Abstract
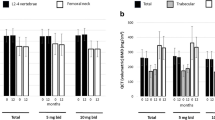
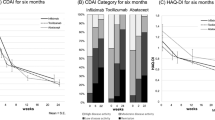
This study aims to investigate the effect of iguratimod, a novel disease-modifying antirheumatic drug, alone or combined with methotrexate (MTX), on the serum levels of regulators of bone remodeling (receptor activator of nuclear factor-κB ligand (RANKL), osteoprotegerin (OPG), and Dickkopf-1 (DKK-1)) and bone remodeling markers (C-telopeptide of type I collagen (CTX-I) and procollagen type I N-terminal propeptide (PINP)) in patients with rheumatoid arthritis (RA). Patients with RA were treated with iguratimod, MTX, or their combination for 12 months. Serum samples were collected before treatment and 6 and 12 months afterwards. RANKL, OPG, DKK-1, CTX-I, and PINP levels were measured, and radiographic progression was assessed. The serum RANKL levels decreased after treatment for 6 and 12 months with iguratimod (median: baseline 565.00 pmol/L vs. 6 months 411.00 pmol/L vs. 12 months 212.00 pmol/L), MTX (median: baseline 562.50 pmol/L vs. 6 months 399.50 pmol/L vs. 12 months 163.50 pmol/L), and their combination (median: baseline 971.00 pmol/L vs. 6 months 272.50 pmol/L vs. 12 months 241.50 pmol/L). Combination therapy showed greater effects 6 months post-treatment compared to single-drug therapy. PINP levels increased significantly 12 months post-treatment with all therapies, but only the combination therapy led to decreased CTX-I levels. OPG and DKK-1 levels showed no significant changes. The three treatments showed no significant differences in radiographic progression. Iguratimod could stimulate bone formation and regulate the RANKL/RANK/OPG system rather than DKK-1levels. Its effects are comparable to those of MTX, and combination therapy showed stronger effects.

Similar content being viewed by others
References
Smolen JS, Aletaha D, McInnes IB (2016) Rheumatoid arthritis. Lancet. doi:10.1016/S0140-6736(16)30173-8
Fardellone P, Sejourne A, Paccou J, Goeb V (2014) Bone remodelling markers in rheumatoid arthritis. Mediat Inflamm 2014:484280. doi:10.1155/2014/484280
Roux C (2011) Osteoporosis in inflammatory joint diseases. Osteoporosis Int 22(2):421–433. doi:10.1007/s00198-010-1319-x
Amarasekara DS, Yu J, Rho J (2015) Bone loss triggered by the cytokine network in inflammatory autoimmune diseases. J Immunol Res 2015:832127. doi:10.1155/2015/832127
Schett G, Gravallese E (2012) Bone erosion in rheumatoid arthritis: mechanisms, diagnosis and treatment. Nat Rev Rheumatol 8(11):656–664. doi:10.1038/nrrheum.2012.153
Diarra D, Stolina M, Polzer K, Zwerina J, Ominsky MS, Dwyer D, Korb A, Smolen J, Hoffmann M, Scheinecker C, van der Heide D, Landewe R, Lacey D, Richards WG, Schett G (2007) Dickkopf-1 is a master regulator of joint remodeling. Nat Med 13(2):156–163. doi:10.1038/nm1538
Kearns AE, Khosla S, Kostenuik PJ (2008) Receptor activator of nuclear factor kappaB ligand and osteoprotegerin regulation of bone remodeling in health and disease. Endocr Rev 29(2):155–192. doi:10.1210/er.2007-0014
Boyle WJ, Simonet WS, Lacey DL (2003) Osteoclast differentiation and activation. Nature 423(6937):337–342. doi:10.1038/nature01658
van Tuyl LH, Voskuyl AE, Boers M, Geusens P, Landewe RB, Dijkmans BA, Lems WF (2010) Baseline RANKL:OPG ratio and markers of bone and cartilage degradation predict annual radiological progression over 11 years in rheumatoid arthritis. Ann Rheum Dis 69(9):1623–1628. doi:10.1136/ard.2009.121764
Krishnan V, Bryant HU, Macdougald OA (2006) Regulation of bone mass by Wnt signaling. J Clin Invest 116(5):1202–1209. doi:10.1172/JCI28551
Daoussis D, Andonopoulos AP (2011) The emerging role of Dickkopf-1 in bone biology: is it the main switch controlling bone and joint remodeling? Semin Arthritis Rheu 41(2):170–177. doi:10.1016/j.semarthrit.2011.01.006
Wang SY, Liu YY, Ye H, Guo JP, Li R, Liu X, Li ZG (2011) Circulating Dickkopf-1 is correlated with bone erosion and inflammation in rheumatoid arthritis. J Rheumatol 38(5):821–827. doi:10.3899/jrheum.100089
Chopin F, Garnero P, le Henanff A, Debiais F, Daragon A, Roux C, Sany J, Wendling D, Zarnitsky C, Ravaud P, Thomas T (2008) Long-term effects of infliximab on bone and cartilage turnover markers in patients with rheumatoid arthritis. Ann Rheum Dis 67(3):353–357. doi:10.1136/ard.2007.076604
Kuriyama K, Higuchi C, Tanaka K, Yoshikawa H, Itoh K (2002) A novel anti-rheumatic drug, T-614, stimulates osteoblastic differentiation in vitro and bone morphogenetic protein-2-induced bone formation in vivo. Biochem Bioph Res Co 299(5):903–909
Gan K, Yang L, Xu L, Feng X, Zhang Q, Wang F, Tan W, Zhang M (2016) Iguratimod (T-614) suppresses RANKL-induced osteoclast differentiation and migration in RAW264.7 cells via NF-kappaB and MAPK pathways. Int Immunopharmacol 35:294–300. doi:10.1016/j.intimp.2016.03.038
Luo Q, Sun Y, Liu W, Qian C, Jin B, Tao F, Gu Y, Wu X, Shen Y, Xu Q (2013) A novel disease-modifying antirheumatic drug, iguratimod, ameliorates murine arthritis by blocking IL-17 signaling, distinct from methotrexate and leflunomide. J Immunol 191(10):4969–4978. doi:10.4049/jimmunol.1300832
Wei Y, Sun X, Hua M, Tan W, Wang F, Zhang M (2015) Inhibitory effect of a novel antirheumatic drug T-614 on the IL-6-induced RANKL/OPG, IL-17, and MMP-3 expression in synovial fibroblasts from rheumatoid arthritis patients. Biomed Res Int 2015:214683. doi:10.1155/2015/214683
Hara M, Abe T, Sugawara S, Mizushima Y, Hoshi K, Irimajiri S, Hashimoto H, Yoshino S, Matsui N, Nobunaga M, Nakano S (2007) Efficacy and safety of iguratimod compared with placebo and salazosulfapyridine in active rheumatoid arthritis: a controlled, multicenter, double-blind, parallel-group study. Mod Rheumatol 17(1):1–9. doi:10.1007/s10165-006-0542-y
Ishiguro N, Yamamoto K, Katayama K, Kondo M, Sumida T, Mimori T, Soen S, Nagai K, Yamaguchi T, Hara M, Iguratimod-Clinical Study G (2013) Concomitant iguratimod therapy in patients with active rheumatoid arthritis despite stable doses of methotrexate: a randomized, double-blind, placebo-controlled trial. Mod Rheumatol 23(3):430–439. doi:10.1007/s10165-012-0724-8
Duan XW, Zhang XL, Mao SY, Shang JJ, Shi XD (2015) Efficacy and safety evaluation of a combination of iguratimod and methotrexate therapy for active rheumatoid arthritis patients: a randomized controlled trial. Clin Rheumatol 34(9):1513–1519. doi:10.1007/s10067-015-2999-6
Xia Z, Lyu J, Hou N, Song L, Li X, Liu H (2015) Iguratimod in combination with methotrexate in active rheumatoid arthritis : therapeutic effects. Z Rheumatol. doi:10.1007/s00393-015-1641-y
Xu S, Wang Y, Lu J, Xu J (2012) Osteoprotegerin and RANKL in the pathogenesis of rheumatoid arthritis-induced osteoporosis. Rheumatol Int 32(11):3397–3403. doi:10.1007/s00296-011-2175-5
Ellabban AS, Kamel SR, Ahmed SS, Osman AM (2012) Receptor activator of nuclear factor kappa B ligand serum and synovial fluid level. A comparative study between rheumatoid arthritis and osteoarthritis. Rheumatol Int 32(6):1589–1596. doi:10.1007/s00296-011-1831-0
Swierkot J, Gruszecka K, Matuszewska A, Wiland P (2015) Assessment of the effect of methotrexate therapy on bone metabolism in patients with rheumatoid arthritis. Arch Immunol Ther Ex 63(5):397–404. doi:10.1007/s00005-015-0338-x
Hensvold AH, Joshua V, Li W, Larkin M, Qureshi F, Israelsson L, Padyukov L, Lundberg K, Defranoux N, Saevarsdottir S, Catrina AI (2015) Serum RANKL levels associate with anti- citrullinated protein antibodies in early untreated rheumatoid arthritis and are modulated following methotrexate. Arthritis Res Ther 17:239. doi:10.1186/s13075-015-0760-9
Boumans MJ, Thurlings RM, Yeo L, Scheel-Toellner D, Vos K, Gerlag DM, Tak PP (2012) Rituximab abrogates joint destruction in rheumatoid arthritis by inhibiting osteoclastogenesis. Ann Rheum Dis 71(1):108–113. doi:10.1136/annrheumdis-2011-200198
Seror R, Boudaoud S, Pavy S, Nocturne G, Schaeverbeke T, Saraux A, Chanson P, Gottenberg JE, Devauchelle-Pensec V, Tobon GJ, Mariette X, Miceli-Richard C (2016) Increased Dickkopf-1 in recent-onset rheumatoid arthritis is a new biomarker of structural severity. Data from the ESPOIR cohort. Scientific reports 6:18421. doi:10.1038/srep18421
Glantschnig H, Hampton RA, Lu P, Zhao JZ, Vitelli S, Huang L, Haytko P et al (2010) Generation and selection of novel fully human monoclonal antibodies that neutralize Dickkopf-1 (DKK-1) inhibitory function in vitro and increase bone mass in vivo. J Biol Chem 285(51):40135–40147. doi:10.1074/jbc.M110.166892
Briot K, Rouanet S, Schaeverbeke T, Etchepare F, Gaudin P, Perdriger A, Vray M, Steinberg G, Roux C (2015) The effect of tocilizumab on bone mineral density, serum levels of Dickkopf-1 and bone remodeling markers in patients with rheumatoid arthritis. Joint Bone Spine 82(2):109–115. doi:10.1016/j.jbspin.2014.10.015
Garnero P (2014) New developments in biological markers of bone metabolism in osteoporosis. Bone 66:46–55. doi:10.1016/j.bone.2014.05.016
Acknowledgements
This study was supported by grants from the Science and Technology Ministry “12th Five-Year Plan” to support science and technology of China (No. 2014BAI07B00). We thank Professor Bo Li and Professor Pinghui Sun from the Department of Epidemiology and Biostatistics, Jilin University School of Public Health for assistance with statistical analysis. We also appreciate the anonymous referees for their valuable suggestions and questions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None.
Rights and permissions
About this article
Cite this article
Wang, X., Ma, C., Li, P. et al. Effects of iguratimod on the levels of circulating regulators of bone remodeling and bone remodeling markers in patients with rheumatoid arthritis. Clin Rheumatol 36, 1369–1377 (2017). https://doi.org/10.1007/s10067-017-3668-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-017-3668-8