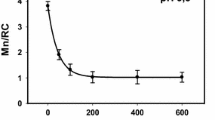
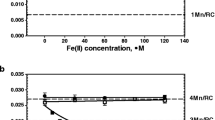
Abstract
Ca-depleted photosystem II membranes obtained by treatment with acidic buffer do not contain Ca2+ in the Mn4CaO5 cluster but contain all extrinsic proteins protecting this cluster (PSII(-Ca/low pH)). However, unlike native photosystem II, Mn cluster in PSII(-Ca/low pH) samples is available for small-sized reductants. Using this property, we investigated the substitution possibility of Mn cation(s) with Fe cation(s) to obtain a chimeric cluster in PSII(-Ca/low pH) samples containing extrinsic proteins. We found that Fe(II) cation replaces Mn cation at pH 6.5, however, PSII(-Ca/low pH) membranes with the 3Mn1Fe chimeric cluster in the oxygen-evolving complex evolve O2 with high intensity in the presence of exogenous Ca2+. The O2 evolution rate is about 80% of the same rate in PSII(-Ca/low pH) membranes.
Graphical abstract




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author, [BS], upon reasonable request.
Abbreviations
- Chl:
-
Chlorophyll
- DCBQ:
-
2,6-Dichloro-1,4-benzoquinone
- HA:
-
High-affinity Mn-binding site
- H2Q:
-
Hydroquinone
- MES:
-
2-(N-morpholino)-ethanesulfonic acid
- OEC:
-
Oxygen-evolving complex
- PSII:
-
Photosystem II
- PSII(-Ca):
-
Ca-depleted PSII
- PSII(-Ca/low pH):
-
Ca-depleted PSII membranes using low pH treatment
- PSII(-Ca/NaCl):
-
Ca-depleted PSII membranes using NaCl treatment
- PSII(-Ca/low pH/3Mn1Fe):
-
Low pH-treated PSII(-Ca) membranes with chimeric cluster 3Mn1Fe
- PSII(-Mn):
-
Mn-depleted PSII membranes
- RC:
-
Reaction center
- TMB:
-
3,3′,5,5′-Tetramethylbenzidine
References
Umena Y, Kawakami K, Shen J-R, Kamiya N (2011) Crystal structure of oxygen-evolving photosystem II at a resolution of 1.9 Å. Nature 473:55–60. https://doi.org/10.1038/nature09913
Ananyev GM, Dismukes GC (1996) Assembly of the tetra-Mn site of photosynthetic water oxidation by photoactivation: Mn stoichiometry and detection of a new intermediate. Biochemistry 35:4102–4109. https://doi.org/10.1021/bi952667h
Ono T-A, Mino H (1999) Unique binding site for Mn2+ ion responsible for reducing an oxidized YZ tyrosine in manganese-depleted photosystem II membranes. Biochemistry 38:8778–8785. https://doi.org/10.1021/bi982949s
Mino H, Asada M (2021) Location of two Mn2+ affinity sites in photosystem II detected by pulsed electron–electron double resonance. Photosynth Res. https://doi.org/10.1007/s11120-021-00885-5
Hoganson CW, Ghanotakis DF, Babcock GT, Yocum CF (1989) Mn2+ reduces Y+ in manganese-depleted photosystem II preparations. Photosynth Res 22:285–293. https://doi.org/10.1007/BF00048306
Becker K, Cormann KU, Nowaczyk MM (2011) Assembly of the water-oxidizing complex in photosystem II. J Photochem Photobiol B 104:204–211. https://doi.org/10.1016/j.jphotobiol.2011.02.005
Semin BK, Ivanov II, Rubin AB, Parak F (1995) High-specific binding of Fe(II) at the Mn-binding site in Mn-depleted PSII membranes from spinach. FEBS Lett 375:223–226. https://doi.org/10.1016/0014-5793(95)01215-Z
Semin BK, Ghirardi ML, Seibert M (2002) Blocking of electron donation by Mn(II) to YZ • following incubation of Mn-depleted photosystem II membranes with Fe(II) in the light. Biochemistry 41:5854–5864. https://doi.org/10.1021/bi0200054
Semin BK, Seibert M (2006) A carboxylic residue at the high-affinity, Mn-binding site participates in the binding of iron cations that block the site. Biochim Biophys Acta 1757:189–197. https://doi.org/10.1016/j.bbabio.2006.02.001
Semin BK, Seibert M (2004) Iron bound to the high-affinity Mn binding site of the oxygen-evolving complex shifts the pK of a component controlling electron transport via Y(Z). Biochemistry 43:6772–6782. https://doi.org/10.1021/bi036047p
Semin BK, Seibert M (2006) Flash-induced blocking of the high-affinity, Mn-binding site by iron cations: dependence on the dark interval between flashes and binary oscillations of fluorescence yield. J Phys Chem B 110:25532–25542. https://doi.org/10.1021/jp0652796
Semin BK, Lovyagina ER, Timofeev KN, Ivanov II, Rubin AB, Seibert M (2005) Iron-blocking the high-affinity Mn-binding site in photosystem II facilitates identification of the type of hydrogen bond participating in proton-coupled electron transport via YZ •. Biochemistry 44:9746–9757. https://doi.org/10.1021/bi047618w
Widdel F, Schnell S, Heising S, Ehrenreich A, Assmus B, Schink B (1993) Ferrous iron oxidation by anoxygenic phototrophic bacteria. Nature 362:834–836. https://doi.org/10.1038/362834a0
Semin BK, Seibert M (2016) Substituting Fe for two of the four Mn ions in photosystem II: effects on water-oxidation. J Bioenerg Biomembr 48:227–240. https://doi.org/10.1007/s10863-016-9651-2
Semin BK, Davletshina LN, Rubin AB (2015) Correlation between pH dependence of O2 evolution and sensitivity of Mn cations in the oxygen-evolving complex to exogenous reductants. Photosynth Res 125:95–103. https://doi.org/10.1007/s11120-015-0155-4
Semin BK, Davletshina LN, Seibert M, Rubin AB (2018) Creation of a 3Mn/1Fe cluster in the oxygen-evolving complex of photosystem II and investigation of its functional activity. J Photochem Photobiol B 178:192–200. https://doi.org/10.1016/j.jphotobiol.2017.11.016
Ono T, Inoue Y (1988) Discrete extraction of the Ca atom functional for O2 evolution in higher plant photosystem II by a simple low pH treatment. FEBS Lett 227:147–152. https://doi.org/10.1016/0014-5793(88)80886-X
Vander Meulen KA, Hobson A, Yocum CF (2002) Calcium depletion modifies the structure of the photosystem II O2-evolving complex. Biochemistry 41:958–966. https://doi.org/10.1021/bi0109414
Vander Meulen KA, Hobson A, Yocum CF (2004) Reconstitution of the photosystem II Ca2+ binding site. Biochim Biophys Acta 1655:179–183. https://doi.org/10.1016/j.bbabio.2003.08.012
Ghanotakis DF, Babcock GT (1983) Hydroxylamine as an inhibitor between Z and P680 in photosystem II. FEBS Lett 153:231–234. https://doi.org/10.1016/0014-5793(83)80154-9
Porra RJ, Thompson WA, Kriedemann PE (1989) Determination of accurate extinction coefficients and simultaneous-equations for assaying chlorophyll a and chlorophyll b extracted with 4 different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim Biophys Acta 975:384–394. https://doi.org/10.1016/S0005-2728(89)80347-0
Ono T, Inoue Y (1990) Abnormal redox reactions in photosynthetic O2- evolving centers in NaCl/EDTA-washed PS II. A dark-stable EPR multiline signal and an unknown positive charge accumulator. Biochim Biophys Acta 1020:269–277. https://doi.org/10.1016/0005-2728(90)90157-Y
Semin BK, Seibert M (2009) A simple colorimetric determination of the manganese content in photosynthetic membranes. Photosynth Res 100:45–48. https://doi.org/10.1007/s11120-009-9421-7
Semin BK, Davletshina LN, Timofeev KN, Ivanov II, Rubin AB, Seibert M (2013) Production of reactive oxygen species in decoupled, Ca2+-depleted PSII and their use in assigning a function to chloride on both sides of PSII. Photosynth Res 117:385–399. https://doi.org/10.1007/s11120-013-9870-x
Serrat FB (1998) 3,3′,5,5′-Tetramethylbenzidme for the colorimetric determination of manganese in water. Microchim Acta 129:77–80. https://doi.org/10.1007/BF01246852
Ghanotakis DF, Babcock GT, Yocum CF (1984) Structural and catalytic properties of the oxygen-evolving complex. Correlation of polypeptide and manganese release with the behavior of Z+ in chloroplasts and a highly resolved preparation of the PSII complex. Biochim Biophys Acta 765:388–398. https://doi.org/10.1016/0005-2728(84)90180-4
Xu Q, Bricker TM (1992) Structural organization of proteins on the oxidizing side of photosystem I. Two molecules of the 33-kDa manganese-stabilizing proteins per reaction center. J Biol Chem 267:25816–25821. https://doi.org/10.1016/S0021-9258(18)35683-7
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
Ghanotakis DF, Babcock GT, Yocum CF (1984) Calcium reconstitutes high rates of oxygen evolution in polypeptide depleted photosystem II preparations. FEBS Lett 167:127–130. https://doi.org/10.1016/0014-5793(84)80846-7
Miyao M, Murata N (1984) Calcium ions can be substituted for the 24-kDa polypeptide in photosynthetic oxygen evolution. FEBS Lett 168:118–120. https://doi.org/10.1016/0014-5793(84)80218-5
Furia TE (1973) CRC handbook of food additives (Vol. 1), CRC press
Kuntzleman T, Yocum CF (2005) Reduction-induced inhibition and Mn(II) release from the photosystem II oxygen-evolving complex by hydroquinone or NH2OH are consistent with a Mn(III)/Mn(III)/Mn(IV)/Mn(IV) oxidation state for the dark-adapted enzyme. Biochemistry 44:2129–2142. https://doi.org/10.1021/bi048460i
Roose JL, Frankel LK, Mummadisetti MP, Bricker TM (2016) The extrinsic proteins of photosystem II: update. Planta 243:889–908. https://doi.org/10.1007/s00425-015-2462-6
Nakatani HY (1984) Photosynthetic oxygen evolution does not require the participation of polypeptides of 16 and 24 kilodaltons. Biochem Biophys Res Commun 120:299–304. https://doi.org/10.1016/0006-291X(84)91448-7
Tomita M, Ifuku K, Sato F, Noguchi T (2009) FTIR evidence that the PsbP extrinsic protein induces protein conformational changes around the oxygen-evolving Mn cluster in photosystem II. Biochemistry 48:6318–6325. https://doi.org/10.1021/bi9006308
Zabret J, Bohn S, Schuller SK, Arnolds O, Möller M, Meier-Credo J et al (2021) Structural insights into photosystem II assembly. Nature plants 7:524–538. https://doi.org/10.1038/s41477-021-00895-0
Avramov AP, Hwang HJ, Burnap RL (2020) The role of Ca2+ and protein scaffolding in the formation of nature’s water oxidizing complex. Proc Natl Acad Sci USA 117:28036–28045. https://doi.org/10.1073/pnas.2011315117
Lubitz W, Cox CM, N, (2019) Water oxidation in photosystem II. Photosynth Res 142:105–125. https://doi.org/10.1007/s11120-019-00648-3
Shen JR (2015) The structure of photosystem II and the mechanism of water oxidation in photosynthesis. Annu Rev Plant Biol 66:23–48. https://doi.org/10.1146/annurev-arplant-050312-120129
Vinyard DJ, Ananyev GM, Dismukes GC (2013) Photosystem II: the reaction center of oxygenic photosynthesis. Annu Rev Biochem 82:577–606. https://doi.org/10.1146/annurev-biochem-070511-100425
Gates C, Ananyev G, Roy-Chowdhury S, Cullinane B, Miller M, Fromme P, Dismukes GC (2022) Why did nature choose manganese over cobalt to make oxygen photosynthetically on the earth? J Phys Chem B 126:3257–3268. https://doi.org/10.1021/acs.jpcb.2c00749
Du P, Eisenberg R (2012) Catalysts made of earth-abundant elements (Co, Ni, Fe) for water splitting: Recent progress and future challenges. Energy Environ Sci 5:6012–6021. https://doi.org/10.1039/c2ee03250c
Acknowledgements
The research was carried out as part of the Scientific Project of the State Order of the Government of Russian Federation to Lomonosov Moscow State University No. 121032500058-7
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Semin, B.К., Davletshina, L.N. High-efficiency oxygen evolution by photosystem II oxygen-evolving complex containing 3Mn per reaction center. J Biol Inorg Chem 28, 393–401 (2023). https://doi.org/10.1007/s00775-023-01987-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-023-01987-2