Abstract
Finegoldia magna is an anaerobic gram-positive bacterium that can cause invasive human infections. Recently, a 52-year-old patient suffering from a periprosthetic joint infection (PJI) due to F. magna was treated with cefepime on hemodialysis; however, treatment failed due to relapse caused by antibiotic-resistant strains. Reports on the antimicrobial susceptibility of F. magna clinical isolates are rare. We collected 57 clinical F. magna isolates from Zurich, Switzerland, between September 2019 and July 2020 and tested their antimicrobial susceptibility to investigate the local resistance pattern. Antimicrobial susceptibility testing (AST) was evaluated for nine antibiotics (benzylpenicillin, amoxicillin/clavulanic acid, cefuroxime, cefepime, levofloxacin, rifampicin, metronidazole, doxycycline, and clindamycin) by E-test according to CLSI guidelines. All F. magna strains were susceptible to benzylpenicillin, amoxicillin/clavulanic acid, and metronidazole, while 75% to clindamycin. F. magna isolates showed MIC values lower than species-unrelated breakpoints for cefuroxime, levofloxacin, and cefepime in 93%, 56%, and 32% of the cases, respectively. MIC values for rifampicin and doxycycline were lower than locally determined ECOFFs in 98% and 72% of the cases, respectively. In summary, we recommend the use of benzylpenicillin, amoxicillin/clavulanic acid, or metronidazole without prior AST as first-line treatment option against F. magna PJI infections. If cefuroxime, cefepime, levofloxacin, rifampicin, doxycycline, or clindamycin are used, AST is mandatory.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Finegoldia magna is a gram-positive anaerobic bacterium formerly known as Peptostreptococcus magnus. It is found in the mucosa of the gastrointestinal tract, the female genito-urinary tract and as part of normal human skin [1, 2]. In addition, it can invade the human tissues and cause opportunistic infections such as periprosthetic joint infections (PJI) and soft-tissue abscesses [2,3,4]. In 2018, a patient at the Balgrist University hospital in Zurich, Switzerland, experienced a treatment failure of a F. magna hip PJI due to cefepime resistance that had not been tested in the initial phase of treatment. This case prompted us to investigate the in vitro antimicrobial susceptibility of F. magna, since data remain scarce (Supplementary Table S1). To provide an overview of the local resistance situation and to give recommendations for the treatment of infections caused by this microorganism, susceptibility of clinical F. magna isolates from Zurich, Switzerland, was assessed.
Case presentation
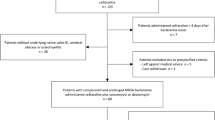
The patient was a 52-year-old woman with diabetes mellitus, chronic kidney insufficiency, and adiposity. She was initially treated for a polymicrobial PJI (Cutibacterium avidum and Citrobacter koseri) after surgical debridement of all infected tissues and explantation of the prosthesis. At time of implantation of the new prosthesis, Pseudomonas aeruginosa and F. magna were found in several intraoperative tissue biopsies. Antibiotic treatment with ciprofloxacin and cefepime was started on hemodialysis and maintained for three months without performing antimicrobial susceptibility testing (AST). Sixteen months later, a relapse with F. magna was diagnosed as the patient presented recurring pain and elevated serum inflammatory parameters. AST revealed susceptibility to amoxicillin, amoxicillin/clavulanic acid, and metronidazole, resistance to clindamycin, and MIC values for cefepime and levofloxacin above the species-unrelated breakpoints (Supplementary Table S2). Because of the relapsing infection, the joint prosthesis was explanted. Intravenous antibiotic treatment with amoxicillin was initiated for two weeks and was followed by metronidazole per os for four weeks prior to implantation of a new hip prosthesis. During the following 24 months, neither a relapse nor a new infection was detected.
Materials and methods
Bacterial isolates
All F. magna strains isolated at the Institute of Medical Microbiology (IMM) of the University of Zurich, Switzerland, between September 2019 and July 2020, were stored in home-made chopped meat carbohydrate broth at room temperature until the beginning of the study. Bacterial identification was performed by MALDI-TOF MS (Bruker Daltonics) using the direct formic acid transfer method [5]. Repeated isolates from same patients were excluded. A total of 57 isolates was analyzed.
AST and analysis of the MIC distributions
AST was performed using E-test strips (ETEST® bioMérieux SA Marcy l’Etoile, France) based on Clinical and Laboratory Standards Institute (CLSI) standard procedure [6]. Briefly, bacterial suspensions corresponding to a McFarland 1.0 were prepared in saline solution from primary sub-cultures and were spread on home-made Brucella agar plates supplemented with 5% laked sheep blood, hemin, and 10 µg/ml vitamin K1. Plates were incubated under anaerobic conditions at 37 °C for 48 h. The following nine antibiotics were tested: benzylpenicillin, amoxicillin/clavulanic acid, cefuroxime, cefepime, levofloxacin, rifampicin, metronidazole, doxycycline and clindamycin. E-tests were chosen because a high correlation between this method and the standard dilution method was shown previously [7, 8]. Internal quality control testing for the antibiotics included in the study performed during 2020–2021 at the IMM is reported in Supplementary Table S4. The median minimum inhibitory concentration (MIC50), the 90th percentile of minimum inhibitory concentration (MIC90), and the MIC range of values were determined for each antibiotic and compared to the following breakpoints. For benzylpenicillin, amoxicillin/clavulanic acid, metronidazole, and clindamycin, the breakpoints from the European Committee on Antimicrobial Susceptibility Testing (EUCAST) for gram-positive anaerobes, Version 10.0, valid from 1st January 2020, were used [9]. For cefuroxime, cefepime, and levofloxacin, EUCAST species-unrelated breakpoints were applied since EUCAST breakpoints for gram-positive anaerobes were not available [9]. For rifampicin and doxycycline, local epidemiological cut-off values (2 mg/l and 1 mg/l, respectively) were determined using “ECOFFinder” version 2.1 provided by CLSI [10].
Detection of rifampicin resistance
Whole-genome sequencing (WGS) was performed on the only clinical isolate with a MIC value for rifampicin above the local ECOFF. DNA extraction, library preparation, and WGS were performed as described recently [11]. Raw sequencing data were trimmed, filtered, and assembled by an in-house bioinformatics pipeline. The complete gene sequence of rpoB was extracted and used to query the Comprehensive Antibiotic Resistance Database (CARD) [12] in order to identify potential point mutations mediating rifampicin resistance.
Results
MIC distributions of nine antibiotics (benzylpenicillin, amoxicillin/clavulanic acid, cefuroxime, cefepime, levofloxacin, rifampicin, metronidazole, doxycycline, and clindamycin) for 57 F. magna isolates are shown in Fig1A–I, while the MIC50, MIC90, and MIC ranges are summarized in Table 1. Based on EUCAST breakpoints for gram-positive anaerobes, F. magna isolates were categorized towards penicillin, amoxicillin/clavulanic acid, clindamycin, and metronidazole as susceptible at standard dosing regimen, susceptible at increased exposure, and resistant with a high likelihood of therapeutic failure even with an increased exposure. F. magna isolates were classified as “wild-type” or “non-wild-type” when the MIC values were below or above the EUCAST non-species-related breakpoints (for cefepime, cefuroxime, and levofloxacin) or in-house determined ECOFF values (for rifampicin and doxycycline).
Distribution of the MIC values (mg/l) of nine tested antibiotics on 57 F. magna strains. Panel A, benzylpenicillin; panel B, amoxicillin/clavulanic acid; panel C, metronidazole; panel D, clindamycin; panel E, cefuroxime; panel F, cefepime; panel G, levofloxacin; panel H, doxycycline; panel I, rifampicin. Dotted line corresponds to the EUCAST breakpoints for gram-positive anaerobes, dashed line corresponds to the EUCAST species-unrelated breakpoints, and continuous line corresponds to local epidemiological cut-off values
All isolates were susceptible to benzylpenicillin, amoxicillin/clavulanic acid, and metronidazole, while 75% were susceptible to clindamycin. F. magna isolates exhibited MICs for rifampicin, doxycycline, and levofloxacin below the susceptibility breakpoint in 98, 72, and 56% of the cases, respectively. For cefuroxime and cefepime, F. magna strains exhibited MICs below the ECOFFs in 75% and only 14% of the cases, respectively. However, based on species-unrelated breakpoints, 93% and 32% of the isolates would be categorized as susceptible at increased dosing regimen.
One isolate exhibited a MIC (3 mg/l) for rifampicin above the distribution of the wild-type population (internally established ECOFF = 2 mg/l, Fig. 1 panel I). Inspection of the rpoB gene (responsible for rifampicin resistance [13]) revealed the presence of a mutation leading to the amino acid substitution Gln486Arg. Moreover, this isolate exhibited MIC values for cefepime, levofloxacin, and cefuroxime above the ECOFFs (Supplementary Table S3).
Discussion
Biofilm infections such as PJIs require surgical debridement to remove infected tissues and prolonged antibiotic treatment to prevent relapse of infection [14, 15]. Antibiotic therapy should be based on AST in order to avoid inadequate treatment. F. magna PJIs are rare and little information is available regarding the best therapeutic options. All 57 F. magna clinical isolates included in this study were susceptible to benzylpenicillin, amoxicillin/clavulanic acid, and metronidazole whereas high rates of isolates showing MICs above the resistance breakpoints for cefepime (68%), levofloxacin (44%), doxycycline (28%), and clindamycin (25%) were observed. Therefore, the latter antibiotics should be taken in consideration only after AST to prevent relapse of infection as documented in our clinical case presentation. Also, MIC values for cefuroxime and rifampicin were above the resistance breakpoints only in 7% and 2% of the cases, respectively.
In accordance with the literature, MIC data of all isolates for penicillin, amoxicillin/clavulanic acid [16,17,18,19], and metronidazole [3, 18,19,20,21,22,23] were within the susceptible range. Penicillin-resistant F. magna strains were found in different countries with rates of 1.7 to 5.7% [18, 20, 21, 24]. Metronidazole-resistant strains have been reported in only a few cases in Germany [18], Kuwait [18], and Spain [17]. As outlined above, we found a high prevalence (68%) of F. magna strains with MIC values for cefepime above the resistance ECOFF. Also, MIC values ranged between 2 and 256 mg/l; the MIC50 and MIC90 were 12 mg/l and 64 mg/l, respectively. In a comparable study, using 30 F. magna strains from diabetic foot infections, Goldstein et al. [25] reported a wide range of cefepime MICs between 1 mg/l and > 32 mg/l with high MIC50 (32 mg/l) and MIC90 (> 32 mg/l).
To our knowledge, we report for the first time AST data for cefuroxime, which is the most common antibiotic used for prophylaxis of implant-associated surgeries [26, 27]. Considering the low rate (7%) of isolates displaying MIC values above the resistance ECOFF and of 18% of isolates that would be classified as susceptible at increased exposure considering species-unrelated breakpoints, cefuroxime deserves better attention and may be discussed in current prophylaxis recommendations.
Comparative analysis with the literature should be performed with caution. While our study included 57 clinical strains isolated in Switzerland, other studies analyzed significantly smaller numbers of isolates. In addition, differences in resistance rates were observed in various countries. For instance, we found a significant lower MIC50 value (0.5 mg/l) for levofloxacin as compared to a Russian study, where a MIC50 of 32 mg/l for 38 F. magna strains isolated from cancer patients was reported [24]. High MIC values for levofloxacin have also been reported in other studies conducted by Veloo et al., Mikamo et al., or Goldstein et al. [28,29,30]. For rifampicin, we found a high MIC50 value (0.38 mg/l) as compared to the only study available in literature from Sweden, where the MIC50 of rifampicin for nine F. magna isolates was as low as 0.064 mg/l [3]. Based on our in-house ECOFF value, we found a moderate portion of strains (28%) falling out the doxycycline wild-type population. This is in stark contrast to the study of Goldstein et al., who reported a high prevalence of non-wild-type strains (85%, n = 100) from the USA, Canada, Germany, Holland, Hungary, and Sweden [19]. The resistance rate of 25% for clindamycin in Switzerland is comparable to that found in 271 F. magna isolates from Slovenia (28%) [18], while significantly lower to what reported in F. magna isolates from Kuwait (50%) [18] and Spain (54%) [17], though smaller numbers were tested.
In conclusion, F. magna clinical isolates are in general susceptible to penicillin, amoxicillin/clavulanic acid, and metronidazole and PJI infections caused by this microorganism can be treated with these antibiotics without prior AST. Treatment with cefuroxime, cefepime, levofloxacin, rifampicin, doxycycline, and clindamycin always requires confirmatory AST due to varying resistance rates.
Availability of data and material
Raw data in supplementary part.
All authors agree with the content and conclusions of this manuscript.
References
Murdoch DA, Shah HN (1999) Reclassification of Peptostreptococcus magnus (Prevot 1933) Holdeman and Moore 1972 as Finegoldia magna comb. nov. and Peptostreptococcus micros (Prevot 1933) Smith 1957 as Micromonas micros comb. nov. Anaerobe 5(5):555–559
Murphy EC, Frick IM (2013) Gram-positive anaerobic cocci–commensals and opportunistic pathogens. FEMS Microbiol Rev 37(4):520–553
Söderquist B et al (2017) Finegoldia magna isolated from orthopedic joint implant-associated infections. J Clin Microbiol 55(11):3283–3291
Walter G et al (2014) Bone and joint infections due to anaerobic bacteria: an analysis of 61 cases and review of the literature. Eur J Clin Microbiol Infect Dis 33(8):1355–1364
Schulthess B et al (2013) Identification of Gram-positive cocci by use of matrix-assisted laser desorption ionization-time of flight mass spectrometry: comparison of different preparation methods and implementation of a practical algorithm for routine diagnostics. J Clin Microbiol 51(6):1834–1840
CLSI (2007) Methods for antimicrobial susceptibility testing of anaerobic bacteria; approved standard—seventh edition (M11-A7). Seventh ed. Vol. 27. CLSI
Wust J, Hardegger U (1992) Comparison of the E test and a reference agar dilution method for susceptibility testing of anaerobic bacteria. Eur J Clin Microbiol Infect Dis 11(12):1169–1173
Citron DM et al (1991) Evaluation of the E test for susceptibility testing of anaerobic bacteria. J Clin Microbiol 29(10):2197–2203
EUCAST. The european committee on antimicrobial susceptibility testing. Breakpoint tables for interpretation of MICs and zone diameters. Version 10.0, 2020. http://www.eucast.org
Turnidge J, Kahlmeter G, Kronvall G (2006) Statistical characterisation of bacterial wild-type MIC value distributions and the determination of epidemiological cut-off values. Clin Microbiol Infect 12(5):418–425
Lauener FN, et al (2019) Genetic determinants and prediction of antibiotic resistance phenotypes in Helicobacter pylori. J Clin Med 8(1)
McArthur AG et al (2013) The comprehensive antibiotic resistance database. Antimicrob Agents Chemother 57(7):3348–3357
Alifano P et al (2015) Rifampicin-resistance, rpoB polymorphism and RNA polymerase genetic engineering. J Biotechnol 202:60–77
Bernard L et al (2021) Antibiotic therapy for 6 or 12 weeks for prosthetic joint infection. N Engl J Med 384(21):1991–2001
Kusejko K et al (2021) The impact of surgical strategy and rifampin on treatment outcome in Cutibacterium periprosthetic joint infections. Clin InFect Dis 72(12):e1064–e1073
Scapaticci M et al (2018) A case of necrotizing fasciitis caused by Finegoldia magna in a patient with type 2 diabetes mellitus. Infez Med 26(4):359–363
Cobo F et al (2019) Antimicrobial susceptibility and clinical findings of significant anaerobic bacteria in southern Spain. Anaerobe 59:49–53
Veloo ACM et al (2020) Antimicrobial susceptibility profiles of anaerobic bacteria, isolated from human clinical specimens, within different European and surrounding countries. A joint ESGAI study. Anaerobe 61:102111
Goldstein EJC, et al (2017) In vitro activities of pexiganan and 10 comparator antimicrobials against 502 anaerobic isolates recovered from skin and skin structure infections. Antimicrob Agents Chemother 61(12)
Hawser SP (2010) Activity of tigecycline and comparators against recent clinical isolates of Finegoldia magna from Europe. Eur J Clin Microbiol Infect Dis 29(8):1011–1013
Brazier J et al (2008) European surveillance study on antimicrobial susceptibility of Gram-positive anaerobic cocci. Int J Antimicrob Agents 31(4):316–320
Lee Y et al (2015) Multicenter study of antimicrobial susceptibility of anaerobic bacteria in Korea in 2012. Ann Lab Med 35(5):479–486
Petrina MAB et al (2017) Susceptibility of bacterial vaginosis (BV)-associated bacteria to secnidazole compared to metronidazole, tinidazole and clindamycin. Anaerobe 47:115–119
Shilnikova II, Dmitrieva NV (2015) Evaluation of antibiotic susceptibility of Gram-positive anaerobic cocci isolated from cancer patients of the N N Blokhin Russian cancer research center. J Pathog 2015:648134
Goldstein EJ et al (2006) In vitro activity of ceftobiprole against aerobic and anaerobic strains isolated from diabetic foot infections. Antimicrob Agents Chemother 50(11):3959–3962
AlBuhairan B, Hind D, Hutchinson A (2008) Antibiotic prophylaxis for wound infections in total joint arthroplasty: a systematic review. J Bone Joint Surg Br 90(7):915–919
Bratzler DW et al (2013) Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health Syst Pharm 70(3):195–283
Veloo AC, Welling GW, Degener JE (2011) Antimicrobial susceptibility of clinically relevant Gram-positive anaerobic cocci collected over a three-year period in the Netherlands. Antimicrob Agents Chemother 55(3):1199–1203
Mikamo H et al (2003) In vitro and in vivo antibacterial activities of telithromycin. Chemotherapy 49(1–2):62–65
Goldstein EJ et al (2006) In vitro activities of dalbavancin and 12 other agents against 329 aerobic and anaerobic gram-positive isolates recovered from diabetic foot infections. Antimicrob Agents Chemother 50(8):2875–2879
Acknowledgements
We thank the technicians of the Institute of Medical Microbiology of the University of Zurich for their technical assistance and collection of the clinical samples.
Funding
Open access funding provided by University of Zurich. The Balgrist PJI cohort is financially supported by the Balgrist foundation, Switzerland.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
The Cantonal Ethic Authority of Zurich confirmed that there is no ethical application required for the AST because the samples had been collected and anonymized before the beginning of the study. For the case description, consent was obtained as part of the following cohort study which was approved on November 2nd, 2017, by the Cantonal Ethic Authority of Zurich, Switzerland: “Prospective observational cohort study on periprosthetic joint infections (PJI): Incidence, clinical manifestations, diagnostic approaches, treatment strategies, and long-term outcome” (BASEC Number 2017–01458).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Patients signed informed consent regarding publishing their data.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Walser, F., Prinz, J., Rahm, S. et al. Antimicrobial susceptibility testing is crucial when treating Finegoldia magna infections. Eur J Clin Microbiol Infect Dis (2022). https://doi.org/10.1007/s10096-022-04439-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10096-022-04439-y