Abstract
Nanoparticles (NPs) of cobalt oxide and Fe/Ni-doped cobalt oxide have been prepared by the co-precipitation method. XPS analysis established the chemical nature of these NPs in terms of atomic as well as molecular formulations, and EDAX analysis complemented the XPS results displaying the presence of Fe and Ni atoms in the cobalt oxide NPs revealing successful doping of Fe and Ni in cobalt oxide NPs. The impact of Fe and Ni-doping on the structure and physico-chemical properties of cobalt oxide NPs was also established by XPS analysis. The presence of metal-oxide bonds was further established by FTIR (Fourier transform infrared spectroscopy) and HRTEM (high-resolution transmission electron microscopy) analysis confirmed the formation of NPs. Among bare and Fe/Ni-doped cobalt oxide NPs, Fe-doped cobalt oxide NPs showed maximum photocatalytic activity against methylene blue as well as Fe-doped cobalt oxide NPs also acted as an excellent catalyst for the sulfoxidation of sulfides with 99% conversion and 95% selectivity under green reaction conditions utilizing water as a solvent.
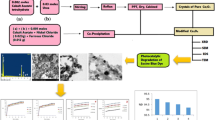
Graphical abstract













Similar content being viewed by others
Data availability
Enquiries about data availability should be directed to the authors.
References
Al-Tuwirqi R, Al-Ghamdi AA, Aal NA et al (2011) Facile synthesis and optical properties of Co3O4 nanostructures by the microwave route. Superlattices Microstruct 49:416–421. https://doi.org/10.1016/j.spmi.2010.12.010
Ashiri R (2013) Detailed FT-IR spectroscopy characterization and thermal analysis of synthesis of barium titanate nanoscale particles through a newly developed process. Vib Spectrosc 66:24–29. https://doi.org/10.1016/j.vibspec.2013.02.001
Bur SK, Padwa A (2004) The pummerer reaction: methodology and strategy for the synthesis of heterocyclic compounds. Chem Rev 104:2401–2432. https://doi.org/10.1021/cr020090l
Callegari A, Tonti D, Chergui M (2003) Photochemically grown silver nanoparticles with wavelength-controlled size and shape. Nano Lett. https://doi.org/10.1021/nl034757a
Carofiglio M, Barui S, Cauda V, Laurenti M (2020) Doped zinc oxide nanoparticles: synthesis, characterization and potential use in nanomedicine. Appl Sci Basel Switz 10:5194. https://doi.org/10.3390/app10155194
Farhadi S, Pourzare K, Sadeghinejad S (2013a) Simple preparation of ferromagnetic Co3O4 nanoparticles by thermal dissociation of the [CoII(NH3)6](NO3)2 complex at low temperature. J Nanostruct Chem 3:16. https://doi.org/10.1186/2193-8865-3-16
Farhadi S, Safabakhsh J, Zaringhadam P (2013b) Synthesis, characterization, and investigation of optical and magnetic properties of cobalt oxide (Co3O4) nanoparticles. J Nanostruct Chem 3:69. https://doi.org/10.1186/2193-8865-3-69
Feldman KS (2006) Modern pummerer-type reactions. Tetrahedron 62:5003–5034. https://doi.org/10.1016/j.tet.2006.03.004
Field L (1978) Some developments in synthetic organic sulfur chemistry since 1970. Synthesis 1978:713–740. https://doi.org/10.1055/s-1978-24870
Garnock-Jones KP, Dhillon S, Scott LJ (2009) Armodafinil. CNS Drugs 23:793–803. https://doi.org/10.2165/11203290-000000000-00000
Garza F, Kearney TE (2012) Chapter 205. Methylene blue. In: Olson KR (ed) Poisoning & drug overdose, (6th edn). The McGraw-Hill Companies, New York
Gola D, Kriti A, Bhatt N et al (2021) Silver nanoparticles for enhanced dye degradation. Curr Res Green Sustain Chem 4:100132. https://doi.org/10.1016/j.crgsc.2021.100132
Iravani S, Varma RS (2020) Sustainable synthesis of cobalt and cobalt oxide nanoparticles and their catalytic and biomedical applications. Green Chem 22:2643–2661. https://doi.org/10.1039/D0GC00885K
Kant S, Kumar S (2013) Effects of alkaline earth metal ions on thermodynamic and ultrasonic properties of ascorbic acid. J Chem Eng Data 58:1294–1300. https://doi.org/10.1021/je301362j
Kant S, Kumar A, Kumar S (2009) Molar volume, viscosity and conductance studies of some alkali metal chlorides in aqueous ascorbic acid. J Mol Liq 150:39–42. https://doi.org/10.1016/j.molliq.2009.09.010
Khan I, Saeed K, Khan I (2019) Nanoparticles: properties, applications and toxicities. Arab J Chem 12:908–931. https://doi.org/10.1016/j.arabjc.2017.05.011
Kumar S, Thakur A, Rangra VS, Sharma S (2016) Synthesis and use of low-band-gap ZnO nanoparticles for water treatment. Arab J Sci Eng 41:2393–2398. https://doi.org/10.1007/s13369-015-1852-1
Kumar S, Kumar M, Thakur A, Patial S (2017) Water treatment using photocatalytic and antimicrobial activities of tin oxide nanoparticles. IJCT Vol244 July 2017
Malwal D, Packirisamy G (2018) Chapter 10-recent advances in the synthesis of metal oxide (MO) nanostructures. In: Mohan Bhagyaraj S, Oluwafemi OS, Kalarikkal N, Thomas S (eds) Synthesis of inorganic nanomaterials. Woodhead Publishing, Sawston, pp 255–281
Medhi R, Marquez MD, Lee TR (2020) Visible-light-active doped metal oxide nanoparticles: review of their synthesis, properties, and applications. ACS Appl Nano Mater 3:6156–6185. https://doi.org/10.1021/acsanm.0c01035
Nakamoto K (2006) Infrared and Raman spectra of inorganic and coordination compounds. https://doi.org/10.1002/0470027320.s4104
Oladoye PO, Ajiboye TO, Omotola EO, Oyewola OJ (2022) Methylene blue dye: toxicity and potential elimination technology from wastewater. Results Eng 16:100678. https://doi.org/10.1016/j.rineng.2022.100678
Petitto SC, Langell MA (2004) Surface composition and structure of Co3O4(110) and the effect of impurity segregation. J Vac Sci Technol Vac Surf Films 22:1690–1696. https://doi.org/10.1116/1.1763899
Pradeev Raj K, Sadaiyandi K, Kennedy A et al (2018) Influence of Mg doping on ZnO nanoparticles for enhanced photocatalytic evaluation and antibacterial analysis. Nanoscale Res Lett 13:229. https://doi.org/10.1186/s11671-018-2643-x
Prakash Sharma V, Sharma U, Chattopadhyay M, Shukla VN (2018) Advance applications of nanomaterials: a review. Mater Today Proc 5:6376–6380. https://doi.org/10.1016/j.matpr.2017.12.248
Qiu H-J, Liu L, Mu Y-P et al (2015) Designed synthesis of cobalt-oxide-based nanomaterials for superior electrochemical energy storage devices. Nano Res 8:321–339. https://doi.org/10.1007/s12274-014-0589-6
Rajaeiyan A, Bagheri-Mohagheghi MM (2013) Comparison of sol-gel and co-precipitation methods on the structural properties and phase transformation of γ and α-Al2O3 nanoparticles. Adv Manuf 1:176–182. https://doi.org/10.1007/s40436-013-0018-1
Ray C, Pal T (2017) Retracted article: recent advances of metal–metal oxide nanocomposites and their tailored nanostructures in numerous catalytic applications. J Mater Chem A 5:9465–9487. https://doi.org/10.1039/C7TA02116J
Reddy KP, Jain R, Ghosalya MK, Gopinath CS (2017) Metallic cobalt to spinel Co 3 O 4 —electronic structure evolution by near-ambient pressure photoelectron spectroscopy. J Phys Chem C 121:21472–21481. https://doi.org/10.1021/acs.jpcc.7b06661
Rojas CM (2015) The Mislow-Evans rearrangement. Molecular rearrangements in organic synthesis. Wiley, Hoboken, pp 569–626
Santhosh C, Velmurugan V, Jacob G et al (2016) Role of nanomaterials in water treatment applications: a review. Chem Eng J 306:1116–1137. https://doi.org/10.1016/j.cej.2016.08.053
Shahabuddin S, Sarih NM, Mohamad S, Baharin SNA (2016) Synthesis and characterization of Co3O4 nanocube-doped polyaniline nanocomposites with enhanced methyl orange adsorption from aqueous solution. RSC Adv 6:43388–43400. https://doi.org/10.1039/C6RA04757B
Shindhal T, Rakholiya P, Varjani S et al (2021) A critical review on advances in the practices and perspectives for the treatment of dye industry wastewater. Bioengineered 12:70–87. https://doi.org/10.1080/21655979.2020.1863034
Simonsen LO, Brown AM, Harbak H et al (2011) Cobalt uptake and binding in human red blood cells. Blood Cells Mol Dis 46:266–276. https://doi.org/10.1016/j.bcmd.2011.02.009
Smith LHS, Coote SC, Sneddon HF, Procter DJ (2010) Beyond the pummerer reaction: recent developments in thionium ion chemistry. Angew Chem Int Ed 49:5832–5844. https://doi.org/10.1002/anie.201000517
Spencer CM, Faulds D (2000) Esomeprazole. Drugs 60:321–329. https://doi.org/10.2165/00003495-200060020-00006
Stoll A, Seebeck E (1951) Chemical investigations on alliin, the specific principle of garlic. Advances in enzymology and related areas of molecular biology. Wiley, Hoboken, pp 377–400
Thakur A, Kumar S, Pathania P et al (2017) Synthesis of rGO–ZnO composites for thermal, electrical and antibacterial studies. Surf Rev Lett 24:1750095. https://doi.org/10.1142/S0218625X17500950
Thakur A, Kumar S, Singh G, Rangra VS (2022a) Electrical, thermal and antimicrobial properties of synthesized TiO2/rGO nanocomposites. Mater Today Proc. https://doi.org/10.1016/j.matpr.2022.09.129
Thakur MS, Singh N, Sharma A et al (2022b) Metal coordinated macrocyclic complexes in different chemical transformations. Coord Chem Rev 471:214739. https://doi.org/10.1016/j.ccr.2022.214739
Trost BM (1978) .alpha.-Sulfenylated carbonyl compounds in organic synthesis. Chem Rev 78:363–382. https://doi.org/10.1021/cr60314a002
Xiang X, Zhang L, Hima H et al (2009) Co-based catalysts from Co/Fe/Al layered double hydroxides for preparation of carbon nanotubes. Appl Clay Sci 42:405–409. https://doi.org/10.1016/j.clay.2008.04.004
Zhang R, Ding H, Pu X et al (2020) Recent advances in the synthesis of sulfides, sulfoxides and sulfones via C-S bond construction from non-halide substrates. Catalysts 10:1339. https://doi.org/10.3390/catal10111339
Acknowledgements
This research was partially supported by the “Project Monitoring Unit of RUSA—Equity Initiative” of Himachal Pradesh University.
Funding
This study was funded by Rashtriya Uchchatar Shiksha Abhiyan, 1-10/2020-HPU(DS)-RUSA.
Author information
Authors and Affiliations
Contributions
AS helped in nanomaterial synthesis, data curation; MSM was involved in catalytic studies, resources; PP helped in catalytic studies; JB was involved in catalytic studies, formal analysis. DK and MK contributed to data curation, formal analysis; MST performed photocatalytic studies; SK helped in supervision, conceptualization, writing–original draft, writing–review & editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Soni, A., Maru, M.S., Patel, P. et al. Fe-doped nano-cobalt oxide green catalysts for sulfoxidation and photo degradation. Clean Techn Environ Policy (2023). https://doi.org/10.1007/s10098-023-02611-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10098-023-02611-2