Abstract
Background
The immune response to acute ischemic stroke (AIS) is implicated in diagnosis, prognosis, and intervention; however, the temporal dynamics of leukocytes following AIS are poorly understood. The purpose of this study was to characterize peripheral leukocyte dynamics following AIS among individuals with poor and favorable outcomes.
Methods
A retrospective chart review was conducted among patients with a diagnosis of AIS who were treated at a comprehensive stroke center across a 3-year timeframe. Groups were defined according to stroke outcomes. Patients with poor outcomes were distinguished from those with favorable outcomes by discharge National Institute of Health Stroke Score, infarct size, and Modified Rankin Scale. Leukocyte counts were compared among controls and AIS outcome groups.
Results
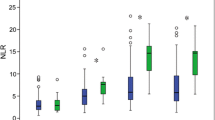
The neutrophil-lymphocyte ratio (NLR) calculated at 48–72 h post-AIS was identified as the strongest predictor of outcome. NLR was significantly higher in the poor outcome group (8.68 ± 0.93) compared with both the favorable outcome (4.5 ± 0.51, p = 0.009) and control group (4.33 ± 0.66, p < 0.001). Patients with a 48–72 h NLR ≥ 4.58 were 5.58 times more likely to have a poor outcome than AIS patients with an NLR < 4.58.
Conclusions
The results of this study improve the understanding of the immune response in AIS. Low neutrophil count relative to high lymphocyte count at 48–72 h post-AIS should be considered a predictor of a favorable stroke outcome. Conversely, high neutrophil count relative to low lymphocyte count at 48–72 h post-AIS should be considered a predictor of a poor stroke outcome.




Similar content being viewed by others
References
Rayasam A, Hsu M, Kijak JA, Kissel L, Hernandez G, Sandor M, Fabry Z (2018) Immune responses in stroke: how the immune system contributes to damage and healing after stroke and how this knowledge could be translated to better cures? Immunology. 154:363–376. https://doi.org/10.1111/imm.12918
Zhang RL, Chopp M, Jiang N, Tang WX, Prostak J, Manning AM, Anderson DC (1995) Anti-intercellular adhesion molecule-1 antibody reduces ischemic cell damage after transient but not permanent middle cerebral artery occlusion in the Wistar rat. Stroke 26:1438–1442 discussion 1443
Zhang L, Zhang ZG, Zhang RL, Lu M, Krams M, Chopp M (2003) Effects of a selective CD11b/CD18 antagonist and recombinant human tissue plasminogen activator treatment alone and in combination in a rat embolic model of stroke. Stroke 34:1790–1795. https://doi.org/10.1161/01.STR.0000077016.55891.2E
Frieler RA, Chung Y, Ahlers CG, Gheordunescu G, Song J, Vigil TM, Shah YM, Mortensen RM (2017) Genetic neutrophil deficiency ameliorates cerebral ischemia-reperfusion injury. Exp Neurol 298:104–111. https://doi.org/10.1016/j.expneurol.2017.08.016
Kong LL, Wang ZY, Han N, Zhuang X, Wang Z, Li H, Chen N (2014) Neutralization of chemokine-like factor 1, a novel C-C chemokine, protects against focal cerebral ischemia by inhibiting neutrophil infiltration via MAPK pathways in rats. J Neuroinflammation 11:112. https://doi.org/10.1186/1742-2094-11-112
Stowe AM, Adair-Kirk TL, Gonzales ER, Perez RS, Shah AR, Park TS, Gidday JM (2009) Neutrophil elastase and neurovascular injury following focal stroke and reperfusion. Neurobiol Dis 35:82–90. https://doi.org/10.1016/j.nbd.2009.04.006
Shimakura A, Kamanaka Y, Ikeda Y, Kondo K, Suzuki Y, Umemura K (2000) Neutrophil elastase inhibition reduces cerebral ischemic damage in the middle cerebral artery occlusion. Brain Res 858:55–60
Jiang N, Moyle M, Soule HR, Rote WE, Chopp M (1995) Neutrophil inhibitory factor is neuroprotective after focal ischemia in rats. Ann Neurol 38:935–942. https://doi.org/10.1002/ana.410380615
Matsuo Y, Onodera H, Shiga Y, Shozuhara H, Ninomiya M, Kihara T, Tamatani T, Miyasaka M, Kogure K (1994) Role of cell adhesion molecules in brain injury after transient middle cerebral artery occlusion in the rat. Brain Res 656:344–352
Buck BH, Liebeskind DS, Saver JL, Bang OY, Yun SW, Starkman S, Ali LK, Kim D, Villablanca JP, Salamon N, Razinia T, Ovbiagele B (2008) Early neutrophilia is associated with volume of ischemic tissue in acute stroke. Stroke 39:355–360. https://doi.org/10.1161/STROKEAHA.107.490128
Fang Y-N, Tong M-S, Sung P-H, Chen YL, Chen CH, Tsai NW, Huang CJ, Chang YT, Chen SF, Chang WN, Lu CH, Yip HK (2017) Higher neutrophil counts and neutrophil-to-lymphocyte ratio predict prognostic outcomes in patients after non-atrial fibrillation-caused ischemic stroke. Biom J 40:154–162. https://doi.org/10.1016/j.bj.2017.03.002
Maestrini I, Strbian D, Gautier S, Haapaniemi E, Moulin S, Sairanen T, Dequatre-Ponchelle N, Sibolt G, Cordonnier C, Melkas S, Leys D, Tatlisumak T, Bordet R (2015) Higher neutrophil counts before thrombolysis for cerebral ischemia predict worse outcomes. Neurology 85:1408–1416. https://doi.org/10.1212/WNL.0000000000002029
Shi J, Peng H, You S, Liu Y, Xu J, Xu Y, Liu H, Shi R, Cao Y, Liu CF (2018) Increase in neutrophils after recombinant tissue plasminogen activator thrombolysis predicts poor functional outcome of ischaemic stroke: a longitudinal study. Eur J Neurol 25:687–e45. https://doi.org/10.1111/ene.13575
Kumar AD, Boehme AK, Siegler JE, Gillette M, Albright KC, Martin-Schild S (2013) Leukocytosis in patients with neurologic deterioration after acute ischemic stroke is associated with poor outcomes. J Stroke Cerebrovasc Dis Off J Natl Stroke Assoc 22:e111–e117. https://doi.org/10.1016/j.jstrokecerebrovasdis.2012.08.008
Petrone AB, O’Connell GC, Regier MD, Chantler PD, Simpkins JW, Barr TL (2016) The role of arginase 1 in post-stroke immunosuppression and ischemic stroke severity. Transl Stroke Res 7:103–110. https://doi.org/10.1007/s12975-015-0431-9
Nayak AR, Kashyap RS, Kabra D, Deoras P, Purohit HJ, Taori GM, Daginawala HF (2011) Evaluation of routinely performed hematological and biochemical parameters for the prognosis of acute ischemic stroke patients. Neurol Sci Off J Ital Neurol Soc Ital Soc Clin Neurophysiol 32:855–860. https://doi.org/10.1007/s10072-011-0718-x
Veltkamp R, Gill D (2016) Clinical trials of immunomodulation in ischemic stroke. Neurother J Am Soc Exp Neurother 13:791–800. https://doi.org/10.1007/s13311-016-0458-y
Haeusler KG, Schmidt WUH, Foehring F, Meisel C, Guenther C, Brunecker P, Kunze C, Helms T, Dirnagl U, Volk HD, Villringer A (2012) Immune responses after acute ischemic stroke or myocardial infarction. Int J Cardiol 155:372–377. https://doi.org/10.1016/j.ijcard.2010.10.053
Haeusler KG, Schmidt WUH, Föhring F, Meisel C, Helms T, Jungehulsing GJ, Nolte CH, Schmolke K, Wegner B, Meisel A, Dirnagl U, Villringer A, Volk HD (2008) Cellular immunodepression preceding infectious complications after acute ischemic stroke in humans. Cerebrovasc Dis Basel Switz 25:50–58. https://doi.org/10.1159/000111499
Hug A, Dalpke A, Wieczorek N, Giese T, Lorenz A, Auffarth G, Liesz A, Veltkamp R (2009) Infarct volume is a major determiner of post-stroke immune cell function and susceptibility to infection. Stroke 40:3226–3232. https://doi.org/10.1161/STROKEAHA.109.557967
Vogelgesang A, Grunwald U, Langner S et al (2008) Analysis of lymphocyte subsets in patients with stroke and their influence on infection after stroke. Stroke 39:237–241. https://doi.org/10.1161/STROKEAHA.107.493635
Brooks SD, Spears C, Cummings C, VanGilder RL, Stinehart KR, Gutmann L, Domico J, Culp S, Carpenter J, Rai A, Barr TL (2014) Admission neutrophil-lymphocyte ratio predicts 90 day outcome after endovascular stroke therapy. J Neurointerventional Surg 6:578–583. https://doi.org/10.1136/neurintsurg-2013-010780
Gokhan S, Ozhasenekler A, Mansur Durgun H et al (2013) Neutrophil lymphocyte ratios in stroke subtypes and transient ischemic attack. Eur Rev Med Pharmacol Sci 17:653–657
Qun S, Tang Y, Sun J, Liu Z, Wu J, Zhang J, Guo J, Xu Z, Zhang D, Chen Z, Hu F, Xu X, Ge W (2017) Neutrophil-to-lymphocyte ratio predicts 3-month outcome of acute ischemic stroke. Neurotox Res 31:444–452. https://doi.org/10.1007/s12640-017-9707-z
Tokgoz S, Kayrak M, Akpinar Z, Seyithanoğlu A, Güney F, Yürüten B (2013) Neutrophil lymphocyte ratio as a predictor of stroke. J Stroke Cerebrovasc Dis Off J Natl Stroke Assoc 22:1169–1174. https://doi.org/10.1016/j.jstrokecerebrovasdis.2013.01.011
Tokgoz S, Keskin S, Kayrak M, Seyithanoglu A, Ogmegul A (2014) Is neutrophil/lymphocyte ratio predict to short-term mortality in acute cerebral infarct independently from infarct volume? J Stroke Cerebrovasc Dis Off J Natl Stroke Assoc 23:2163–2168. https://doi.org/10.1016/j.jstrokecerebrovasdis.2014.04.007
Yu S, Arima H, Bertmar C, Clarke S, Herkes G, Krause M (2018) Neutrophil to lymphocyte ratio and early clinical outcomes in patients with acute ischemic stroke. J Neurol Sci 387:115–118. https://doi.org/10.1016/j.jns.2018.02.002
Zhao L, Dai Q, Chen X, Li S, Shi R, Yu S, Yang F, Xiong Y, Zhang R (2016) Neutrophil-to-lymphocyte ratio predicts length of stay and acute hospital cost in patients with acute ischemic stroke. J Stroke Cerebrovasc Dis Off J Natl Stroke Assoc 25:739–744. https://doi.org/10.1016/j.jstrokecerebrovasdis.2015.11.012
Kocaturk O, Besli F, Gungoren F, Kocaturk M, Tanriverdi Z (2019) The relationship among neutrophil to lymphocyte ratio, stroke territory, and 3-month mortality in patients with acute ischemic stroke. Neurol Sci Off J Ital Neurol Soc Ital Soc Clin Neurophysiol 40:139–146. https://doi.org/10.1007/s10072-018-3604-y
Ferrarese C, Longoni M, Stanzani L (2001) Cytokine network in acute stroke. Neurol Sci Off J Ital Neurol Soc Ital Soc Clin Neurophysiol 22:287–288
Iadecola C, Anrather J (2011) The immunology of stroke: from mechanisms to translation. Nat Med 17:796–808. https://doi.org/10.1038/nm.2399
Khoshnam SE, Winlow W, Farzaneh M, Farbood Y, Moghaddam HF (2017) Pathogenic mechanisms following ischemic stroke. Neurol Sci Off J Ital Neurol Soc Ital Soc Clin Neurophysiol 38:1167–1186. https://doi.org/10.1007/s10072-017-2938-1
Gelderblom M, Leypoldt F, Steinbach K, Behrens D, Choe CU, Siler DA, Arumugam TV, Orthey E, Gerloff C, Tolosa E, Magnus T (2009) Temporal and spatial dynamics of cerebral immune cell accumulation in stroke. Stroke 40:1849–1857. https://doi.org/10.1161/STROKEAHA.108.534503
Pagram H, Bivard A, Lincz LF, Levi C (2016) Peripheral immune cell counts and advanced imaging as biomarkers of stroke outcome. Cerebrovasc Dis Extra 6:120–128. https://doi.org/10.1159/000450620
Pillay J, Ramakers BP, Kamp VM, Loi ALT, Lam SW, Hietbrink F, Leenen LP, Tool AT, Pickkers P, Koenderman L (2010) Functional heterogeneity and differential priming of circulating neutrophils in human experimental endotoxemia. J Leukoc Biol 88:211–220. https://doi.org/10.1189/jlb.1209793
Chu HX, Kim HA, Lee S, Moore JP, Chan CT, Vinh A, Gelderblom M, Arumugam TV, Broughton BRS, Drummond GR, Sobey CG (2014) Immune cell infiltration in malignant middle cerebral artery infarction: comparison with transient cerebral ischemia. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 34:450–459. https://doi.org/10.1038/jcbfm.2013.217
Perini F, Morra M, Alecci M et al (2001) Temporal profile of serum anti-inflammatory and pro-inflammatory interleukins in acute ischemic stroke patients. Neurol Sci Off J Ital Neurol Soc Ital Soc Clin Neurophysiol 22:289–296
Ullrich N, Strecker J-K, Minnerup J, Schilling M (2014) The temporo-spatial localization of polymorphonuclear cells related to the neurovascular unit after transient focal cerebral ischemia. Brain Res 1586:184–192. https://doi.org/10.1016/j.brainres.2014.08.037
Ortolano F, Maffia P, Dever G, Rodolico G, Millington OR, de Simoni MG, Brewer JM, Bushell TJ, Garside P, Carswell HV (2010) Advances in imaging of new targets for pharmacological intervention in stroke: real-time tracking of T-cells in the ischaemic brain. Br J Pharmacol 159:808–811. https://doi.org/10.1111/j.1476-5381.2009.00527.x
Bank U, Reinhold D, Schneemilch C et al (1999) Selective proteolytic cleavage of IL-2 receptor and IL-6 receptor ligand binding chains by neutrophil-derived serine proteases at foci of inflammation. J Interferon Cytokine Res Off J Int Soc Interferon Cytokine Res 19:1277–1287. https://doi.org/10.1089/107999099312957
Munder M, Schneider H, Luckner C, Giese T, Langhans CD, Fuentes JM, Kropf P, Mueller I, Kolb A, Modolell M, Ho AD (2006) Suppression of T-cell functions by human granulocyte arginase. Blood 108:1627–1634. https://doi.org/10.1182/blood-2006-11-010389
Gill D, Veltkamp R (2016) Dynamics of T cell responses after stroke. Curr Opin Pharmacol 26:26–32. https://doi.org/10.1016/j.coph.2015.09.009
Gabrilovich DI, Nagaraj S (2009) Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol 9:162–174. https://doi.org/10.1038/nri2506
Pillay J, Tak T, Kamp VM, Koenderman L (2013) Immune suppression by neutrophils and granulocytic myeloid-derived suppressor cells: similarities and differences. Cell Mol Life Sci CMLS 70:3813–3827. https://doi.org/10.1007/s00018-013-1286-4
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics statement
Research Involving Human Participants: All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee, and this study received approval from the WVU Institutional Review Board.
Informed consent
This study was a retrospective chart review, thus did not require informed consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(JPG 15 kb)
Rights and permissions
About this article
Cite this article
Petrone, A.B., Eisenman, R.D., Steele, K.N. et al. Temporal dynamics of peripheral neutrophil and lymphocytes following acute ischemic stroke. Neurol Sci 40, 1877–1885 (2019). https://doi.org/10.1007/s10072-019-03919-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-019-03919-y