Abstract
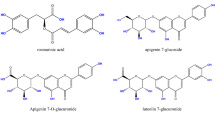
Antimicrobial effects of Melipona beecheii honey have been attributed to diverse factors, in this sense, certain components such as proteins and phenolics could explain relevant aspects of its antimicrobial activity. The aim of this study was to evaluate the antibacterial activity of phenolic and protein extracts from M. beecheii honey against two bacterial pathogens: Staphylococcus aureus and Escherichia coli. With respect to phenolic content, HPLC analysis allowed the identification of phenolic acids like chlorogenic acid, caffeic acid, and flavonoids like catechin, myricetin, quercetin and apigenin. On the other hand, seven bands with molecular weight from 7.6 to 95 kDa were detected in protein extract by SDS-PAGE system. It was determined the antibacterial activity of both extracts, with MICs lower than 145 µg/mL and 60 µg/mL for the phenolic and protein extracts respectively. These results indicate that phenolic and protein components of M. beecheii honey contribute significantly to the antibacterial activity.




Similar content being viewed by others
References
Alvarez-Suarez M, Giampieri F, Brenciani A, Mazzoni L, Gasparrini M, González-Paramás A, Santos-Buelga C, Morroni G, Simoni S, Forbes-Hernández T, Afrin S, Giovanetti E, Battino M. Apis mellifera vs Melipona beecheii Cuban polifloral honeys: a comparison based on their physicochemical parameters, chemical composition and biological properties. LWT Food Sci. Technol. 87: 272-279 (2018)
Asgary S, Naderi GH, Askari N. Protective effect of flavonoids against red blood cell hemolysis by free radicals. Exp. Clin. Cardiol. 10: 88-90 (2005)
Blasa M, Candiracci M, Accorsi A, Piacentini MP, Piatti E. Honey flavonoids as protection agents against oxidative damage to human red blood cells. Food Chem. 104: 1635-1640 (2007)
Bogdanov S. Nature and origin of the antibacterial substances in honey. LWT Food Sci. Technol. 30: 748-753 (1997)
Boorn KL, Khor YY, Sweetman E, Tan F, Heard TA, Hammer KA. Antimicrobial activity of honey from the stingless bee Trigona carbonaria determined by agar diffusion, agar dilution, broth microdilution and time-kill methodology. J. Appl. Microbiol. 108: 1534-1543 (2010)
Bouarab-Chibane L, Forquet V, Lantéri P, Clément Y, Léonard-Akkari L, Oulahal N, Degraeve P, Bordes C. Antibacterial properties of polyphenols: characterization and QSAR (quantitative structure–activity relationship) models. Front. Microbiol. 10: 829 (2019)
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle protein-dye binding. Anal. Biochem. 72: 248-254 (1976)
Brudzynski K, Sjaarda C. Antibacterial compounds of Canadian honeys target bacterial cell wall inducing phenotype changes, growth inhibition and cell lysis that resemble action of β-lactam antibiotics. PLoS ONE 9: e106967 (2014)
Chan-Rodríguez D, Ramón-Sierra J, Lope-Ayora J, Sauri-Duch E, Cuevas-Glory L, Ortiz-Vázquez E. Antibacterial properties of honey produced by Melipona beecheii and Apis mellifera against foodborn microorganisms. Food Sci. Biotechnol. 21: 905-909 (2012)
de Queiroz Pimentel RB, da Costa CA, Albuquerque PM, Junior SD. Antimicrobial activity and rutin identification of honey produced by the stingless bee Melipona compressipes manaosensis and commercial honey. BMC Complement. Altern. Med. 13: 151 (2013)
Ferreres F, Tomás-Barberán FA, Soler C, García-Viguera C, Ortiz A, Tomás-Lorente F. A simple extractive technique for honey flavonoid HPLC analysis. Apidologie 25: 21-30 (1994)
Ganske F, Bornscheuer UT. Growth of Escherichia coli, Pichia pastoris and Bacillus cereus in the presence of the ionic liquids [BMIM] [BF4] and [BMIM] [PF6] and organic solvents. Biotechnol. Lett. 28: 465-469 (2006)
Giorgi A, Madeo M, Baumgartner J, Lozzia GC. The relationships between phenolic content, pollen diversity, physicochemical information and radical scavenging activity in honey. Molecules 16: 336-347 (2011)
Hsu CH, Chen C, Jou ML, Lee AYL, Lin YC, Yu YP, Huang WT, Wu SH. Structural and DNA-binding studies on the bovine antimicrobial peptide, indolicidin: evidence for multiple conformations involved in binding to membranes and DNA. Nucleic Acids Res. 33: 4053-4064 (2005)
Irish J, Carter DA, Blair SE, Heard TA. Antibacterial activity of honey from the Australian stingless bee Trigona carbonaria. Int. J. Antimicrob. Agents 32: 89-90 (2008)
Kanakis CD, Tarantilis PA, Polissiou MG, Diamantoglou S, Tajmir-Riahi HA. DNA interaction with naturally occurring antioxidant flavonoids quercetin, kaempferol, and delphinidin. J. Biomol. Struct. Dyn. 22: 719-724 (2005)
Kono Y. Apparent antibacterial activity of catalase: role of lipid hydroperoxide contamination. J. Biochem. 117: 42-46 (1995)
Kwakman PH, de Boer L, Ruyter-Spira CP, Creemers-Molenaar T, Helsper J, Vandenbroucke-Grauls CM, Zaat S, te Velde AA. Medical-grade honey enriched with antimicrobial peptides has enhanced activity against antibiotic-resistant pathogens. Eur. J. Clin. Microbiol. 30: 251-257 (2011a)
Kwakman PH, te Velde AA, de Boer L, Vandenbroucke-Grauls CM, Zaat SA. Two major medicinal honeys have different mechanisms of bactericidal activity. PLoS ONE 6: e17709 (2011b)
Miorin PL, Levy NC, Custodio AR, Bretz WA, Marcucci MC. Antibacterial activity of honey and propolis from Apis mellifera and Tetragonisca angustula against Staphylococcus aureus. J. Appl. Microbiol. 95: 913-920 (2003)
Nishio EK, Ribeiro JM, Oliveira AG, Andrade CGTJ, Proni EA, Kobayashi RKT, Nakazato G. Antibacterial synergic effect of honey from two stingless bees: Scaptotrigona bipunctata Lepeletier, 1836, and S. postica Latreille, 1807. Scientific reports 6: 21641 (2016)
Ortiz-Vázquez E, Cuevas-Glory L, Zapata-Baas G, Martínez-Guevara J, Ramón-Sierra J. Which bee honey components contribute to its antimicrobial activity? A review. Afr. J. Microbiol. Res. 7: 5758-5765 (2013)
Park SC, Lee JR, Kim JY, Hwang I, Nah JW, Cheong H, Park Y, Hahm KS. Pr-1, a novel antifungal protein from pumpkin rinds. Biotechnol. Lett. 32: 125-130 (2010)
Peterson GL. A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal. Biochem. 83: 346-356 (1977)
Puupponen-Pimiä R, Nohynek L, Meier C, Kähkönen M, Heinonen M, Hopia A, Oksman-Caldentey KM. Antimicrobial properties of phenolic compounds from berries. J. Appl. Microbiol. 90: 494-507 (2001)
Ramón-Sierra J, Ruiz-Ruiz J, Ortiz-Vázquez E. Electrophoresis characterisation of protein as a method to establish the entomological origin of stingless bee honeys. Food Chem. 183: 43-48 (2015)
Schneider T, Kruse T, Wimmer R, Wiedemann I, Sass V, Pag U, Jansen A, Nielsen AK, Mygind PH, Raventós DS, Neve S, Ravn B, Bonvin AM, De Maria L, Andersen AS, Gammelgaard LK, Sahl H-G, Kristensen H.H. Plectasin, a fungal defensin, targets the bacterial cell wall precursor Lipid II. Science 328: 1168-1172 (2010)
Schwalbe R, Steele-Moore L, Goodwin AC. Antimicrobial susceptibility testing protocols. CRC Press, Boca Raton (2007)
Temaru E, Shimura S, Amano K, Karasawa T. Antibacterial activity of honey from stingless honeybees (Hymenoptera; Apidae; Meliponinae). Pol. J. Microbiol. 56: 281-285 (2007)
Valadez-Moctezuma E, Kahl G. Huellas de ADN en genomas de plantas (Teorías y Protocolos de Laboratorio). 1st ed. Mundi-Prensa México, SA de CV, México (2000)
Vit P, Medina M, Enríquez ME. Quality standards for medicinal uses of Meliponinae honey in Guatemala, Mexico and Venezuela. Bee World 85: 2-5 (2004)
Xu J, Zhao X, Wang X, Zhao Z, Du Y. Oligochitosan inhibits Phytophthora capsici by penetrating the cell membrane and putative binding to intracellular targets. Pestic. Biochem. Physiol. 88: 167-175 (2007)
Yadav SP, Ahmad A, Pandey BK, Verma R, Ghosh JK. Inhibition of lytic activity of Escherichia coli toxin hemolysin E against human red blood cells by a leucine zipper peptide and understanding the underlying mechanism. Biochemistry 47: 2134-2142 (2008)
Zainol MI, Yusoff KM, Yusof MY. Antibacterial activity of selected Malaysian honey. BMC Complement. Altern. Med. 13: 129 (2013)
Acknowledgements
The authors would like to thank Dr. Luis Cuevas for providing the stingless bee honey used in this study, National Council for Science and Technology (CONACYT, México) Grant of Basic Science CB-221624 and National Technological Institute of Mexico (TecNM) Grant No. 6748.18-P for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10068_2020_744_MOESM1_ESM.tiff
Figure S1. Effect of methanol (15% v/v) in the growth of cultures of E. coli (a) and S. aureus, the kinetics of bacterial growth showed a minimal effect of inhibition in presence of methanol (TIFF 190 kb)
Rights and permissions
About this article
Cite this article
Ramón-Sierra, J., Martínez-Guevara, J.L., Pool-Yam, L. et al. Effects of phenolic and protein extracts from Melipona beecheii honey on pathogenic strains of Escherichia coli and Staphylococcus aureus. Food Sci Biotechnol 29, 1013–1021 (2020). https://doi.org/10.1007/s10068-020-00744-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-020-00744-4