Abstract
Objectives
To analyze clinical characteristics, risk factors, pathogen distribution, and prognostic markers in primary Sjögren’s syndrome (pSS) patients with severe pneumonia (SP) compared to those without severe pneumonia (NSP).
Methods
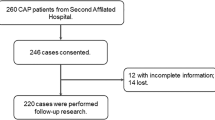
This case–control study included 24 hospitalized pSS patients with SP and 96 NSP at the first affiliated hospital of Soochow university from June 2014 to May 2023. Data encompassing demographics, comorbidities, treatments, and laboratory results were retrospectively collected. Univariate and multivariate regression analyses, ROC curves, and statistical analyses using SPSS 23.0 assessed risk factors. The study retrospectively analyzed clinical features and risk factors, highlighting distinct parameters between pSS patients with and without SP.
Results
Marked differences were observed in several parameters: pSS activity(P < 0.001), white blood cell (P = 0.043), lymphocyte (P < 0.001), neutrophils (P = 0.042), C-reactive protein (P = 0.042), and CD8+ T cell (P = 0.017). Notably, lymphocyte count and SS activity demonstrated robust discrimination ability (AUC > 0.85). C-reactive protein (CRP), procalcitonin, CD4+ T cell, and IgA showed significant associations with SP; higher CRP levels correlated with increased risk, while lower CD4+ T cell and IgA levels associated with increased risk. SS activity significantly impacted outcomes. Various biomarkers exhibited diverse discriminatory abilities but lacked strong predictive associations with outcomes.
Conclusion
pSS patients with SP exhibited higher disease activity and altered immune profiles compared to those NSP. Lymphocyte count and SS activity emerged as robust discriminators. Higher CRP levels correlated with increased risk of SP, while lower CD4+T cell and IgA levels associated with increased risk. SS activity significantly impacted patient outcomes.
Key Points • pSS patients with SP exhibited higher disease activity and altered immune profiles compared to those NSP. • Lymphocyte count and SS activity emerged as robust discriminators. • Higher CRP levels correlated with increased risk of SP, while lower CD4+ T cell and IgA levels associated with decreased risk. • SS activity significantly impacted patient outcomes. |


Similar content being viewed by others
Data availability
The data with regard to this article will be shared on reasonable request to the corresponding author.
References
Andréasson K, Ohlsson B, Mandl T (2016) Elevated levels of faecal calprotectin in primary Sjögren’s syndrome is common and associated with concomitant organic gastrointestinal disease. Arthritis Res Ther 18:9. https://doi.org/10.1186/s13075-015-0907-8
Auteri S, Alberti ML, Fernández ME et al (2021) Occult primary Sjögren syndrome in patients with interstitial pneumonia with autoimmune features. Respir Med 182:106405. https://doi.org/10.1016/j.rmed.2021.106405
Dong X, Gao YL, Lu Y, Zheng Y (2021) Characteristics of primary Sjögren’s syndrome related lymphocytic interstitial pneumonia. Clin Rheumatol 40(2):601–612. https://doi.org/10.1007/s10067-020-05236-8
Heus A, Arends S, Van Nimwegen JF, Stel AJ, Nossent GD, Bootsma H (2020) Pulmonary involvement in primary Sjögren’s syndrome, as measured by the ESSDAI. Scand J Rheumatol 49(1):38–46. https://doi.org/10.1080/03009742.2019.1634221
Işık ÖÖ, Yazıcı A, Çefle A (2022) The respiratory manifestations in patients with primary Sjögren’s syndrome: is interstitial lung disease related to disease activity. Turk J Med Sci 52(5):1737–1743. https://doi.org/10.55730/1300-0144.5513
Goulabchand R, Makinson A, Morel J et al (2022) Hospitalizations for infections in primary Sjögren’s syndrome patients: a nationwide incidence study. Ann Med 54(1):2672–2680. https://doi.org/10.1080/07853890.2022.2126517
Luo J, Xu S, Lv Y et al (2019) Clinical features and potential relevant factors of renal involvement in primary Sjögren’s syndrome. Int J Rheum Dis 22(2):182–190. https://doi.org/10.1111/1756-185X.13429
Bjordal O, Norheim KB, Rødahl E, Jonsson R, Omdal R (2020) Primary Sjögren’s syndrome and the eye. Surv Ophthalmol 65(2):119–132. https://doi.org/10.1016/j.survophthal.2019.10.004
Manfrè V, Cafaro G, Riccucci I et al (2020) One year in review 2020: comorbidities, diagnosis and treatment of primary Sjögren’s syndrome. Clin Exp Rheumatol 38 Suppl 126(4):10–22
Shiboski CH, Shiboski SC, Seror R et al (2017) 2016 American College of Rheumatology/European League Against Rheumatism classification criteria for primary Sjögren’s syndrome: a consensus and data-driven methodology involving three international patient cohorts. Arthritis Rheumatol 69(1):35–45. https://doi.org/10.1002/art.39859
Zhou X, Li H, Wang N, Jin Y, He J (2023) Respiratory infection risk in primary Sjögren’s syndrome complicated with interstitial lung disease: a retrospective study. Clin Rheumatol. https://doi.org/10.1007/s10067-023-06803-5
Roca F, Dominique S, Schmidt J et al (2017) Interstitial lung disease in primary Sjögren’s syndrome. Autoimmun Rev 16(1):48–54. https://doi.org/10.1016/j.autrev.2016.09.017
Palm O, Garen T, Berge Enger T et al (2013) Clinical pulmonary involvement in primary Sjogren’s syndrome: prevalence, quality of life and mortality–a retrospective study based on registry data. Rheumatology (Oxford) 52(1):173–179. https://doi.org/10.1093/rheumatology/kes311
Ramos-Casals M, Solans R, Rosas J et al (2008) Primary Sjögren syndrome in Spain: clinical and immunologic expression in 1010 patients. Medicine (Baltimore) 87(4):210–219. https://doi.org/10.1097/MD.0b013e318181e6af
Yazisiz V, Arslan G, Ozbudak IH et al (2010) Lung involvement in patients with primary Sjögren’s syndrome: what are the predictors. Rheumatol Int 30(10):1317–1324. https://doi.org/10.1007/s00296-009-1152-8
Mathai SC, Danoff SK (2016) Management of interstitial lung disease associated with connective tissue disease. BMJ 352:h6819. https://doi.org/10.1136/bmj.h6819
Barnes H, Holland AE, Westall GP, Goh NS, Glaspole IN (2018) Cyclophosphamide for connective tissue disease-associated interstitial lung disease. Cochrane Database Syst Rev 1(1):CD010908. https://doi.org/10.1002/14651858.CD010908.pub2
Cheng Y, Wang TY, Yuan HL et al (2021) Nocardia infection in nephrotic syndrome patients: three case studies and a systematic literature review. Front Cell Infect Microbiol 11:789754. https://doi.org/10.3389/fcimb.2021.789754
Wang H, Aloe C, Wilson N, Bozinovski S (2019) G-CSFR antagonism reduces neutrophilic inflammation during pneumococcal and influenza respiratory infections without compromising clearance. Sci Rep 9(1):17732. https://doi.org/10.1038/s41598-019-54053-w
Liampas A, Parperis K, Erotocritou MF et al (2023) Primary Sjögren syndrome-related peripheral neuropathy: a systematic review and meta-analysis. Eur J Neurol 30(1):255–265. https://doi.org/10.1111/ene.15555
Delalande S, de Seze J, Fauchais AL et al (2004) Neurologic manifestations in primary Sjögren syndrome: a study of 82 patients. Medicine (Baltimore) 83(5):280–291. https://doi.org/10.1097/01.md.0000141099.53742.16
Gong B, Zheng L, Lu Z et al (2021) Mesenchymal stem cells negatively regulate CD4+ T cell activation in patients with primary Sjögren syndrome through the miRNA‑125b and miRNA‑155 TCR pathway. Mol Med Rep 23(1). https://doi.org/10.3892/mmr.2020.11681
Carvalho CN, Carmo do RF, Duarte AL, Carvalho AA, Leão JC, Gueiros LA (2016) IL-17A and IL-17F polymorphisms in rheumatoid arthritis and Sjögren’s syndrome. Clin Oral Investig 20(3):495–502. https://doi.org/10.1007/s00784-015-1540-2
Balmforth D, Harky A, Adams B et al (2019) Is there a role for biomarkers in thoracic aortic aneurysm disease. Gen Thorac Cardiovasc Surg 67(1):12–19. https://doi.org/10.1007/s11748-017-0855-0
Geng YD, Chen YR, Jin J, Wang XD, Zhang S, Li DJ (2019) Prognostic value of D-dimer in patients with diffuse large B-cell lymphoma: a retrospective study. Curr Med Sci 39(2):222–227. https://doi.org/10.1007/s11596-019-2023-5
An Q, Zhao J, Zhu X et al (2022) Exploiting the role of T cells in the pathogenesis of Sjögren’s syndrome for therapeutic treatment. Front Immunol 13:995895. https://doi.org/10.3389/fimmu.2022.995895
Yang M, Akbar U, Mohan C (2019) Curcumin in autoimmune and rheumatic diseases. Nutrients 11(5):1004. https://doi.org/10.3390/nu11051004
Joy GM, Arbiv OA, Wong CK et al (2023) Prevalence, imaging patterns and risk factors of interstitial lung disease in connective tissue disease: a systematic review and meta-analysis. Eur Respir Rev 32(167):220210. https://doi.org/10.1183/16000617.0210-2022
Tanaka Y (2023) A review of Janus kinase inhibitors for the treatment of Covid-19 pneumonia. Inflamm Regen 43(1):3. https://doi.org/10.1186/s41232-022-00253-3
Tang X, Feng YM, Ni JX et al (2021) Early use of corticosteroid may prolong SARS-CoV-2 shedding in non-intensive care unit patients with COVID-19 pneumonia: a multicenter, single-blind, randomized control trial. Respiration 100(2):116–126. https://doi.org/10.1159/000512063
Tang Q, Chen Q, Li Y, Wang Z (2022) Association between glucocorticoids and mortality in patients with severe pneumonia: a systematic review and meta-analysis based on randomized controlled trials. Comput Math Methods Med 2022:1191205. https://doi.org/10.1155/2022/1191205
Lin W, Xin Z, Zhang J et al (2022) Interstitial lung disease in primary Sjögren’s syndrome. BMC Pulm Med 22(1):73. https://doi.org/10.1186/s12890-022-01868-5
Luppi F, Sebastiani M, Sverzellati N, Cavazza A, Salvarani C, Manfredi A (2020) Lung complications of Sjogren syndrome. Eur Respir Rev 29(157):200021. https://doi.org/10.1183/16000617.0021-2020
Bowman SJ, Fox R, Dörner T et al (2022) Safety and efficacy of subcutaneous ianalumab (VAY736) in patients with primary Sjögren’s syndrome: a randomised, double-blind, placebo-controlled, phase 2b dose-finding trial. Lancet 399(10320):161–171. https://doi.org/10.1016/S0140-6736(21)02251-0
Seror R, Ravaud P, Bowman SJ et al (2010) EULAR Sjogren’s syndrome disease activity index: development of a consensus systemic disease activity index for primary Sjogren’s syndrome. Ann Rheum Dis 69(6):1103–1109. https://doi.org/10.1136/ard.2009.110619
Acknowledgements
The authors would like to thank all the participants of this study who made this research possible.
Funding
This study was financially supported by the National Natural Science Foundation of China (82101893), the First Affiliated Hospital of Soochow University BOXI Clinical Research Project (BXLC016), Fujian provincial health technology project (2023GGA008), and the Gusu District Health Talent Training Project (GSWS2022073).
Author information
Authors and Affiliations
Contributions
BC and ZLG drafted the manuscript and performed most of the data collection. QY, HL, and HS carried out the data collection and data analysis and revised the paper. YG and XML conceived the study and participated in its design and coordination and interpreted the data. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
All procedures involving human participants conformed to the ethical guidelines of the institutional and national research committee and the 1964 Declaration of Helsinki. The study was approved by the hospital ethics committee (approval ID:2021–023).
Disclosures
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cai, B., Guo, Z., Yan, Q. et al. Clinical features and risk factors of primary Sjögren’s syndrome complicated with severe pneumonia: a case–control study. Clin Rheumatol 43, 1665–1674 (2024). https://doi.org/10.1007/s10067-024-06942-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-024-06942-3