Abstract
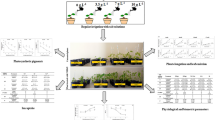
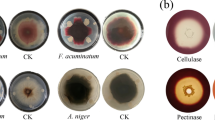
Cyamopsis tetragonoloba (L.) Taub. (guar) is a commercially important crop known for its galactomannan content in seeds. Drought stress is a significant global concern that compromises the productivity of major legumes including guar. The endophytic microbes associated with plants play a significant role in enhancing plant growth and modulating the impact of abiotic stress(s). The present study involved the isolation of 73 endophytic bacteria from the guar seeds of drought-tolerant (RGC-1002 and RGC-1066) and sensitive (Sarada and Varsha) varieties. Based on multiple PGP attributes and drought tolerance, at 50% PEG6000 w/v, 11 efficient isolates were selected and identified through 16S rRNA gene sequencing. Isolates belonging to ten different species of bacilli including Cytobacillus oceanisediminis, Mesobacillus fermenti, Peribacillus simplex from sensitive and Bacillus zanthoxyli, B. safensis, B. velezensis, B. altitudinis, B. licheniformis, B. tequilensis, and B. paralicheniformis isolated from tolerant varieties. A greenhouse experiment with a drought-sensitive guar variety demonstrated that inoculation of selected isolates showed comparatively better plant growth, higher relative water content (RWC), decreased carbon isotope discrimination ratio (Δ13C), increased chlorophyll, carotenoids, anthocyanin, and proline content, decreased malondialdehyde (MDA) and modulated defense enzymes as compared to their uninoculated controls. Tolerant variety isolates B. tequilensis NBRI14G and B. safensis NBRI10R showed the most promising results in improving plant growth and also drought stress tolerance in guar plants. This study represents for the first time that seed endophytic bacterial strains from guar can be utilized to develop the formulation for improving the productivity of guar under drought-stress conditions.






Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article.
Abbreviations
- A :
-
Net photosynthetic rate
- ACC-1:
-
Aminocyclopropane-1-carboxylate
- APX:
-
Ascorbate peroxidase
- CAT:
-
Catalase
- DW:
-
Dry weight
- E:
-
Transpiration
- EPS:
-
Exopolysaccharide
- FW:
-
Fresh weight
- GPX:
-
Guaiacol peroxidase
- g s :
-
Stomatal conductance
- IAA:
-
Indole-3-acetic acid
- IRMS:
-
Isotope ratio mass spectrometer
- MDA:
-
Malondialdehyde
- PEG:
-
Polyethylene glycol 6000
- PGPB:
-
Plant growth-promoting bacteria
- RL:
-
Root length
- RWC:
-
Relative water content
- SL:
-
Shoot length
- SOD:
-
Superoxide dismutase
- TW:
-
Turgid weight
- WUE:
-
Water use efficiency
- Δ13C, CID:
-
Carbon isotope discrimination ratio
References
Abideen Z, Cardinale M, Zulfiqar F, Koyro HW, Rasool SG, Hessini K, Darbali W, Zhao F, Siddique KH (2022) Seed endophyte bacteria enhance drought stress tolerance in Hordeum vulgare by regulating, physiological characteristics, antioxidants and minerals uptake. Fron Plant Sci 13:980046. https://doi.org/10.3389/fpls.2022.980046
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Badawy IH, Hmed AA, Sofy MR, Al-Mokadem AZ (2022) Alleviation of cadmium and nickel toxicity and phyto-stimulation of tomato plant L. by endophytic micrococcus luteus and enterobacter cloacae. Plants 11:2018. https://doi.org/10.3390/plants11152018
Bajoria S, Varshney AK, Pareek RP, Mohan MK, Ghosh P (2008) Screening and characterization of antifungal clusterbean (Cyamopsis tetragonoloba) rhizobacteria. Biocontrol Sci Technol 18:139–156. https://doi.org/10.1080/09583150701818972
Barrs HD, Weatherley PE (1962) A re-examination of the relative turgidity technique for estimating water deficits in leaves. Aust J Biol Sci 15:413–428. https://doi.org/10.1071/BI9620413
Bates LS, Waldren RA, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Bric JM, Bostock RM, Silverstone SE (1991) Rapid in situ assay for indoleacetic acid production by bacteria immobilized on a nitrocellulose membrane. Appl Environ Microbiol 57:535–538. https://doi.org/10.1128/aem.57.2.535-538.1991
Dixit VK, Misra S, Mishra SK, Tewari SK, Joshi N, Chauhan PS (2020) Characterization of plant growth-promoting alkalotolerant Alcaligenes and Bacillus strains for mitigating the alkaline stress in Zea mays. Antonie Van Leeuwenhoek 113:889–905. https://doi.org/10.1007/s10482-020-01399-1
Dowarah B, Agarwal H, Krishnatreya DB, Sharma PL, Kalita N, Agarwala N (2021) Evaluation of seed associated endophytic bacteria from tolerant chilli cv. Firingi Jolokia for their biocontrol potential against bacterial wilt disease. Microbiol Res 248:126751. https://doi.org/10.1016/j.micres.2021.126751
DuBois M, Gilles KA, Hamilton JK, Rebers PT, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356. https://doi.org/10.1021/ac60111a017
Farquhar GD, O’Leary MH, Berry JA (1982) On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Funct Plant Biol 9:121–137. https://doi.org/10.1071/PP9820121
Fonseca MDCD, Bossolani JW, de Oliveira SL, Moretti LG, Portugal JR, Scudeletti D, de Oliveira EF, Crusciol CAC (2022) Bacillus subtilis inoculation improves nutrient uptake and physiological activity in sugarcane under drought stress. Microorganisms 10:809. https://doi.org/10.3390/microorganisms10040809
Gothwal RK, Nigam VK, Mohan MK, Sasmal D, Ghosh P (2008) Screening of nitrogen fixers from rhizospheric bacterial isolates associated with important desert plants. Appl Ecol Environ Res 6:101–109. https://doi.org/10.15666/aeer/0602_101109
Hagagy N, AbdElgawad H (2023) Rapeseed plant: bio-stimulation effects of plant growth-promoting actinobacteria on metabolites and antioxidant defense system under elevated CO2 conditions. J Sci Food Agric. https://doi.org/10.1002/jsfa.12909
Haroon U, Munis MFH, Liaquat F, Khizar M, Elahi M, Chaudhary HJ (2023) Biofilm formation and flocculation potential analysis of halotolerant Bacillus tequilensis and its inoculation in soil to mitigate salinity stress of chickpea. Physiol Mol Biol Plants 29:277–288. https://doi.org/10.1007/s12298-023-01280-1
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Hemeda HM, Klein BP (1990) Effects of naturally occurring antioxidants on peroxidase activity of vegetable extracts. J Food Sci 55:184–185. https://doi.org/10.1111/j.1365-2621.1990.tb06048.x
Johnston-Monje D, Gutiérrez JP, Becerra Lopez-Lavalle LA (2022) Stochastic inoculum, biotic filtering and species-specific seed transmission shape the rare microbiome of plants. Life 12:1372. https://doi.org/10.3390/life12091372
Kumar M, Mishra S, Dixit V, Kumar M, Agarwal L, Chauhan PS, Nautiyal CS (2016) Synergistic effect of Pseudomonas putida and Bacillus amyloliquefaciens ameliorates drought stress in chickpea (Cicer arietinum L.). Plant Signal Behav 11:1071004. https://doi.org/10.1080/15592324.2015.1071004
Mahmood A, Kataoka R (2020) Metabolite profiling reveals a complex response of plants to application of plant growth-promoting endophytic bacteria. Microbiol Res 234:126421. https://doi.org/10.1016/j.micres.2020.126421
Mariotti L, Scartazza A, Curadi M, Picciarelli P, Toffanin A (2021) Azospirillum baldaniorum Sp245 induces physiological responses to alleviate the adverse effects of drought stress in purple basil. Plants 10:1141. https://doi.org/10.3390/plants10061141
Mehta S, Nautiyal CS (2001) An efficient method for qualitative screening of phosphate-solubilizing bacteria. Curr Microbiol 43:51–56. https://doi.org/10.1007/s002840010259
Mekureyaw MF, Pandey C, Hennessy RC, Nicolaisen MH, Liu F, Nybroe O, Roitsch T (2022) The cytokinin-producing plant beneficial bacterium Pseudomonas fluorescens G20–18 primes tomato (Solanum lycopersicum) for enhanced drought stress responses. J Plant Physiol 270:153629. https://doi.org/10.1016/j.jplph.2022.153629
Meyer JA, Abdallah MA (1978) The fluorescent pigment of Pseudomonas fluorescens: biosynthesis, purification and physicochemical properties. Microbiol 107:319–328. https://doi.org/10.1099/00221287-107-2-319
Misra S, Chauhan PS (2020) ACC deaminase-producing rhizosphere competent Bacillus spp. mitigate salt stress and promote Zea mays growth by modulating ethylene metabolism. 3 Biotech 10:119. https://doi.org/10.1007/s13205-020-2104-y
Misra S, Dixit VK, Khan MH, Mishra SK, Dviwedi G, Yadav S, Lehri A, Chauhan PS (2017) Exploitation of agro-climatic environment for selection of 1-aminocyclopropane-1-carboxylic acid (ACC) deaminase producing salt-tolerant indigenous plant growth promoting rhizobacteria. Microbiol Res 205:25–34. https://doi.org/10.1016/j.micres.2017.08.007
Misra S, Semwal P, Pandey DD, Mishra SK, Chauhan PS (2023) Siderophore-producing Spinacia Oleracea bacterial endophytes enhance nutrient status and vegetative growth under iron-deficit conditions. J Plant Growth Regul. https://doi.org/10.1007/s00344-023-11185-8
Murali M, Singh SB, Gowtham HG, Shilpa N, Prasad M, Aiyaz M, Amruthesh KN (2021) Induction of drought tolerance in Pennisetum glaucum by ACC deaminase producing PGPR-Bacillus amyloliquefaciens through antioxidant defense system. Microbiol Res 253:126891. https://doi.org/10.1016/j.micres.2021.126891
Murray JR, Hackett WP (1991) Dihydroflavonol reductase activity in relation to differential anthocyanin accumulation in juvenile and mature phase Hedera helix L. Plant Physiol 97:343–351. https://doi.org/10.1104/pp.97.1.343
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Narayan S, Sharma RK, Kumar V, Sanyal I, Shirke PA (2023) Alterations in plant anatomy and higher lignin synthesis provides drought tolerance in cluster bean [Cyamopsis tetragonoloba (L.) Taub.]. Plant Physiol Biochem 201:107905. https://doi.org/10.1016/j.plaphy.2023.107905
Nautiyal CS (1999) An efficient microbiological growth medium for screening phosphate solubilizing microorganisms. FEMS Microbiol Lett 170:265–270. https://doi.org/10.1111/j.1574-6968.1999.tb13383.x
Newcombe G, Marlin M, Barge E, Heitmann S, Ridout M, Busby PE (2023) Plant seeds commonly host Bacillus spp., potential antagonists of phytopathogens. Microb Ecol 85:1356–1366. https://doi.org/10.1007/s00248-022-02024-6
Paganin P, Isca C, Tasso F, Calandrelli T, Migliore G, Marras PA, Medas D, Dore E, De Giudici G, Sprocati AR, Alisi C (2023) A bacterial formula with native strains as alternative to chemical fertiliser for tomato crop. Plant Growth Regul. https://doi.org/10.1007/s10725-023-00993-3
Pandey K, Kumar RS, Prasad P, Pande V, Trivedi PK, Shirke PA (2022) Coordinated regulation of photosynthesis and sugar metabolism in guar increases tolerance to drought. Environ Exp Bot 194:104701. https://doi.org/10.1016/j.envexpbot.2021.104701
Patel P, Patil T, Maiti S, Paul D, Amaresan N (2022) Screening of osmotic stress-tolerant bacteria for plant growth promotion in wheat (Triticum aestivum L.) and brinjal (Solanum melongena L.) under drought conditions. Lett Appl Microbiol 75:1286–1292. https://doi.org/10.1111/lam.13797
Penrose DM, Glick BR (2003) Methods for isolating and characterizing ACC deaminase-containing plant growth-promoting rhizobacteria. Physiol Plant 118:10–15. https://doi.org/10.1034/j.1399-3054.2003.00086.x
Semwal P, Misra S, Misra A, Kar S, Majhi B, Mishra SK, Srivastava S, Chauhan PS (2023) Endophytic Bacillus strains enhance biomass and bioactive metabolites of Gloriosa superba. Ind Crops Prod 204:117296. https://doi.org/10.1016/j.indcrop.2023.117296
Srivastava S, Yadav A, Seem K, Mishra S, Chaudhary V, Nautiyal CS (2008) Effect of high temperature on Pseudomonas putida NBRI0987 biofilm formation and expression of stress sigma factor RpoS. Curr Microbiol 56:453–457. https://doi.org/10.1007/s00284-008-9105-0
Titus S, Gaonkar SN, Srivastava RB and Karande AA (1995) Exopolymer production by a fouling marine bacterium Pseudomonas alcaligenes. Indian J Geomarine Sci 24:45‒48. http://nopr.niscpr.res.in/handle/123456789/37241. Accessed 22 Nov 2016
Udeh EL, Ngmenzuma TY, Maseko ST, Nyila MA, Kanu SA, Mohale KC (2023) Gas exchange and carbon assimilation of Bambara groundnut landraces co-inoculated with Bradyrhizobium japonicum and Bacillus subtilis under simulated drought stress. Sci Afr 21:01823. https://doi.org/10.1016/j.sciaf.2023.e01823
Vishnupradeep R, Bruno LB, Taj Z, Karthik C, Challabathula D, Kumar A, Freitas H, Rajkumar M (2022) Plant growth promoting bacteria improve growth and phytostabilization potential of Zea mays under chromium and drought stress by altering photosynthetic and antioxidant responses. Environ Technol Innov 25:102154. https://doi.org/10.1016/j.eti.2021.102154
Wang G, Zhang L, Zhang S, Li B, Li J, Wang X, Zhang J, Guan C, Ji J (2023) The combined use of a plant growth promoting Bacillus sp. strain and GABA promotes the growth of rice under salt stress by regulating antioxidant enzyme system, enhancing photosynthesis and improving soil enzyme activities. Microbiol Res 266:127225. https://doi.org/10.1016/j.micres.2022.127225
Wellburn AR (1994) The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 144:307–313. https://doi.org/10.1016/S0176-1617(11)81192-2
Acknowledgements
Senior Research Fellowships provided to Vaishali Umrao, Sushma Yadav, and Pradeep Semwal by University Grants Commission (UGC), New Delhi, is gratefully acknowledged. Authors are thankful to the Director, CSIR-National Botanical Research Institute, Lucknow, for providing the necessary facilities. This manuscript bears CSIR-NBRI communication number, CSIR-NBRI_MS/2024/02/04.
Author information
Authors and Affiliations
Contributions
Vaishali Umrao performed the experiments, analyzed the data, and drafted the manuscript; Sushma Yadav helped in carrying out the experiments and drafting the manuscript; Pradeep Semwal contributed in the validation of experiments; Sankalp Misra helped in writing original draft and editing; Shashank Mishra helped with methodology and arranged the resources; Puneet Singh Chauhan helped in conceptualization, critical revision, and final approval of the manuscript; Pramod Arvind Shirke conceptualized, supervised the research, and revised and finalized the manuscript. All authors have read and approved the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
N/A.
Consent for publication
All authors approved the manuscript for publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Umrao, V., Yadav, S., Semwal, P. et al. Endophytic bacilli from Cyamopsis tetragonoloba (L.) Taub. induces plant growth and drought tolerance. Int Microbiol (2024). https://doi.org/10.1007/s10123-024-00499-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10123-024-00499-6