Abstract
Background
Juvenile myoclonic epilepsy (JME) is characterized by altered patterns of brain functional connectivity (FC). However, the nature and extent of alterations in the spatiotemporal characteristics of dynamic FC in JME patients remain elusive. Dynamic networks effectively encapsulate temporal variations in brain imaging data, offering insights into brain network abnormalities and contributing to our understanding of the seizure mechanisms and origins.
Methods
Resting-state functional magnetic resonance imaging (rs-fMRI) data were procured from 37 JME patients and 37 healthy counterparts. Forty-seven network nodes were identified by group-independent component analysis (ICA) to construct the dynamic network. Ultimately, patients’ and controls’ spatiotemporal characteristics, encompassing temporal clustering and variability, were contrasted at the whole-brain, large-scale network, and regional levels.
Results
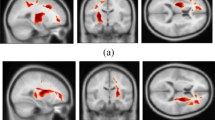
Our findings reveal a marked reduction in temporal clustering and an elevation in temporal variability in JME patients at the whole-brain echelon. Perturbations were notably pronounced in the default mode network (DMN) and visual network (VN) at the large-scale level. Nodes exhibiting anomalous were predominantly situated within the DMN and VN. Additionally, there was a significant correlation between the severity of JME symptoms and the temporal clustering of the VN.
Conclusions
Our findings suggest that excessive temporal changes in brain FC may affect the temporal structure of dynamic brain networks, leading to disturbances in brain function in patients with JME. The DMN and VN play an important role in the dynamics of brain networks in patients, and their abnormal spatiotemporal properties may underlie abnormal brain function in patients with JME in the early stages of the disease.







Similar content being viewed by others
Data availability
The datasets analyzed in the current study are available from the corresponding author upon reasonable request.
References
Zhang Z, Liu G, Zheng W, Shi J, Liu H, Sun Y (2020) Altered dynamic effective connectivity of the default mode network in newly diagnosed drug-naive juvenile myoclonic epilepsy. Neuroimage Clin 28:102431. https://doi.org/10.1016/j.nicl.2020.102431
Appleton R, Beirne M, Acomb B (2000) Photosensitivity in juvenile myoclonic epilepsy. Seizure 9:108–111. https://doi.org/10.1053/seiz.1999.0376
Spencer SS (2002) Neural networks in human epilepsy: evidence of and implications for treatment. Epilepsia 43:219–227. https://doi.org/10.1046/j.1528-1157.2002.26901.x
Sporns O (2011) The human connectome: a complex network. Ann N Y Acad Sci 1224:109–125. https://doi.org/10.1111/j.1749-6632.2010.05888.x
Bassett DS, Bullmore ET (2009) Human brain networks in health and disease. Curr Opin Neurol 22:340. https://doi.org/10.1097/WCO.0b013e32832d93dd
van Diessen E, Diederen SJ, Braun KP, Jansen FE, Stam CJ (2013) Functional and structural brain networks in epilepsy: what have we learned? Epilepsia 54:1855–1865. https://doi.org/10.1111/epi.12350
Zhang Z, Xu Q, Liao W, Wang Z, Li Q, Yang F et al (2015) Pathological uncoupling between amplitude and connectivity of brain fluctuations in epilepsy. Hum Brain Mapp 36:2756–2766. https://doi.org/10.1002/hbm.22805
Preti MG, Bolton TA, Van De Ville D (2017) The dynamic functional connectome: state-of-the-art and perspectives. Neuroimage 160:41–54. https://doi.org/10.1016/j.neuroimage.2016.12.061
Holme P, Saramäki J (2012) Temporal networks. Phys Rep 519:97–125. https://doi.org/10.1016/j.physrep.2012.03.001
Zou H, Yang J (2021) Temporal variability-based functional brain lateralization study in ADHD. J Atten Disord 25:839–847. https://doi.org/10.1177/1087054719859074
Long Y, Cao H, Yan C, Chen X, Li L, Castellanos FX et al (2020) Altered resting-state dynamic functional brain networks in major depressive disorder: findings from the REST-meta-MDD consortium. Neuroimage Clin 26:102163. https://doi.org/10.1016/j.nicl.2020.102163
Scheffer IE, Berkovic S, Capovilla G, Connolly MB, French J, Guilhoto L et al (2017) ILAE classification of the epilepsies: position paper of the ILAE commission for classification and terminology. Epilepsia 58:512–521. https://doi.org/10.1111/epi.13709
O’donoghue M, Duncan J, Sander J (1996) The national hospital seizure severity scale: a further development of the chalfont seizure severity scale. Epilepsia 37:563–571. https://doi.org/10.1111/j.1528-1157.1996.tb00610.x
Bell AJ, Sejnowski TJ (1995) An information-maximization approach to blind separation and blind deconvolution. Neural Comput 7:1129–1159. https://doi.org/10.1162/neco.1995.7.6.1129
Allen EA, Damaraju E, Plis SM, Erhardt EB, Eichele T, Calhoun VD (2014) Tracking whole-brain connectivity dynamics in the resting state. Cereb Cortex 24:663–676. https://doi.org/10.1093/cercor/bhs352
Shirer WR, Ryali S, Rykhlevskaia E, Menon V, Greicius MD (2012) Decoding subject-driven cognitive states with whole-brain connectivity patterns. Cereb Cortex 22:158–165. https://doi.org/10.1093/cercor/bhr099
Luo L, Li Q, You W, Wang Y, Tang W, Li B et al (2021) Altered brain functional network dynamics in obsessive-compulsive disorder. Hum Brain Mapp 42:2061–2076. https://doi.org/10.1002/hbm.25345
Allen EA, Erhardt EB, Damaraju E, Gruner W, Segall JM, Silva RF et al (2011) A baseline for the multivariate comparison of resting-state networks. Front Syst Neurosci 5:2. https://doi.org/10.3389/fnsys.2011.00002
Stanley ML, Moussa MN, Paolini BM, Lyday RG, Burdette JH, Laurienti PJ (2013) Defining nodes in complex brain networks. Front Comput Neurosci 7:169. https://doi.org/10.3389/fncom.2013.00169
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N et al (2002) Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 15:273–289. https://doi.org/10.1006/nimg.2001.0978
Smith SM, Miller KL, Salimi-Khorshidi G, Webster M, Beckmann CF, Nichols TE et al (2011) Network modelling methods for FMRI. Neuroimage 54:875–891. https://doi.org/10.1016/j.neuroimage.2010.08.063
Sizemore AE, Bassett DS (2018) Dynamic graph metrics: tutorial, toolbox, and tale. Neuroimage 180:417–427. https://doi.org/10.1016/j.neuroimage.2017.06.081
Tewarie P, Hillebrand A, Schoonheim MM, van Dijk BW, Geurts JJ, Barkhof F et al (2014) Functional brain network analysis using minimum spanning trees in multiple sclerosis: an MEG source-space study. Neuroimage 88:308–318. https://doi.org/10.1016/j.neuroimage.2013.10.022
Wang X, Cui X, Ding C, Li D, Cheng C, Wang B et al (2021) Deficit of cross-frequency integration in mild cognitive impairment and Alzheimer’s disease: a multilayer network approach. J Magn Reson Imaging 53:1387–1398. https://doi.org/10.1002/jmri.27453
Dong D, Duan M, Wang Y, Zhang X, Jia X, Li Y et al (2019) Reconfiguration of dynamic functional connectivity in sensory and perceptual system in schizophrenia. Cereb Cortex 29:3577–3589. https://doi.org/10.1093/cercor/bhy232
Hophing L, Kyriakopoulos P, Bui E (2022) Sex and gender differences in epilepsy. Int Rev Neurobiol 164:235–276. https://doi.org/10.1016/bs.irn.2022.06.012
Chen Y, Chen J, Chen X, Wang R, Zeng J, Wang F et al (2020) Predictors of outcome in juvenile myoclonic epilepsy. Risk Manag Healthcare Policy 13:609–613. https://doi.org/10.2147/RMHP.S244725
Power JD, Schlaggar BL, Petersen SE (2015) Recent progress and outstanding issues in motion correction in resting state fMRI. Neuroimage 105:536–551. https://doi.org/10.1016/j.neuroimage.2014.10.044
Wang Y, Berglund IS, Uppman M, Li TQ (2019) Juvenile myoclonic epilepsy has hyper dynamic functional connectivity in the dorsolateral frontal cortex. Neuroimage Clin 21:101604. https://doi.org/10.1016/j.nicl.2018.11.014
Shine JM, van den Brink RL, Hernaus D, Nieuwenhuis S, Poldrack RA (2018) Catecholaminergic manipulation alters dynamic network topology across cognitive states. Network Neuroscience 2:381–396. https://doi.org/10.1162/netn_a_00042
Dong L, Luo C, Zhu Y, Hou C, Jiang S, Wang P et al (2016) Complex discharge-affecting networks in juvenile myoclonic epilepsy: a simultaneous EEG-fMRI study. Hum Brain Mapp 37:3515–3529. https://doi.org/10.1002/hbm.23256
Lee HJ, Park KM (2019) Structural and functional connectivity in newly diagnosed juvenile myoclonic epilepsy. Acta Neurol Scand 139:469–475. https://doi.org/10.1111/ane.13079
Zhong C, Liu R, Luo C, Jiang S, Dong L, Peng R et al (2018) Altered structural and functional connectivity of juvenile myoclonic epilepsy: an fMRI study. Neural Plasticity 2018:1. https://doi.org/10.1155/2018/7392187
Buckner RL, DiNicola LM (2019) The brain’s default network: updated anatomy, physiology and evolving insights. Nat Rev Neurosci 20:593–608. https://doi.org/10.1038/s41583-019-0212-7
Liu F, Wang Y, Li M, Wang W, Li R, Zhang Z et al (2017) Dynamic functional network connectivity in idiopathic generalized epilepsy with generalized tonic–clonic seizure. Hum Brain Mapp 38:957–973. https://doi.org/10.1002/hbm.23430
Jia X, Ma S, Jiang S, Sun H, Dong D, Chang X et al (2018) Disrupted coupling between the spontaneous fluctuation and functional connectivity in idiopathic generalized epilepsy. Front Neurol 9:838. https://doi.org/10.3389/fneur.2018.00838
Wang H, Wang B, Cen Z, Wang J, Zang Y-F, Yang D et al (2022) Aberrant visual-related networks in familial cortical myoclonic tremor with epilepsy. Parkinsonism Relat Disord 101:105–110. https://doi.org/10.1016/j.parkreldis.2022.07.001
Ke M, Wang C, Liu G (2023) Multilayer brain network modeling and dynamic analysis of juvenile myoclonic epilepsy. Front Behav Neurosci 17:1123534. https://doi.org/10.3389/fnbeh.2023.1123534
Cavanna AE, Trimble MR (2006) The precuneus: a review of its functional anatomy and behavioural correlates. Brain 129:564–583. https://doi.org/10.1093/brain/awl004
Qin Y, Li S, Yao D, Luo C (2022) Causality analysis to the abnormal subcortical-cortical connections in idiopathic generalized epilepsy. Front Neurosci 16:1003. https://doi.org/10.3389/fnins.2022.925968
Andrews-Hanna JR, Smallwood J, Spreng RN (2014) The default network and self-generated thought: component processes, dynamic control, and clinical relevance. Ann N Y Acad Sci 1316:29–52. https://doi.org/10.1111/nyas.12360
Klugah-Brown B, Luo C, He H, Jiang S, Armah GK, Wu Y et al (2019) Altered dynamic functional network connectivity in frontal lobe epilepsy. Brain Topogr 32:394–404. https://doi.org/10.1007/s10548-018-0678-z
Chilosi AM, Brovedani P, Moscatelli M, Bonanni P, Guerrini R (2006) Neuropsychological findings in idiopathic occipital lobe epilepsies. Epilepsia 47(Suppl 2):76–78. https://doi.org/10.1111/j.1528-1167.2006.00696.x
Park KM, Kim SE, Lee BI, Hur YJ (2018) Brain morphology in patients with genetic generalized epilepsy: its heterogeneity among subsyndromes. Eur Neurol 80:236–244. https://doi.org/10.1159/000496698
Wolf P, Goosses R (1986) Relation of photosensitivity to epileptic syndromes. J Neurol Neurosurg Psychiatry 49:1386–1391. https://doi.org/10.1136/jnnp.49.12.1386
Binder JR, Desai RH, Graves WW, Conant LL (2009) Where is the semantic system? A critical review and meta-analysis of 120 functional neuroimaging studies. Cereb Cortex 19:2767–2796. https://doi.org/10.1093/cercor/bhp055
Benbadis SR, Gerson WA, Harvey JH, Luders HO (1996) Photosensitive temporal lobe epilepsy. Neurology 46:1540–1542. https://doi.org/10.1212/wnl.46.6.1540
Winston GP, Daga P, Stretton J, Modat M, Symms MR, McEvoy AW et al (2012) Optic radiation tractography and vision in anterior temporal lobe resection. Ann Neurol 71:334–341. https://doi.org/10.1002/ana.22619
Besseling RM, Jansen JF, Overvliet GM, van der Kruijs SJ, Vles JS, Ebus SC et al (2013) Reduced functional integration of the sensorimotor and language network in rolandic epilepsy. Neuroimage Clin 2:239–246. https://doi.org/10.1016/j.nicl.2013.01.004
Bzdok D, Hartwigsen G, Reid A, Laird AR, Fox PT, Eickhoff SB (2016) Left inferior parietal lobe engagement in social cognition and language. Neurosci Biobehav Rev 68:319–334. https://doi.org/10.1016/j.neubiorev.2016.02.024
McGill ML, Devinsky O, Kelly C, Milham M, Castellanos FX, Quinn BT et al (2012) Default mode network abnormalities in idiopathic generalized epilepsy. Epilepsy Behav 23:353–359. https://doi.org/10.1016/j.yebeh.2012.01.013
Xu Y (2018) The posterior parietal cortex in adaptive visual processing. Trends Neurosci 41:806–822. https://doi.org/10.1016/j.tins.2018.07.012
Blakemore S-J (2012) Development of the social brain in adolescence. J R Soc Med 105:111–116. https://doi.org/10.1258/jrsm.2011.110221
Silbereis JC, Pochareddy S, Zhu Y, Li M, Sestan N (2016) The cellular and molecular landscapes of the developing human central nervous system. Neuron 89:248–268. https://doi.org/10.1016/j.neuron.2015.12.008
Supekar K, Uddin LQ, Prater K, Amin H, Greicius MD, Menon V (2010) Development of functional and structural connectivity within the default mode network in young children. Neuroimage 52:290–301. https://doi.org/10.1016/j.neuroimage.2010.04.009
Dadario NB, Sughrue ME (2023) The functional role of the precuneus. Brain 146:3598–3607. https://doi.org/10.1093/brain/awad181
Silva-Barrat C, Menini C, Bryere P, Naquet R (1986) Multiunitary activity naalysis of cortical and subcortical structures in paroxysmal discharges and grand mal seizures in photosensitve baboons. Electroencephalogr Clin Neurophysiol 64:455–468. https://doi.org/10.1016/0013-4694(86)90079-9
Bartolini E, Pesaresi I, Fabbri S, Cecchi P, Giorgi FS, Sartucci F et al (2014) Abnormal response to photic stimulation in juvenile myoclonic epilepsy: an EEG-fMRI study. Epilepsia 55:1038–1047. https://doi.org/10.1111/epi.12634
Wolf P (2017) Reflex epileptic mechanisms in humans: lessons about natural ictogenesis. Epilepsy Behav 71:118–123. https://doi.org/10.1016/j.yebeh.2015.01.009
Liston C, Chen AC, Zebley BD, Drysdale AT, Gordon R, Leuchter B et al (2014) Default mode network mechanisms of transcranial magnetic stimulation in depression. Biol Psychiat 76:517–526. https://doi.org/10.1016/j.biopsych.2014.01.023
Kellogg M, Meador KJ (2017) Neurodevelopmental effects of antiepileptic drugs. Neurochem Res 42:2065–2070. https://doi.org/10.1007/s11064-017-2262-4
Funding
This work was supported by a grant from the National Natural Science Foundation of China (grant numbers 61966023 and 82160326) and the Key Research and Development (R&D) Program Project of Gansu (grant numbers 22YF7FA089).
Author information
Authors and Affiliations
Contributions
MK, XL, and GL designed the experiment and revised the manuscript. MK and XL wrote the manuscript. GL recorded and collected the data. XL performed the data analysis. JZ and XR designed computer programs. YG designed computer programs and revised the manuscript. All authors contributed to this article and approved the version submitted.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Medical Research Ethics Committee of the Lanzhou University Second Hospital (No. 2019A-102). All individuals understood the purpose and latent risks and signed informed consent.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ke, M., Luo, X., Guo, Y. et al. Alterations in spatiotemporal characteristics of dynamic networks in juvenile myoclonic epilepsy. Neurol Sci (2024). https://doi.org/10.1007/s10072-024-07506-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10072-024-07506-8