Abstract
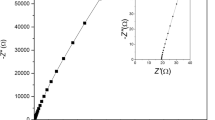
Super capacitors have emerged as a promising substitute for batteries and conventional capacitors which have played a key role as energy storage devices. The continuous concerns over green and low-cost concepts have motivated the consideration of natural materials as they are non-toxic and low cost. On the other hand, the realization of the dangers of liquid electrolytes has promoted the attention on gel polymer electrolytes to be used for devices. The prime objective of the present study is fabricating an electrochemical double-layer capacitor (EDLC) which is a type of super capacitor make using natural graphite and a gel polymer electrolyte. The electrolyte having the composition 0.5 PVdF-Co-HFP:0.70 MgTF:1 EC:1 DEC showed an optimum room temperature conductivity of 3.45 × 10−3 S cm−1. It was purely an ionic conductor having improved amorphous nature. EDLCs were fabricated with two identical natural graphite electrodes. Evaluation of EDLC was carried out using cyclic voltammetry, electrochemical impedance spectroscopy, and galvanostatic charge-discharge test. The proposed electrode/electrolyte combination seemed to be suitable for the application in EDLCs. Specific capacitance values obtained are satisfactory to carry forward further investigations to improve performance.












Similar content being viewed by others
References
Liew CW, Ramesh S (2014) Comparing triflate and hexafluorophosphate anions of ionic liquids in polymer electrolytes for super capacitor applications. Materials 7(5):4019–4033
Li J, Cheng X, Shashurin A, Keidar M (2012) Review of electrochemical capacitors based on carbon nano tubes and graphene. Graphene 1(01):1–13
Ramya R, Sivasubramanian R, Sangaranarayan MV (2013) Conducting polymers based electrochemical super capacitors – progress and prospects. Electrochim Acta 101:109–129
Nandhini R, Mini PA, Avinash B, Nair SV, Subramanian KRV (2012) Super capacitor electrodes using nano scale activated carbon from graphite by ball milling. Mater Lett 87:165–168
Li HQ, Wang YG, Wang CX, Xia YY (2008) A competitive candidate material for aqueous super capacitors : high surface area graphite. J Power Sources 185(2):1557–1562
Pandey GP, Hashmi SA, Kumar Y (2010) Performance studies of activated charcoal based electrical double layer capacitors with ionic liquid gel polymer electrolytes. Energy Fuels 24(12):6644–6652
Bandaranayake CM, Samarakkody GS, Perera KS, Vidanapathirana KP (2016) Variation of performance of dye sensitized solar cells with the salt concentration of the electrolyte. J Electrochem Energy Convers Storage 13:0110071–0110074
Osman Z, Samin SM, Othman L, Mid Isa KB (2012) Ionic transport in PMMA – NaCF3SO3 gel polymer electrolyte. Adv Mater Res 545:259–263
Kumar GG, Munichandraiah N (2000) A gel polymer electrolyte for Mg triflate. Solid State Ionics 128(1-4):203–210
Harankahawa N, Weerasinghe S, Vidanapathirana K, Perera K (2017) Investigation of a pseudo capacitor with polyacrylonitrile based gel polymer electrolytes. J Electrochem Sci Technol 8(1):1–8
Ramesh S, Chai MF (2007) Conductivity dielectric behavior and FTIR studies of high molecular weight poly (vinyl chloride)-lithium triflate polymer electrolytes. Mater Sci Eng B 139(2-3):240–245
Zainol NH, Samin SM, Othman L, Mid Isa KB, Chong WG, Osman Z (2013) Magnesium ion based gel polymer electrolytes : ionic conduction and infrared spectroscopy studies. Int J Electrochem Sci 8:3602–3614
Priya WLM, Suthanthiraraj SA (2013) Preparation and characterization of polymer electrolytes containing PVdF co HFP and zinc triflate. Chem Sci Trans 2(4):1232–1237
Rajendran S, Bama VS (2010) A study on the effect of various plasticizers in poly (vinylacetate) – poly (methylmethacrylate) based gel polymer electrolyte. J Nano Crys Solids 356(50-51):2764–2768
Wang H, Pilon L (2012) Physical interpretation of cyclic voltammetry for measuring electric double layer capacitances. Electrochim Acta 64:130–139
Prabaharan SRS, Vimala R, Zainal Z (2006) Nanostructured meso porous carbon as electrodes for super capacitors. J Power Sources 161(1):730–736
Kim BK, Sy S, Yu A, Zhang J (2015) Electrochemical super capacitors for energy storage and conversion. Hand Book of Clean Energy Systems. John Wiley and Sons Ltd
Du C, Pang N (2006) Super capacitors using carbon nanotubes films by electrophoretic deposition. J Power Sources 160(2):1487–1494
Wang W, Guo S, Penchev M, Ruiz I, Bozhilov KN, Yan D, Ozkan M, Ozkan CS (2013) Three dimensional few layer graphene and carbon nao tube foam architectures for high fidelity super capacitors. Nano Energy 2(2):294–303
Das S, Ghosh A (2017) Solid polymer electrolyte based on PVdF-HFP and ionic liquid embedded with TiO2 nanoparticle for electric double layer capacitor (EDLC) application. J Electrochem Soc 164(13):F1348–F1353
Tey JP, Careem MA, Yarmo MA, Arof AK (2016) Durian shell based activated carbon electrode for EDLCs. Ionics 22(7):1209–1216
Senthilkumar ST, Selvan RK, Melo JS, Sanjeeviraja C (2013) High performance solid state electric double layer capacitor from redox mediated gel polymer electrolyte and renewable tamarind fruit shell derived porous carbon. Appl Mater Interfaces 5(21):10541–10550
Funding
This study was financially sponsored by Wayamba University of Sri Lanka via the research grant, SRHDC/RP/04/17/01. In addition, authors wish to acknowledge the assistance extended by National Research Council, Sri Lanka (17-006) and National Science Foundation, Sri Lanka (RG/2017/BS/02, RG/2015/EQ/07).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Karunarathne, K.A.J.K., Perera, K.S., Vidanapathirana, K.P. et al. Fabrication and evaluation of an electrochemical double-layer capacitor with natural graphite electrodes and magnesium trifluoromethanesulfonate–based gel polymer electrolyte. J Solid State Electrochem 23, 2165–2171 (2019). https://doi.org/10.1007/s10008-019-04309-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-019-04309-2