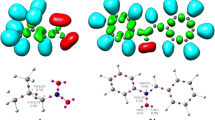
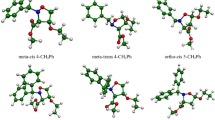
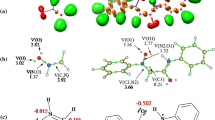
Abstract
The mechanism of the [3+2] cycloaddition (32CA) reaction of C-phenyl-N-methylnitrone with ethyl trifluoroacetoacetate has been theoretically studied at the MPWB1K/6-311G(d,p) level. This 32CA reaction, in which the enol form of the β-keto ester participates as the ethylene component, takes place with complete ortho regioselectivity and exo stereoselectivity. The presence of the CF3 group in the β-position in the enol acetate accelerates the 32CA reaction, but it does not modify the regioselectivity, which is controlled by the presence of the ester group. While ortho regioselectivity is reproduced by the MPWB1K calculations, the endo selectivity is not. The inclusion of solvent effects slightly decreases the reactivity but does not modify the gas phase selectivities. Analysis of the DFT global reactivity indices and the Parr functions in reagents provide a rationalization for the participation of ethyl trifluoroacetoacetate and the regioselectivity in this zw-type 32CA reaction.









Similar content being viewed by others
References
Gothelf KV, Jorgensen KA (1999) Asymmetric catalysis of 1,3-dipolar cycloaddition reactions–the concepts of activation and induction of asymmetry. Chem Rev 82:327–350
Karlsson S, Hogberg HE (2001) Asymmetric 1,3-dipolar cycloadditions for the construction of enantiomerically pure heterocycles. A review. Org Prep Proced Int 33:105–172
Piperno A, Giofre SV, Iannazzo D, Romeo R, Romeo G, Chiacchio U, Rescifina A, Piotrowska DG (2010) Synthesis of C-4’-truncated phosphonated carbocyclic 2’-Oxa-3’-azanucleosides as antiviral agents. J Org Chem 75:2798–2805
Domingo LR, Sáez JA (2009) Understanding the mechanism of polar Diels–Alder reactions. Org Biomol Chem 7:3576–3583
Domingo LR, Emamian SR (2014) Understanding the mechanisms of [3+2] cycloaddition reactions. The pseudoradical versus the zwitterionic mechanism. Tetrahedron 70:1267–1273
Padwa A, Pearson WH (2002) Synthetic applications of 1,3-dipolar cycloaddition chemistry toward heterocycles and natural products, vol 59. Wiley, Hoboken
Domingo LR, Aurell MJ, Pérez P (2014) A DFT analysis of the participation of TACs in zw-type [3+2] cycloaddition reactions. Tetrahedron 70:4519–4525
Bravo P, Bruché L, Fronza G, Zecchi G (1992) A cycloadditive route to trifluoromethyl-substituted aminoalcohols. Tetrahedron 48:9775–9788
Tanaka K, Mori T, Mitsuhashi K (1989) Novel ring transformation of 3-benzoylisoxazolidines into 2-hydroxydihydrofurans - N-O cleavage vs 1,3-dipolar cycloreversion. Chem Lett 18:1115–1118
Bravo P, Bruché L, Crucianelli M, Farina A, Valdo Meille S, Merli A, Seresini P (1996) Asymmetric 1.3-Dipolar cycloadditions of nitrile oxides and nitrones with fluorosubstituted chiral vinyl sulfoxides. J Chem Res Synop 8:348–349
Tanaka K, Mitsuhashi K (1987) Synthesis of trifluoromethyl azoles using building-blocks. J Synth Org Chem Jpn 45:269–283
Sobhi C, Nacereddine AK, Djerourou A, Aurell MJ, Domingo LR (2012) The role of the trifluoromethyl group in reactivity and selectivity in polarcycloaddition reactions. A DFT study. Tetrahedron 68:8457–8462
BonnetDelpon D, Begue JP, Lequeux T, Ourevitch M (1996) Trifluoromethylalkenes in cycloaddition reactions. Tetrahedron 52:59–70
Camps F, Coll J, Messeguer A, Roca A (1977) NMR-study of keto-enol equilibrium of ethyl gamma, gamma, gamma-trifluoroacetoacetate and its reaction with water and alcohols. Tetrahedron 33:1637–1640
Zhao Y, Truhlar DG (2004) Hybrid meta density functional theory methods for thermochemistry, thermochemical kinetics, and noncovalent interactions: The MPW1B95 and MPWB1K models and comparative assessments for hydrogen bonding and van der Waals interactions. J Phys Chem A 108:6908–6918
Hehre WJ, Radom L, Schleyer PR, Pople JA (1986) Ab initio molecular orbital theory, vol 33. Wiley, New York
Soto-Delgado J, Aizman A, Contreras R, Domingo LR (2012) On the catalytic effect of water in the intramolecular Diels–Alder reaction of quinone systems: a theoretical study. Molecules 17:13687–13703
Rhyman L, Ramasami P, Joule JA, Saez JA, Domingo LR (2013) Understanding the formation of [3+2] and [2 + 4] cycloadducts in the Lewis acid catalysed reaction between methyl glyoxylate oxime and cyclopentadiene: a theoretical study. RSC Adv 3:447–457
Domingo LR, Sáez JA, Arnó M (2014) A DFT study on the NHC catalysed Michael addition of enols to α, β-unsaturated acyl-azoliums. A base catalysed C–C bond-formation step. Org Biomol Chem 12:895–904
Schlegel HB (1982) Optimization of equilibrium geometries and transition structures. J Comput Chem 3:214–218
Schlegel HB (1995) Geometry optimization on potential energy surfaces. Mod Electron Struct Theory 2:459–500
Tomasi J, Persico M (1994) Molecular-interactions in solution—an overview of methods based on continuous distributions of the solvent. Chem Rev 94:2027–2094
Simkin B, Sheikhet II (1995) Quantum chemical and statistical theory of solutions: a computational approach. Horwood, London
Mennucci B, Cances E, Tomasi J (1997) Evaluation of solvent effects in isotropic and anisotropic dielectrics and in ionic solutions with a unified integral equation method: theoretical bases, computational implementation, and numerical applications. J Phys Chem B 101:10506–10517
Cossi M, Barone V, Cammi R, Tomasi J (1996) Ab initio study of solvated molecules: a new implementation of the polarizable continuum model. Chem Phys Lett 255:327–335
Barone V, Cossi M, Tomasi J (1998) Geometry optimization of molecular structures in solution by the polarizable continuum model. J Comput Chem 19:404–417
Reed AE, Weinstock RB, Weinhold F (1985) Natural-population analysis. J Chem Phys 83:735–746
Reed AE, Curtiss LA, Weinhold F (1988) Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem Rev 88:899–926
Frisch M (2009) Gaussian 09, revision A. 02. Gaussian. Inc, Wallingford
Parr RG, Szentpály L, Liu S (1999) Electrophilicity index. J Am Chem Soc 121:1922–1924
Parr RG, Pearson RG (1983) Absolute hardness: companion parameter to absolute electronegativity. J Am Chem Chem Soc 105:7512–7516
Parr RG, Yang W (1989) Density functional theory of atoms and molecules. Oxford University Press, New York
Domingo LR, Chamorro E, Pérez P (2008) Understanding the reactivity of captodative ethylenes in polar cycloaddition reactions. A theoretical study. J Org Chem 73:4615–4624
Domingo LR, Pérez P (2011) The nucleophilicity N index in organic chemistry. Org Biomol Chem 9:7168–7175
Benchouk W, Mekelleche SM, Silvi B, Aurell MJ, Domingo LR (2011) Understanding the kinetic solvent effects on the 1,3-dipolar cycloaddition of benzonitrile N-oxide: a DFT study. J Phys Org Chem 24:611–618
Domingo LR, Pérez P, Aurell MJ, Sáez JA (2012) Understanding the bond formation in hetero-Diels-Alder reactions. An ELF analysis of the reaction of nitroethylene with dimethylvinylamine. Curr Org Chem 16:2343–2351
Zhao Y, Truhlar DG (2008) The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor Chem Accounts 120:215–241
Chai J-D, Head-Gordon M (2008) Long-range corrected hybrid density functionals with damped atom–atom dispersion corrections. Phys Chem Chem Phys 10:6615–6620
Geerlings P, De Proft F, Langenaeker W (2003) Conceptual density functional theory. Chem Rev 103:1793–1873
Ess DH, Jones GO, Houk KN (2006) Conceptual, qualitiative, and quantitative theories of 1,3-dipolar and Diels-Alder cycloadditions used in synthesis. Adv Synth Catal 348:2337–2361
Domingo LR, Pérez P, Sáez JA (2013) Understanding the regioselectivity in hetero Diels-Alder reactions. An ELF analysis of the reaction between nitrosoethylene and 1-vinylpyrrolidine. Tetrahedron 69:107–114
Domingo LR, Pérez P, Sáez JA (2013) Understanding the local reactivity in polar organic reactions through electrophilic and nucleophilic Parr functions. RSC Adv 3:1486–1494
Acknowledgments
L.R.D thanks to Ministerio de Economía y Competitividad of the Spanish Government, project CTQ2013-45646-P, for financial support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supporting information
MPWB1K/6-311G(d,p) total energies, and enthalpies, entropies, and Gibbs free energies, computed at 384.15 K and 1 atm in toluene, of the stationary points involved in the 32CA reaction between nitrone 20 and enol acetate 22. B3LYP, MPWB1K, M062X, ωB97X-D, and MP2 total energies and energetic differences between the stereoisomeric ortho TSs involved in the 32CA reaction of nitrone 20 with enol acetate 22. MPWB1K/6-311+G(d,p) total and relative gas phase energies of the stationary points involved in the 32CA reaction between nitrone 20 and enol acetate 22. MPWB1K/6-311+G(d,p) geometries of the TSs involved in the 32CA reaction of nitrone 20 with enol acetate 22. Top view of the geometries of the stereoisomeric ortho TSs involved in the 32CA reaction of nitrone 20 with enol acetate 22. MPWB1K/6-311G(d,p) total energies, enthalpies, entropies, and Gibbs free energies, computed at 384.15 K and 1 atm in toluene, of the stationary points involved in the 32CA reaction between nitrone 20 and enol acetate 24. Complete reference [29. (DOC 777 kb)
Rights and permissions
About this article
Cite this article
Layeb, H., Nacereddine, A.K., Djerourou, A. et al. Understanding the role of the trifluoromethyl group in reactivity and regioselectivity in [3+2] cycloaddition reactions of enol acetates with nitrones. A DFT study. J Mol Model 21, 104 (2015). https://doi.org/10.1007/s00894-015-2658-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-015-2658-5