/es201811s
Mathalon A, Hill P (2014) Microplastic fibers in the intertidal ecosystem surrounding Halifax Harbor, Nova Scotia. Mar Pollut Bull 81(1):69–79. https://doi.org/10.1016/j.marpolbul.2014.02.018
Rochman CM et al (2015) Anthropogenic debris in seafood: plastic debris and fibers from textiles in fish and bivalves sold for human consumption. Sci Rep 5(1):14340. https://doi.org/10.1038/srep14340
Wang J, Tan Z, Peng J, Qiu Q, Li M (2016) The behaviors of microplastics in the marine environment. Mar Environ Res 113:7–17. https://doi.org/10.1016/j.marenvres.2015.10.014
Eerkes-Medrano D, Thompson RC, Aldridge DC (2015) Microplastics in freshwater systems: a review of the emerging threats, identification of knowledge gaps and prioritisation of research needs. Water Res 75:63–82. https://doi.org/10.1016/j.watres.2015.02.012
Guillaume SM (2022) Sustainable and degradable plastics. Nat Chem 14(3):245–246. https://doi.org/10.1038/s41557-022-00901-8
Bhargava N, Sharanagat VS, Mor RS, Kumar K (2020) Active and intelligent biodegradable packaging films using food and food waste-derived bioactive compounds: a review. Trends Food Sci Technol 105:385–401. https://doi.org/10.1016/j.tifs.2020.09.015
Bubner P, Plank H, Nidetzky B (2013) Visualizing cellulase activity. Biotechnol Bioeng 110(6):1529–1549. https://doi.org/10.1002/bit.24884
Lin X, Wu L, Huang S, Qin Y, Qiu X, Lou H (2019) Effect of lignin-based amphiphilic polymers on the cellulase adsorption and enzymatic hydrolysis kinetics of cellulose. Carbohydr Polym 207:52–58. https://doi.org/10.1016/j.carbpol.2018.11.070
Sheldon RA, Norton M (2020) Green chemistry and the plastic pollution challenge: towards a circular economy. Green Chem 22(19):6310–6322. https://doi.org/10.1039/D0GC02630A
Ahola S, Turon X, Österberg M, Laine J, Rojas OJ (2008) Enzymatic hydrolysis of native cellulose nanofibrils and other cellulose model films: effect of surface structure. Langmuir 24(20):11592–11599. https://doi.org/10.1021/la801550j
Salehudin MH, Salleh E, Mamat SNH, Muhamad II (2014) Starch based active packaging film reinforced with empty fruit bunch (EFB) cellulose nanofiber. Procedia Chem 9:23–33. https://doi.org/10.1016/j.proche.2014.05.004
Othman SH, Majid NA, Tawakkal ISMA, Basha RK, Nordin N, Shapi’i RA (2019) Tapioca starch films reinforced with microcrystalline cellulose for potential food packaging application. Food Sci Technol 39(3):605–612. https://doi.org/10.1590/fst.36017
Yu J, Chang PR, Ma X (2010) The preparation and properties of dialdehyde starch and thermoplastic dialdehyde starch. Carbohydr Polym 79(2):296–300. https://doi.org/10.1016/j.carbpol.2009.08.005
Xu H, Canisag H, Mu B, Yang Y (2015) Robust and flexible films from 100% starch cross-linked by biobased disaccharide derivative. ACS Sustain Chem Eng 3(11):2631–2639. https://doi.org/10.1021/acssuschemeng.5b00353
Rhim JW, Gennadios A, Weller CL, Cezeirat C, Hanna MA (1998) Soy protein isolate–dialdehyde starch films. Ind Crops Prod 8(3):195–203. https://doi.org/10.1016/S0926-6690(98)00003-X
Plappert SF et al (2018) Transparent, flexible, and strong 2,3-dialdehyde cellulose films with high oxygen barrier properties. Biomacromol 19(7):2969–2978. https://doi.org/10.1021/acs.biomac.8b00536
Soni R, Asoh T-A, Uyama H (2020) Cellulose nanofiber reinforced starch membrane with high mechanical strength and durability in water. Carbohydr Polym 238:116203. https://doi.org/10.1016/j.carbpol.2020.116203
Soni R, Asoh T-A, Hsu Y-I, Shimamura M, Uyama H (2020) Effect of starch retrogradation on wet strength and durability of cellulose nanofiber reinforced starch film. Polym Degrad Stab 177:109165
Soni R, Hsu Y-I, Asoh T-A, Uyama H (2021) Synergistic effect of hemiacetal crosslinking and crystallinity on wet strength of cellulose nanofiber-reinforced starch films. Food Hydrocoll 120:106956. https://doi.org/10.1016/j.foodhyd.2021.106956
Soni R, Asoh T-A, Hsu Y-I, Uyama H (2022) Freshwater-durable and marine-degradable cellulose nanofiber reinforced starch film. Cellulose 29(3):1667–1678
Soni R, Hsu Y-I, Asoh T-A, Uyama H (2022) Cellulose nanofiber reinforced starch film with rapid disintegration in marine environments. J Appl Polym Sci 139(32):e52776
Soykeabkaew N, Nishino T, Peijs T (2009) All-cellulose composites of regenerated cellulose fibres by surface selective dissolution. Compos Part A Appl Sci Manuf 40(4):321–328. https://doi.org/10.1016/j.compositesa.2008.10.021
Han Q, Gao X, Zhang H, Chen K, Peng L, Jia Q (2019) Preparation and comparative assessment of regenerated cellulose films from corn (Zea mays) stalk pulp fines in DMAc/LiCl solution. Carbohydr Polym 218:315–323. https://doi.org/10.1016/j.carbpol.2019.04.083
Xu Q, Chen C, Rosswurm K, Yao T, Janaswamy S (2016) A facile route to prepare cellulose-based films. Carbohydr Polym 149:274–281. https://doi.org/10.1016/j.carbpol.2016.04.114
Hietala M, Varrio K, Berglund L, Soini J, Oksman K (2018) Potential of municipal solid waste paper as raw material for production of cellulose nanofibres. Waste Manage 80:319–326. https://doi.org/10.1016/j.wasman.2018.09.033
Yang W et al (2023) Cellulose nanocrystal preparation via rapid hydrolysis of wood cellulose fibers using recyclable molten ferric chloride hexahydrate. ACS Sustain Chem Eng 11(27):10172–10182. https://doi.org/10.1021/acssuschemeng.3c02460
Cai J et al (2008) Dynamic self-assembly induced rapid dissolution of cellulose at low temperatures. Macromolecules 41(23):9345–9351. https://doi.org/10.1021/ma801110g
Cai J, Zhang L (2005) Rapid dissolution of cellulose in LiOH/urea and NaOH/urea aqueous solutions. Macromol Biosci 5(6):539–548. https://doi.org/10.1002/mabi.200400222
Cai J, Zhang L, Zhou J, Li H, Chen H, Jin H (2004) Novel fibers prepared from cellulose in NaOH/urea aqueous solution. Macromol Rapid Commun 25(17):1558–1562. https://doi.org/10.1002/marc.200400172
Cao Y, Li H, Zhang Y, Zhang J, He J (2010) Structure and properties of novel regenerated cellulose films prepared from cornhusk cellulose in room temperature ionic liquids. J Appl Polym Sci 116(1):547–554. https://doi.org/10.1002/app.31273
Sixta H et al (2015) Ion cell-F: a high-strength regenerated cellulose fibre. Nord Pulp Paper Res J 30(1):43–57. https://doi.org/10.3183/npprj-2015-30-01-p043-057
Liu G, Yu C, Chen C, Ma W, Ji H, Zhao J (2011) A new type of covalent-functional graphene donor-acceptor hybrid and its improved photoelectrochemical performance. Sci China Chem 54(10):1622–1626. https://doi.org/10.1007/s11426-011-4366-z
Ghasemi M, Alexandridis P, Tsianou M (2018) Dissolution of cellulosic fibers: impact of crystallinity and fiber diameter. Biomacromol 19(2):640–651. https://doi.org/10.1021/acs.biomac.7b01745
DiFlavio J-L, Pelton R, Leduc M, Champ S, Essig M, Frechen T (2007) The role of mild TEMPO–NaBr–NaClO oxidation on the wet adhesion of regenerated cellulose membranes with polyvinylamine. Cellulose 14(3):257–268. https://doi.org/10.1007/s10570-006-9104-x
Erlandsson J et al (2018) On the mechanism behind freezing-induced chemical crosslinking in ice-templated cellulose nanofibril aerogels. J Mater Chem A Mater 6(40):19371–19380. https://doi.org/10.1039/C8TA06319B
Przystas TJ, Fife TH (1981) The mechanism of hemiacetal decomposition. Substituent effects in breakdown of substituted benzaldehyde ethyl hemiacetals. J Am Chem Soc 103(16):4884–4890. https://doi.org/10.1021/ja00406a036
Sorensen PE, Jencks WP (1987) Acid- and base-catalyzed decomposition of acetaldehyde hydrate and hemiacetals in aqueous solution. J Am Chem Soc 109(15):4675–4690. https://doi.org/10.1021/ja00249a034
Azofra LM, Alkorta I, Elguero J, Toro-Labbé A (2012) Mechanisms of formation of hemiacetals: intrinsic reactivity analysis. J Phys Chem A 116(31):8250–8259. https://doi.org/10.1021/jp304495f
Tsalagkas D, Zhai L, Kim HC, Kim J (2017) Optical and mechanical properties of cellulose nanopaper structures. In: Varadan VK (eds). https://doi.org/10.1117/12.2259839.
Soni R, Asoh T-A, Hsu Y-I, Uyama H (2022) Freshwater-durable and marine-degradable cellulose nanofiber reinforced starch film. Cellulose. https://doi.org/10.1007/s10570-021-04410-8
Saito T, Isogai A (2005) Ion-exchange behavior of carboxylate groups in fibrous cellulose oxidized by the TEMPO-mediated system. Carbohydr Polym 61(2):183–190. https://doi.org/10.1016/j.carbpol.2005.04.009
Royer S-J, Wiggin K, Kogler M, Deheyn DD (2021) Degradation of synthetic and wood-based cellulose fabrics in the marine environment: comparative assessment of field, aquarium, and bioreactor experiments. Sci Total Environ 791:148060. https://doi.org/10.1016/j.scitotenv.2021.148060
Acknowledgements
This work was supported by a Japan Society for the Promotion of Science (JSPS) KAKENHI Grant (No. 20H02797) and the New Energy and Industrial Technology Development Organization (NEDO) (JPNP 20004). This work was also supported by Japan Science and Technology Agency (JST) Grant Number JPMJPF2218. We would like to express our appreciation to Nihon Shokuhin Kako Co. Ltd. (Japan) for providing the starch used in the study.
Funding
Open access funding provided by Osaka University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Haji Abdul Hamid, I.D., Soni, R., Hsu, YI. et al. Effect of pH on water durability of cellulose nanofiber-reinforced starch film. J Mater Cycles Waste Manag 26, 723–730 (2024). https://doi.org/10.1007/s10163-023-01828-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10163-023-01828-z