Abstract
Objectives
The present study aimed to identify proteins obtained from pulp tissue and correlate with each clinical diagnosis (healthy pulp, inflamed pulp, and necrotic pulp).
Materials and methods
A total of forty-five molars were used. Three biological replicas were evaluated. Lysis and sonication were used for protein extraction. Protein quantification was assessed by using the Bradford technique, and shotgun proteome analysis was performed by nanoUPLC-MSE using a Synapt G2 mass spectrometer. Mass spectra data were processed using the Waters PLGS software, and protein identification was done using the human Uniprot database appended to the PLGS search engine.
Results
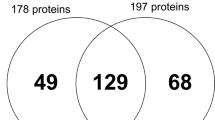
A total of 123 different proteins were identified in all evaluated pulp conditions. Among these, 66 proteins were observed for healthy pulp, 66 for inflamed pulp, and 91 for necrotic pulp. Most protein identification was related to immune response, multi-organism process, platelet activation, and stress in inflamed pulp samples compared to healthy pulp. Proteins related to cellular component organization or biogenesis, developmental process, growth, immune response, multi-organism process, response to stimulus, signaling, stress, and transport were identified in cases of apical periodontitis compared to inflamed pulp.
Conclusions
The progression of the disease to inflamed pulp promoted a high abundance of proteins related to the immune system and stress. Comparing the necrotic pulp with inflamed pulp conditions, a high abundance of proteins was noticed related to metabolism, transport, and response between organisms.
Clinical relevance
This finding may assist in future studies of new markers, understanding of tissue engineering, and development of future products.



Similar content being viewed by others
References
Kawashima N, Okiji T (2016) Odontoblasts: specialized hard-tissue-forming cells in the dentin-pulp complex. Congenit Anom (Kyoto) 56:144–153. https://doi.org/10.1111/cga.12169
Goldberg M, Smith AJ (2004) Cells and extracellular matrices of dentin and pulp: a biological basis for repair and tissue engineering. Crit Rev Oral Biol Med 15:13–27
Abbott PV, Yu C (2007) A clinical classification of the status of the pulp and the root canal system. Aust Dent J 52:S17–S31
Farges JC, Alliot-Licht B, Renard E, Ducret M, Gaudin A, Smith AJ, Cooper PR (2015) Dental pulp defence and repair mechanisms in dental caries. Mediators Inflamm 2015:230251–230216. https://doi.org/10.1155/2015/230251
Yu C, Abbott PV (2007) An overview of the dental pulp: its functions and responses to injury. Aust Dent J 52:S4–S16
Izumi T, Kobayashi I, Okamura K, Sakai H (1995) Immunohistochemical study on the immunocompetent cells of the pulp in human non-carious and carious teeth. Arch Oral Biol 40:609–614
Bletsa A, Berggreen E, Fristad I, Tenstad O, Wiig H (2006) Cytokine signalling in rat pulp interstitial fluid and transcapillary fluid exchange during lipopolysaccharide-induced acute inflammation. J Physiol 573:225–236. https://doi.org/10.1113/jphysiol.2006.104711
Mejare IA, Axelsson S, Davidson T, Frisk F, Hakeberg M, Kvist T, Norlund A, Petersson A, Portenier I, Sandberg H, Tranaeus S, Bergenholtz G (2012) Diagnosis of the condition of the dental pulp: a systematic review. Int Endod J 45:597–613. https://doi.org/10.1111/j.1365-2591.2012.02016.x
Nair PN (2004) Pathogenesis of apical periodontitis and the causes of endodontic failures. Crit Rev Oral Biol Med 15:348–381
Siqueira JF Jr, Rocas IN (2007) Bacterial pathogenesis and mediators in apical periodontitis. Braz Dent J 18:267–280
Pecora GE, Pecora CN (2015) A new dimension in endo surgery: micro endo surgery. J Conserv Dent 18:7–14. https://doi.org/10.4103/0972-0707.148864
Atmeh AR, Watson TF (2016) Root dentine and endodontic instrumentation: cutting edge microscopic imaging. Interface Focus 6:20150113. https://doi.org/10.1098/rsfs.2015.0113
Aminsobhani M, Khalatbari MS, Meraji N, Ghorbanzadeh A, Sadri E (2016) Evaluation of the fractured surface of five endodontic rotary instruments: a metallurgical study. Iran Endod J 11:286–292. https://doi.org/10.22037/iej.2016.6
Yared G (2008) Canal preparation using only one Ni-Ti rotary instrument: preliminary observations. Int Endod J 41:339–344. https://doi.org/10.1111/j.1365-2591.2007.01351.x
Prado MC, Leal F, Simao RA, Gusman H, do Prado M (2017) The use of auxiliary devices during irrigation to increase the cleaning ability of a chelating agent. Restor Dent Endod 42:105–110. https://doi.org/10.5395/rde.2017.42.2.105
Rezende TM, Lima SM, Petriz BA, Silva ON, Freire MS, Franco OL (2013) Dentistry proteomics: from laboratory development to clinical practice. J Cell Physiol 228:2271–2284. https://doi.org/10.1002/jcp.24410
Rechenberg DK, Galicia JC, Peters OA (2016) Biological markers for pulpal inflammation: a systematic review. PLoS One 11:e0167289. https://doi.org/10.1371/journal.pone.0167289
Yue W, Kim S, Jung HS, Lee JM, Lee S, Kim E (2019) Differential protein expression in human dental pulp: comparison of healthy, inflamed, and traumatic pulp. J Clin Med 8. https://doi.org/10.3390/jcm8081234
Provenzano JC, Siqueira JF Jr, Rocas IN, Domingues RR, Paes Leme AF, Silva MR (2013) Metaproteome analysis of endodontic infections in association with different clinical conditions. PLoS One 8:e76108. https://doi.org/10.1371/journal.pone.0076108
Eckhardt A, Jagr M, Pataridis S, Miksik I (2014) Proteomic analysis of human tooth pulp: proteomics of human tooth. J Endod 40:1961–1966. https://doi.org/10.1016/j.joen.2014.07.001
Eckhard U, Marino G, Abbey SR, Tharmarajah G, Matthew I, Overall CM (2015) The human dental pulp proteome and n-terminome: levering the unexplored potential of semitryptic peptides enriched by TAILS to identify missing proteins in the human proteome project in underexplored tissues. J Proteome Res 14:3568–3582. https://doi.org/10.1021/acs.jproteome.5b00579
Belda-Ferre P, Williamson J, Simon-Soro A, Artacho A, Jensen ON, Mira A (2015) The human oral metaproteome reveals potential biomarkers for caries disease. Proteomics 15:3497–3507. https://doi.org/10.1002/pmic.201400600
Costa MG, Pazzini CA, Pantuzo MC, Jorge ML, Marques LS (2013) Is there justification for prophylactic extraction of third molars? A systematic review. Braz Oral Res 27:183–188
Hupp JR (2007) Legal implications of third molar removal. Oral Maxillofac Surg Clin North Am 19:129–36, viii. https://doi.org/10.1016/j.coms.2006.11.008
Lopes V, Mumenya R, Feinmann C, Harris M (1995) Third molar surgery: an audit of the indications for surgery, post-operative complaints and patient satisfaction. Br J Oral Maxillofac Surg 33:33–35
Huang GT, Sonoyama W, Chen J, Park SH (2006) In vitro characterization of human dental pulp cells: various isolation methods and culturing environments. Cell Tissue Res 324:225–236. https://doi.org/10.1007/s00441-005-0117-9
Murad AM, Rech EL (2012) NanoUPLC-MSE proteomic data assessment of soybean seeds using the Uniprot database. BMC Biotechnol 12:82. https://doi.org/10.1186/1472-6750-12-82
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Alfenas CF, Mendes TAO, Ramos HJO, Bruckner FP, Antunes HS, Rocas IN, Siqueira JF Jr, Provenzano JC (2017) Human exoproteome in acute apical abscesses. J Endod 43:1479–1485. https://doi.org/10.1016/j.joen.2017.04.019
Petriz BA, Almeida JA, Gomes CP, Pereira RW, Murad AM, Franco OL (2015) NanoUPLC/MS(E) proteomic analysis reveals modulation on left ventricle proteome from hypertensive rats after exercise training. J Proteomics 113:351–365. https://doi.org/10.1016/j.jprot.2014.10.010
Li GZ, Vissers JP, Silva JC, Golick D, Gorenstein MV, Geromanos SJ (2009) Database searching and accounting of multiplexed precursor and product ion spectra from the data independent analysis of simple and complex peptide mixtures. Proteomics 9:1696–1719. https://doi.org/10.1002/pmic.200800564
Silva JC, Denny R, Dorschel CA, Gorenstein M, Kass IJ, Li GZ, McKenna T, Nold MJ, Richardson K, Young P, Geromanos S (2005) Quantitative proteomic analysis by accurate mass retention time pairs. Anal Chem 77:2187–2200. https://doi.org/10.1021/ac048455k
Jagr M, Eckhardt A, Pataridis S, Foltan R, Mysak J, Miksik I (2016) Proteomic analysis of human tooth pulp proteomes-comparison of caries-resistant and caries-susceptible persons. J Proteomics 145:127–136. https://doi.org/10.1016/j.jprot.2016.04.022
Jagr M, Eckhardt A, Pataridis S, Miksik I (2012) Comprehensive proteomic analysis of human dentin. Eur J Oral Sci 120:259–268. https://doi.org/10.1111/j.1600-0722.2012.00977.x
Moule AJ, Li H, Bartold PM (1995) Donor variability in the proliferation of human dental pulp fibroblasts. Aust Dent J 40:110–114
Renard E, Gaudin A, Bienvenu G, Amiaud J, Farges JC, Cuturi MC, Moreau A, Alliot-Licht B (2016) Immune cells and molecular networks in experimentally induced pulpitis. J Dent Res 95:196–205. https://doi.org/10.1177/0022034515612086
Fristad I, Bletsa A, Byers M (2010) Inflammatory nerve responses in the dental pulp. Endodontic Topics 17:12–41
Cooper PR, Holder MJ, Smith AJ (2014) Inflammation and regeneration in the dentin-pulp complex: a double-edged sword. J Endod 40:S46–S51. https://doi.org/10.1016/j.joen.2014.01.021
Zhang R, Cooper PR, Smith G, Nor JE, Smith AJ (2011) Angiogenic activity of dentin matrix components. J Endod 37:26–30. https://doi.org/10.1016/j.joen.2010.08.042
Loureiro C, Buzalaf MAR, Pessan JP, Moraes FRN, Pela VT, Ventura TMO, Jacinto RC (2020) Comparative analysis of the proteomic profile of the dental pulp in different conditions. A pilot study. Braz Dent J 31:319–336. https://doi.org/10.1590/0103-6440202003167
Deng R, Hao J, Han W, Ni Y, Huang X, Hu Q (2015) Gelsolin regulates proliferation, apoptosis, migration and invasion in human oral carcinoma cells. Oncol Lett 9:2129–2134. https://doi.org/10.3892/ol.2015.3002
Tsikandelova R, Mladenov P, Planchon S, Kalenderova S, Praskova M, Mihaylova Z, Stanimirov P, Mitev V, Renaut J, Ishkitiev N (2018) Proteome response of dental pulp cells to exogenous FGF8. J Proteomics 183:14–24. https://doi.org/10.1016/j.jprot.2018.05.004
Li GH, Arora PD, Chen Y, McCulloch CA, Liu P (2012) Multifunctional roles of gelsolin in health and diseases. Med Res Rev 32:999–1025. https://doi.org/10.1002/med.20231
Hahn CL, Liewehr FR (2007) Update on the adaptive immune responses of the dental pulp. J Endod 33:773–781. https://doi.org/10.1016/j.joen.2007.01.002
McHeyzer-Williams M, Okitsu S, Wang N, McHeyzer-Williams L (2011) Molecular programming of B cell memory. Nat Rev Immunol 12:24–34. https://doi.org/10.1038/nri3128
Deimling D, Hannig C, Hoth-Hannig W, Schmitz P, Schulte-Monting J, Hannig M (2007) Non-destructive visualisation of protective proteins in the in situ pellicle. Clin Oral Investig 11:211–216. https://doi.org/10.1007/s00784-007-0112-5
D'Acquisto F, Merghani A, Lecona E, Rosignoli G, Raza K, Buckley CD, Flower RJ, Perretti M (2007) Annexin-1 modulates T-cell activation and differentiation. Blood 109:1095–1102. https://doi.org/10.1182/blood-2006-05-022798
Fu H, Subramanian RR, Masters SC (2000) 14-3-3 proteins: structure, function, and regulation. Annu Rev Pharmacol Toxicol 40:617–647. https://doi.org/10.1146/annurev.pharmtox.40.1.617
Wang H, Ma D, Zhang X, Xu S, Ning T, Wu B (2018) Comparative proteomic profiling of human dental pulp stem cells and periodontal ligament stem cells under in vitro osteogenic induction. Arch Oral Biol 89:9–19. https://doi.org/10.1016/j.archoralbio.2018.01.015
Huynh AH, Veith PD, McGregor NR, Adams GG, Chen D, Reynolds EC, Ngo LH, Darby IB (2015) Gingival crevicular fluid proteomes in health, gingivitis and chronic periodontitis. J Periodontal Res 50:637–649. https://doi.org/10.1111/jre.12244
Nair PN (1997) Apical periodontitis: a dynamic encounter between root canal infection and host response. Periodontol 2000(13):121–148
Bonfoco E, Krainc D, Ankarcrona M, Nicotera P, Lipton SA (1995) Apoptosis and necrosis: two distinct events induced, respectively, by mild and intense insults with N-methyl-D-aspartate or nitric oxide/superoxide in cortical cell cultures. Proc Natl Acad Sci U S A 92:7162–7166
Chen R, Kang R, Fan XG, Tang D (2014) Release and activity of histone in diseases. Cell Death Dis 5:e1370. https://doi.org/10.1038/cddis.2014.337
Ivaska J, Pallari HM, Nevo J, Eriksson JE (2007) Novel functions of vimentin in cell adhesion, migration, and signaling. Exp Cell Res 313:2050–2062. https://doi.org/10.1016/j.yexcr.2007.03.040
D’Alessandro A, Nemkov T, Moore HB, Moore EE, Wither M, Nydam T, Slaughter A, Silliman CC, Banerjee A, Hansen KC (2016) Metabolomics of trauma-associated death: shared and fluid-specific features of human plasma vs lymph. Blood Transfus 14:185–194. https://doi.org/10.2450/2016.0208-15
Wu JQ, Jiang JH, Xu L, Liang C, Wang XJ, Bai Y (2018) Magnetic bead-based salivary peptidome profiling for accelerated osteogenic orthodontic treatments. Chin J Dent Res 21:41–49. https://doi.org/10.3290/j.cjdr.a39917
Cooper PR, Chicca IJ, Holder MJ, Milward MR (2017) Inflammation and regeneration in the dentin-pulp complex: net gain or net loss? J Endod 43:S87–S94. https://doi.org/10.1016/j.joen.2017.06.011
Byers MR, Taylor PE, Khayat BG, Kimberly CL (1990) Effects of injury and inflammation on pulpal and periapical nerves. J Endod 16:78–84. https://doi.org/10.1016/S0099-2399(06)81568-2
Bowles WR, Withrow JC, Lepinski AM, Hargreaves KM (2003) Tissue levels of immunoreactive substance P are increased in patients with irreversible pulpitis. J Endod 29:265–267. https://doi.org/10.1097/00004770-200304000-00009
Jacobi-Gresser E, Schutt S, Huesker K, Von Baehr V (2015) Methyl mercaptan and hydrogen sulfide products stimulate proinflammatory cytokines in patients with necrotic pulp tissue and endodontically treated teeth. J Biol Regul Homeost Agents 29:73–84
Tarze A, Deniaud A, Le Bras M, Maillier E, Molle D, Larochette N, Zamzami N, Jan G, Kroemer G, Brenner C (2007) GAPDH, a novel regulator of the pro-apoptotic mitochondrial membrane permeabilization. Oncogene 26:2606–2620. https://doi.org/10.1038/sj.onc.1210074
Baik JE, Choe HI, Hong SW, Kang SS, Ahn KB, Cho K, Yun CH, Han SH (2016) Human salivary proteins with affinity to lipoteichoic acid of Enterococcus faecalis. Mol Immunol 77:52–59. https://doi.org/10.1016/j.molimm.2016.07.013
Stashenko P, Teles R, D'Souza R (1998) Periapical inflammatory responses and their modulation. Crit Rev Oral Biol Med 9:498–521
Cappello P, Principe M, Bulfamante S, Novelli F (2017) Alpha-enolase (ENO1), a potential target in novel immunotherapies. Front Biosci (Landmark Ed) 22:944–959
Curciarello R, Steele A, Cooper D, MacDonald TT, Kruidenier L, Kudo T (2014) The role of galectin-1 and galectin-3 in the mucosal immune response to Citrobacter rodentium infection. PLoS One 9:e107933. https://doi.org/10.1371/journal.pone.0107933
Brito LNS, de Lemos Almeida MMR, de Souza LB, Alves PM, Nonaka CFW, Godoy GP (2018) Immunohistochemical analysis of galectins-1, -3, and -7 in periapical granulomas, radicular cysts, and residual radicular cysts. J Endod 44:728–733. https://doi.org/10.1016/j.joen.2018.01.008
Asea A (2005) Stress proteins and initiation of immune response: chaperokine activity of hsp72. Exerc Immunol Rev 11:34–45
Ricucci D, Siqueira JF Jr, Loghin S, Lin LM (2016) Pulp and apical tissue response to deep caries in immature teeth: a histologic and histobacteriologic study. J Dent. 56:19–32. https://doi.org/10.1016/j.jdent.2016.10.005
Siqueira JF Jr (2001) Aetiology of root canal treatment failure: why well-treated teeth can fail. Int Endod J 34:1–10
Ricucci D, Siqueira JF Jr (2010) Fate of the tissue in lateral canals and apical ramifications in response to pathologic conditions and treatment procedures. J Endod 36:1–15. https://doi.org/10.1016/j.joen.2009.09.038
Funding
This study was supported by Universidade Católica de Brasília (UCB), Conselho Nacional de Desenvolvimento Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo do Distrito Federal (FAPDF) and Fundação de Apoio ao Desenvolvimento do Ensino, Ciência e Tecnologia do Estado de Mato Grosso do Sul (Fundect).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The research protocol was approved by the University of Brasília human ethics committee (CEP/FS-UnB n. 018137/2015).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Silva, P.A.O., Lima, S., Freire, M.d. et al. Proteomic analysis of human dental pulp in different clinical diagnosis. Clin Oral Invest 25, 3285–3295 (2021). https://doi.org/10.1007/s00784-020-03660-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-020-03660-3