Abstract
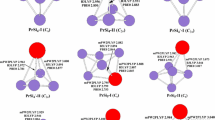
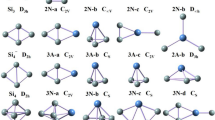
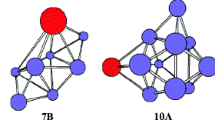
The neutral PrSi n (n = 12–21) species considering various spin configurations were systematically studied using PBE0 and B3LYP schemes in combination with relativistic small-core potentials (ECP28MWB) for Pr atoms and cc-pVTZ basis set for Si atoms. The total energy, growth-pattern, equilibrium geometry, relative stability, hardness, charge transfer, and magnetic moments are calculated and discussed. The results reveal that when n < 20, the ground-state structure of PrSi n evaluated to be prolate clusters. Starting from n = 20, the ground-state structures of PrSi n are evaluated to be endohedral cagelike clusters. Although the relative stabilities based on various binding energies and different functional is different from each other, the consensus is that the PrSi13, PrSi16, PrSi18, and PrSi20 are more stable than the others, especially the PrSi20. Analyses of hardness show that introducing Pr into Si n (n = 12–21) elevates the photochemical sensitivity, especially for PrSi20. Calculated result of magnetic moment and charge transfer shows that the 4f electrons of Pr in the clusters are changed, especially in endohedral structures such as PrSi20, in which one electron transfers from 4f to 5d orbital. That is, the 4f electron of Pr in the clusters participates in bonding. The way to participate in bonding is that a 4f electron transfers to 5d orbital. Although the 4f electron of Pr atom participates in bonding, the total magnetic moment of PrSi n is equal to that of isolated Pr atom. The charge always transfers from Pr atom to Si n cluster for the ground state structures of PrSin (n = 12–19), but charge transfer is reverse for n ≥ 20. The largest charge transfer for endohedral structure reveals that the bonding between Pr and Si n is ionic in nature and very strong. The fullerenelike structure of PrSi20 is the most stable among all of these clusters and can act as the building blocks for novel functional nanotubes.






Similar content being viewed by others
References
Avaltroni F, Steinmann SN, Corminboeuf C (2012) Phys Chem Chem Phys 14:14842–14849
Tam NM, Tai TB, Ngan VT, Nguyen MT (2013) J Phys Chem A 117:6867–6882
Lin L, Yang JC (2015) J Mol Model 21(155):1–13
Ohara M, Miyajima K, Pramann A, Nakajima A, Kaya K (2002) J Chem Phys A 106:3702–3705
Koyasu K, Atobe J, Furuse S, Nakajima A (2008) J Chem Phys 129(214301):1–7
Grubisic A, Ko YJ, Wang H, Bowen KH (2009) J Am Chem Soc 131:10783–10790
Li JR, Wang GH, Yao CH, Mu YW, Wan JG, Han M (2009) J Chem Phys 130(164514):1–9
Guo LJ, Zhao GF, Gu YZ, Liu X, Zeng Z (2008) Phys Rev B 77(195417):1–8
Wang JG, Zhao JJ, Ma L, Wang BL, Wang GH (2007) Phys Lett A 367:335–344
Kenyon AJ (2005) Semicond Sci Technol 20:R65–R84
Hou LY, Yang JC, Liu YM (2016) J Mol Model 22(193):1–10
Peng Q, Shen J (2008) J. Chem. Phys. 128(084711):1–11
Zhao GF, Sun JM, Gu YZ, Wang YX (2009) J Chem Phys 131:114312-1–114312-7
Li CG, Pan LJ, Shao P, Ding LP, Feng HT, Luo DB, Liu B (2015) Theor Chem Accounts 134(34):1–11
Liu TG, Zhao GF, Wang YX (2011) Phys Lett A 375:1120–1127
Liu TG, Zhang WQ, Li YL (2014) Front Phys 9:210–218
Zhao RN, Ren ZY, Guo P, Bai JT, Zhang CH, Han JG (2006) J Phys Chem A 110:4071–4079
Zhao RN, Han JG, Bai JT, Liu FY, Sheng LS (2010) Chem Phys 372:89–95
Zhao RN, Han JG, Bai JT, Sheng LS (2010) Chem Phys 378:82–87
Cao TT, Zhao LX, Feng XJ, Lei YM, Luo YH (2009) J Mol Struct 895:148–155
Wang HQ, Li HF (2014) RSC Adv 4:29782–29793
Zhao RN, Han JG (2014) RSC Adv 4:64410–64418
Kumar V, Singh AK, Kawazoe Y (2006) Phys Rev B 74(125411):1–5
Yang JC, Wang J, Hao YR (2015) Theor Chem Accounts 134(81):1–11
Xie XH, Hao DS, Liu YM, Yang JC (2015) Comput Theor Chem 1074:1–8
Xie XH, Hao DS, Yang JC (2015) Chem Phys 461:11–19
Becke AD (1993) J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Adamo C, Barone V (1999) J Chem Phys 110:6158–6170
Woon DE, Dunning Jr TH (1993) J Chem Phys 98:1358–1371
Cao X, Dolg M (2002) J Mol Struct THEOCHEM 581:139–147
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, et al. (2010) Gaussian 09 revision C.01. Gaussian Inc, Wallingford
Yang JC, Feng YT, Xie XH, Wu HW (2016) Theor Chem Accounts 135(204):1–12
Zhu XL, Zeng XC, Lei YA, Pan B (2004) J Chem Phys 120:8985–8995
Grossman JC, Mitáš L (1995) Phys Rev Lett 74:1323–1326
Nigam S, Majumder C, Kulshreshtha SK (2006) J Chem Phys 125(074303):1–11
Yoo S, Zeng XC (2005) J Chem Phys 123(164303):1–6
Yoo S, Zeng XC (2006) J. Chem. Phys. 124(054304):1–6
Baerends EJ, Gritsenko OV, van Meer R (2013) 15:(39)16408-16425
Acknowledgments
This study was economically supported by the National Natural Science Foundation of China (Grant No. 21263010), by the Inner Mongolia Natural Science Foundation (Grant No. 2015MS0216), and by the Program for Innovative Research Team in University of the Inner Mongolia Autonomous Region (Grant no. NMGIRT-A-1603).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Feng, Y., Yang, J. Stability and electronic properties of praseodymium-doped silicon clusters PrSin (n = 12–21). J Mol Model 23, 180 (2017). https://doi.org/10.1007/s00894-017-3352-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-017-3352-6