Abstract
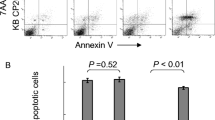
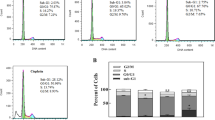
(1R,2R-diaminocyclohexane)(dihydropyrophosphato) platinum(II), also abbreviated as RRD2, belongs to a class of potent antitumor platinum cytostatics called phosphaplatins. Curiously, several published studies have suggested significant mechanistic differences between phosphaplatins and conventional platinum antitumor drugs. Controversial findings have been published regarding the role of RRD2 binding to DNA in the mechanism of its antiproliferative activity in cancer cells. This prompted us to perform detailed studies to confirm or rule out the role of RRD2 binding to DNA in its antiproliferative effect in cancer cells. Here, we show that RRD2 exhibits excellent antiproliferative activity in various cancer cell lines, with IC50 values in the low micromolar or submicromolar range. Moreover, the results of this study demonstrate that DNA lesions caused by RRD2 contribute to killing cancer cells treated with this phosphaplatin derivative. Additionally, our data indicate that RRD2 accumulates in cancer cells but to a lesser extent than cisplatin. On the other hand, the efficiency of cisplatin and RRD2, after they accumulate in cancer cells, in binding to nuclear DNA is similar. Our results also show that RRD2 in the medium, in which the cells were cultured before RRD2 accumulated inside the cells, remained intact. This result is consistent with the view that RRD2 is activated by releasing free pyrophosphate only in the environment of cancer cells, thereby allowing RRD2 to bind to nuclear DNA.
Graphical abstract





Similar content being viewed by others
Data availability
Data will be made available on request.
References
Johnstone TC, Suntharalingam K, Lippard SJ (2016) The next generation of platinum drugs: targeted Pt(II) agents, nanoparticle delivery, and Pt(IV) prodrugs. Chem Rev 116:3436–3486
Dilruba S, Kalayda GV (2016) Platinum-based drugs: past, present and future. Cancer Chemother Pharmacol 77:1103–1124
Brabec V, Hrabina O, Kasparkova J (2017) Cytotoxic platinum coordination compounds. DNA binding agents. Coord Chem Rev 351:2–31
Jung Y, Lippard SJ (2007) Direct cellular responses to platinum-induced DNA damage. Chem Rev 107:1387–1407
Babak MV, Zhi Y, Czarny B, Toh TB, Hooi L, Chow EK-H, Ang WH, Gibson D, Pastorin G (2019) Dual-targeting dual-action platinum(IV) platform for enhanced anticancer activity and reduced nephrotoxicity. Angew Chem Int Ed 58:8109–8114
Novohradsky V, Pracharova J, Kasparkova J, Imberti C, Bridgewater HE, Sadler PJ, Brabec V (2020) Induction of immunogenic cell death in cancer cells by a photoactivated platinum(IV) prodrug. Inorg Chem Front 7:4150–4159
Imberti C, Zhang P, Huang H, Sadler PJ (2020) New designs for phototherapeutic transition metal complexes. Angew Chem Int Ed 59:61–73
Bose RN, Moghaddas S, Belkacemi L, Tripathi S, Adams NR, Majmudar P, McCall K, Dezvareh H, Nislow C (2015) Absence of activation of DNA repair genes and excellent efficacy of phosphaplatins against human ovarian cancers: implications to treat resistant cancers. J Med Chem 58:8387–8401
Bose RN, Maurmann L, Mishur RJ, Yasui L, Gupta S, Grayburn WS, Hofstetter H, Salley T (2008) Non-DNA-binding platinum anticancer agents: cytotoxic activities of platinum-phosphato complexes towards human ovarian cancer cells. Proc Natl Acad Sci USA 105:18314–18319
Moghaddas S, Majmudar P, Marin R, Dezvareh H, Qi C, Soans E, Bose RN (2012) Phosphaplatins, next generation platinum antitumor agents: a paradigm shift in designing and defining molecular targets. Inorg Chim Acta 393:173–181
Belkacemi L, Atkins JL, Yang LU, Gadgil P, Sater AK, Chow DS, Bose RN, Zhang SX (2018) Phosphaplatin antitumor effect enhanced by liposomes partly via an up-regulation of PEDF in breast cancer. Anticancer Res 38:623–646
Corte-Rodríguez M, Espina M, Sierra LM, Blanco E, Ames T, Montes-Bayón M, Sanz-Medel A (2015) Quantitative evaluation of cellular uptake, DNA incorporation and adduct formation in cisplatin sensitive and resistant cell lines: comparison of different Pt-containing drugs. Biochem Pharmacol 98:69–77
Curci A, Gandin V, Marzano C, Hoeschele JD, Natile G, Margiotta N (2017) Novel kiteplatin pyrophosphate derivatives with improved efficacy. Inorg Chem 56:7482–7493
Kasparkova J, Kostrhunova H, Novohradsky V, Pracharova J, Curci A, Margiotta N, Natile G, Brabec V (2017) Anticancer kiteplatin pyrophosphate derivatives show unexpected target selectivity for DNA. Dalton Trans 46:14139–14148
Pracharova J, Saltarella T, Radosova Muchova T, Scintilla S, Novohradsky V, Novakova O, Intini FP, Pacifico C, Natile G, Ilik P, Brabec V, Kasparkova J (2015) Novel antitumor cisplatin and transplatin derivatives containing 1-methyl-7-azaindole: synthesis, characterization, and cellular responses. J Med Chem 58:847–859
Mackay FS, Woods JA, Moseley H, Ferguson J, Dawson A, Parsons S, Sadler PJ (2006) A photoactivated trans diammine platinum complex as cytotoxic as cisplatin. Chem Eur J 12:3155–3161
Woods JA, Bilton RF, Young AJ (1999) b-Carotene enhances hydrogen peroxide-induced DNA damage in human hepatocellular HepG2 cells. FEBS Lett 449:255–258
Robichova S, Slamenova D, Gabelova A, Sedlak J, Jakubikova J (2004) An investigation of the genotoxic effects of N-nitrosomorpholine in mammalian cells. Chem Biol Interact 148:163–171
Johnson NP, Butour J-L, Villani G, Wimmer FL, Defais M, Pierson V, Brabec V (1989) Metal antitumor compounds: the mechanism of action of platinum complexes. Prog Clin Biochem Med 10:1–24
Spanswick VJ, Hartley JM, Hartley JA (2010) Measurement of DNA interstrand crosslinking in individual cells using the single cell gel electrophoresis (Comet) assay. Methods Mol Biol 613:267–282
Heringova P, Woods J, Mackay FS, Kasparkova J, Sadler PJ, Brabec V (2006) Transplatin is cytotoxic when photoactivated: enhanced formation of DNA cross-links. J Med Chem 49:7792–7798
Kaminski R, Darbinyan A, Merabova N, Deshmane SL, White MK, Khalili K (2008) Protective role of Pur alpha to cisplatin. Cancer Biol Ther 7:1926–1935
Kreja L, Seidel HJ (2002) Evaluation of the genotoxic potential of some microbial volatile organic compounds (MVOC) with the comet assay, the micronucleus assay and the HPRT gene mutation assay. Mutation Res 513:143–150
Silva MJ, Costa P, Dias A, Valente M, Louro H, Boavida MG (2005) Comparative analysis of the mutagenic activity of oxaliplatin and cisplatin in the Hprt gene of CHO cells. Environ Mol Mutagenesis 46:104–115
Halámiková A, Heringová P, Kašpárková J, Intini FP, Natile G, Nemirovski A, Gibson D, Brabec V (2008) Cytotoxicity, mutagenicity, cellular uptake, DNA and glutathione interactions of lipophilic trans-platinum complexes tethered to 1-adamantylamine. J Inorg Biochem 102:1077–1089
Ames T, Price M (2019). Phosplatin Therapeutics, Inc USA
Yang L, Moghaddas S, Dezvareh H, Belkacemi L, Bark SJ, Bose RN, Do LH (2016) Insights into the anti-angiogenic properties of phosphaplatins. J Inorg Biochem 164:5–16
Acknowledgements
This work was supported by the Czech Science Foundation [grant number 21-27514S].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Prachařová, J., Kostrhunová, H., Barbanente, A. et al. The mechanism of antiproliferative activity of the oxaliplatin pyrophosphate derivative involves its binding to nuclear DNA in cancer cells. J Biol Inorg Chem 28, 669–678 (2023). https://doi.org/10.1007/s00775-023-02017-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-023-02017-x